New drug
Aclidinium bromide for chronic obstructive pulmonary disease
- Aust Prescr 2014;37:172-9
- 13 August 2014
- DOI: 10.18773/austprescr.2014.069
Some of the views expressed in the following notes on newly approved products should be regarded as preliminary, as there may have been limited published data at the time of publication, and little experience in Australia of their safety or efficacy. However, the Editorial Executive Committee believes that comments made in good faith at an early stage may still be of value. Before new drugs are prescribed, the Committee believes it is important that more detailed information is obtained from the manufacturer's approved product information, a drug information centre or some other appropriate source.
Approved indication: chronic obstructive
pulmonary disease
Bretaris Genuair (A
Menarini)
375 microgram as dry powder for
inhalation
Australian
Medicines Handbook section 19.1.2
The treatment of chronic obstructive pulmonary disease aims to relieve symptoms and prevent deterioration. Patients whose symptoms are not relieved by short-acting bronchodilators are usually given maintenance treatment with a long-acting bronchodilator. This could be a beta agonist or an anticholinergic. Aclidinium adds to the choice of inhaled long-acting anticholinergic drugs. It is a muscarinic receptor antagonist which mediates airway smooth muscle contraction.
A multidose device is used to deliver aclidinium powder. (The metered dose is 400 microgram. This delivers 375 microgram of aclidinium bromide from the mouthpiece, which is equivalent to 322 microgram of aclidinium.) After inhalation, the forced expiratory volume in one second (FEV1) increases within 30 minutes. The peak effect is reached within three hours and bronchodilation is sustained for 12 hours. A twice-daily dose is therefore recommended. The peak plasma concentration is reached within 15 minutes and very little aclidinium reaches the systemic circulation as it is rapidly hydrolysed. Most of the metabolites are excreted in the urine, but renal impairment is unlikely to have a clinically significant effect on clearance. Similarly no dose adjustment is recommended for people with liver disease.
In a phase II trial aclidinium bromide 400 microgram (twice daily) was compared with placebo and tiotropium 18 microgram (once daily) in 30 smokers or ex-smokers with chronic obstructive pulmonary disease. Each patient inhaled each treatment for 15 days with a washout period between each course. The mean baseline FEV1 was 1.5 L. After 15 days the morning pre-dose FEV1 had increased by 143 mL with aclidinium, 107 mL with tiotropium, and decreased by 43 mL with placebo.1
A larger trial randomised 561 patients to inhale aclidinium or placebo twice daily for 12 weeks. These patients had FEV1 values between 30% and 80% of their predicted value. At the start of the study the mean FEV1 was 1.36 L, which was approximately 47% of the predicted value. The primary outcome of the trial was the change in FEV1measured just before the morning dose (trough value). After 12 weeks the trough FEV1 had increased by 62 mL with aclidinium 200 microgram and by 99 mL with 400 microgram. The mean trough FEV1 in the placebo group declined by 25 mL. Treatment with aclidinium also improved the patients' peak FEV1 significantly more than placebo.2
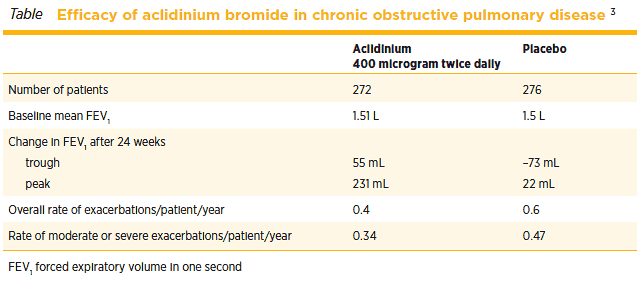
Another trial compared aclidinium 200 and 400 microgram twice daily with placebo over 24 weeks. The 828 smokers and ex-smokers in the study had FEV1 values less than 80% of the predicted value. At baseline the mean FEV1 was about 53% of the predicted value. After 24 weeks the trough FEV1 was 99 mL higher than placebo with aclidinium 200 microgram and 128 mL higher with 400 microgram. The peak FEV1 was 185 mL greater than placebo with aclidinium 200 microgram and 209 mL greater with 400 microgram (see Table). There were also improvements in symptoms, and patients taking aclidinium needed to use a short-acting bronchodilator for relief less often than patients taking placebo. As the effect of aclidinium was greater at the higher dose, 400 microgram twice daily is the recommended dose.3
Some of the adverse effects, such as dry mouth, can be predicted from the anticholinergic actions of aclidinium. Patients with narrow angle glaucoma, bladder outflow obstruction or unstable cardiac disease were excluded from the trials because of the potential for acute glaucoma, urinary retention and arrhythmia. A few patients had prolongation of the QTc interval on the ECG.3 Adverse events which were more frequent with aclidinium than with placebo included headache, nasopharyngitis and diarrhoea.
The 24-week trial showed that aclidinium has an advantage over placebo. However, most of the advantage in lung function was because of the deterioration of trough FEV1 in the placebo group. After 24 weeks of treatment with aclidinium 400 microgram twice daily the increase in trough FEV1 from baseline was 55 mL (see Table).3 While improving symptoms is an important part of the management of chronic obstructive pulmonary disease, not all patients will benefit from aclidinium. A clinically significant improvement was seen in 57% of the patients taking aclidinium compared with 41% of the placebo group. Although aclidinium reduced the rate of exacerbations, the reduction in moderate or severe exacerbations was not statistically different from placebo (see Table).3
What is needed now is a comparison of the
effectiveness of the inhaled long-acting
anticholinergic bronchodilators in chronic
obstructive pulmonary disease. The phase II study
showed some advantages for twice-daily aclidinium
over once-daily tiotropium, but the study was too
small and the duration too short to show if one drug
and device was more effective than the other.1
Aclidinium bromide is not indicated for asthma.

manufacturer did not respond to request for data

) is explained in New drugs: transparency, Vol 37 No 1, Aust Prescr 2014;37:27.
At the time the comment was prepared, information about this drug was available on the web site of the Food and Drug Administration in the USA (www.fda.gov).
At the time the comment was prepared, a scientific discussion about this drug was available on the website of the European Medicines Agency (www.emea.europa.eu).