Although it has been in use since 1951, methotrexate is the current 'gold standard' of rheumatoid arthritis treatment. It has the highest rate of continued long-term treatment, maintaining efficacy without excessive toxicity and can be used alone or in combination. Methotrexate is proven to slow radiographic progression of disease, but it takes 6-8 weeks for the onset of benefit.
Mechanism of action
Methotrexate is a folic acid antagonist cytotoxic drug. By binding to dihydrofolate reductase, methotrexate interferes with DNA synthesis and cell replication.
Maintenance dose
For maintenance, patients ordinarily take a SINGLE WEEKLY dose of 7.5-20 mg(orally, or by intramuscular or subcutaneous injection). Intramuscular or subcutaneous weekly administration of the same methotrexate dose may reduce any nausea experienced with the oral route. It is very important that patients know that they should only take methotrexate once a week. Naming the day they should take their tablet can help them remember.3,4 For example, methotrexate can be prescribed as 'Methotrexate 10 mg, take one tablet on Tuesdays ONLY'.
Co-prescription of both 2.5 mg and 10 mg tablets is not recommended, as the tablets may be confused especially by those with impaired eyesight.
Adverse effects
About 60% of patients may have mild toxicity, but less than 30% cease the drug in the first year because of adverse effects. Common adverse effects include nausea the day after the dose is taken, mouth ulcers, reversible alopecia, rash, and increased rheumatoid nodule formation. Rarer adverse effects include bone marrow suppression, hepatic fibrosis/cirrhosis (increased with alcohol consumption) and pulmonary infiltrates/allergic pneumonitis (possibly increased in smokers). Folic acid (0.5-1 mg/day) reduces gastrointestinal and mucosal adverse effects and is recommended as a concomitant prescription.
Monitoring
Patients should have a fortnightly full blood count, creatinine, and liver function tests for the first three months, monthly for three months, then six weekly thereafter once the dose is stable. Watch for changes in blood cells and monitor for 2-3 fold elevation (above the upper limit of the normal range)in liver enzymes (AST and ALT), or reduction in albumin. If, despite dosage adjustment or cessation of methotrexate, the AST or albumin are abnormal in at least five of the routine 6-weekly blood tests performed over one year, then a liver biopsy should be considered.
Contraindications
Methotrexate should not be used in patients with pre-existing bone marrow aplasia or cytopenias, immunodeficiency, severe hepatic disorders, or active infectious disease. Concomitant alcohol intake or hepatotoxic drugs are also contraindicated. Patients frequently ask about a safe level of alcohol consumption, but this has not been studied.
Drug interactions
Trimethoprim or trimethoprimsulfamethoxazole can increase bone marrow suppression, probably by an additive antifolate effect. Non-steroidal anti-inflammatory drugs (NSAIDs) and salicylates can inhibit the renal excretion of methotrexate. This is important for chemotherapeutic doses, but NSAIDs have no effect on the low doses of methotrexate used for rheumatoid arthritis and they can be cautiously co-prescribed. Patients and general practitioners must be aware of the pharmacy and prescribing software alerts that do not distinguish between low and high doses. Hepatotoxicity is potentially increased with the co-administration of azathioprine, sulfasalazine or leflunomide as part of combination therapy.
Advice in pregnancy/breastfeeding
Methotrexate was originally used as an abortifacient and is associated with congenital abnormalities. Breastfeeding is contraindicated because of neonatal immunosuppression, neutropenia and growth retardation.
|
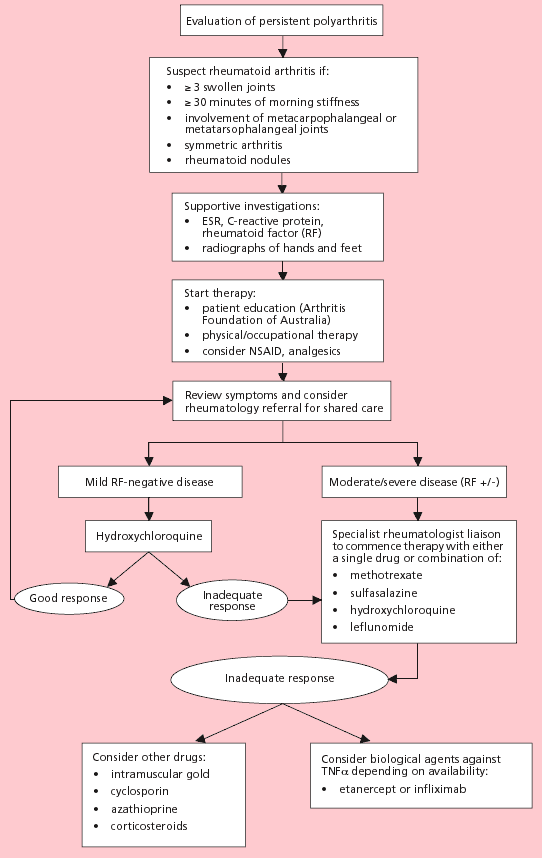
Fig. 1
Management of rheumatoid arthritis
|
|
|