SUMMARY CMI
AURORIX®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using AURORIX?
AURORIX contains the active ingredient moclobemide. AURORIX belongs to a group of medicines called antidepressants. Antidepressants are used to treat depression and work on the central nervous system.
For more information, see Section 1. Why am I using AURORIX? in the full CMI.
2. What should I know before I use AURORIX?
Do not use if you have ever had an allergic reaction to AURORIX or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use AURORIX? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with AURORIX and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use AURORIX?
- The usual dose is between 300 mg and 600 mg per day.
More instructions can be found in Section 4. How do I use AURORIX? in the full CMI.
5. What should I know while using AURORIX?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using AURORIX? in the full CMI.
6. Are there any side effects?
Less serious side effects include disturbed sleep, dizziness, nausea, and headache. Serious side effects include thoughts or talk of death or suicide, thoughts or talk of self-harm or harm to others, any recent attempts of self-harm, increase in aggressive behaviour, irritability or agitation and worsening of depression. Call your doctor or a mental health professional straight away, or go straight to the Emergency Department at your nearest hospital if you or someone you know notice any of these serious side effects. All thoughts or talk of suicide or violence must be taken seriously.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
FULL CMI
AURORIX®
Active ingredient(s): moclobemide
Consumer Medicine Information (CMI)
This leaflet provides important information about using AURORIX. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using AURORIX.
Where to find information in this leaflet:
1. Why am I using AURORIX?
2. What should I know before I use AURORIX?
3. What if I am taking other medicines?
4. How do I use AURORIX?
5. What should I know while using AURORIX?
6. Are there any side effects?
7. Product details
1. Why am I using AURORIX?
AURORIX contains the active ingredient moclobemide. AURORIX belongs to a group of medicines called antidepressants. Antidepressants are used to treat depression and work on the central nervous system. They are thought to work by their action on brain chemicals called amines, which are involved in controlling mood.
There are many different types of medicines used to treat depression. AURORIX belongs to a group of medicines called reversible inhibitors of monoamine oxidase A.
Your doctor, however, may have prescribed AURORIX for another purpose.
Ask your doctor if you have any questions about why AURORIX has been prescribed for you.
This medicine is available only with a doctor's prescription.
Do not give AURORIX to children or adolescents under 18 years of age.
The safety and effectiveness of AURORIX in this age group has not been established.
2. What should I know before I use AURORIX?
Warnings
Do not use AURORIX if:
- you are allergic to moclobemide, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
- you are suffering from severe confusion.
- you are taking:
- clomipramine
- selegiline
- bupropion
- triptans (a family of medicines commonly used to treat migraines e.g. zolmitriptan)
- pethidine
- tramadol
- dextromethorphan (often found in cough and cold medicines)
- linezolid. - you are taking other medications known as selective serotonin reuptake inhibitors or tricyclic antidepressants. Taking AURORIX with these medicines may cause a serious reaction called serotonin syndrome. This can cause a sudden increase in body temperature, high blood pressure and convulsions.
- after the expiry date (EXP) printed on the pack. It may have no effect at all or, worse, an entirely unexpected effect if you take it after the expiry date.
- the packaging is torn or shows signs of tampering or if the tablets appear damaged in some way.
If it has expired or is damaged, return the product to your pharmacist for disposal.
If you are not sure if you should be taking AURORIX, talk to your doctor.
Check with your doctor if you:
- have any other medical conditions including:
- liver disease
- high blood pressure
- a personal history or family history of bipolar disorder
- mental illness other than depression, including schizophrenia, agitation and excitation
- thyrotoxicosis (a condition of excessive thyroid hormones)
- phaeochromocytoma (a rare tumour of adrenal gland)
- rare hereditary problems of galactose intolerance, the Lapp lactose deficiency or glucose-galactose malabsorption. - take any medicines for any other condition
- are allergic to any other medicines, foods, dyes or preservatives.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Your doctor will discuss the risks and benefits of using AURORIX when pregnant and while breastfeeding.
If you have not told your doctor about any of the above, tell them before you start taking AURORIX.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with AURORIX and affect how it works.
These include:
- cimetidine
- dextropropoxyphene
- proton pump inhibitors
- serotonin agonists (e.g. buspirone, sumatriptan)
- St. John's Wort (hypericum) - containing phytotherapeutic products.
- opiates e.g. morphine, fentanyl and codeine
- adrenergics
- sibutramine
- Other antidepressant medicines that may interfere with AURORIX include: fluoxetine, fluvoxamine, paroxetine, sertraline, amitriptyline and nortriptyline, trimipramine and maprotiline, venlafaxine, clomipramine, citalopram and paroxetine.
- Metoprolol. Moclobemide may cause an additional drop in blood pressure if you are taking metoprolol.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect AURORIX.
4. How do I use AURORIX?
How much to take
- Follow the instructions provided by your doctor and use AURORIX until your doctor tells you to stop.
- The usual dose is between 300 mg and 600 mg per day.
When to take
- AURORIX should be taken morning and evening at the end of your meal.
How to take AURORIX
- Tablets should be swallowed whole with a glass of water after meals.
- You should follow your doctor's instructions carefully if changing from one antidepressant to another and report any unexpected effects if they occur.
How long to take AURORIX
For depression, the length of treatment will depend on how quickly your symptoms improve. Most antidepressants take time to work so don't be discouraged if you don't feel better right away. Some of your symptoms may improve in 1 or 2 weeks but, it can take up to 4 or 6 weeks to feel any real improvement. Even when you feel well, you will usually have to take AURORIX for several months or even longer to make sure the benefits will last. Continue taking it until your doctor tells you to stop.
If you forget to use AURORIX
AURORIX should be used regularly at the same time each day. If you miss your dose at the usual time, skip the dose you missed and take your next dose when you are meant to. Otherwise, take it as soon as you remember then go back to taking it as soon as you would normally.
Do not take a double dose to make up for the dose you missed.
If you use too much AURORIX
If you think that you have used too much AURORIX, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(Australia telephone 13 11 26) for advice, or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
Keep telephone numbers for these places handy.
Some signs and symptoms of overdose include nausea, vomiting, drowsiness, slurred speech, reduced reflexes and agitation.
5. What should I know while using AURORIX?
Things you should do
If you are being treated for depression, tell your doctor immediately if you feel your condition has worsened or if you are experiencing suicidal thoughts.
Be sure to discuss with your doctor any problems you may have and how you feel. This will help your doctor to determine the best treatment for you.
Be sure to keep all of your appointments with your doctor so that your progress can be checked.
Call your doctor straight away if you:
- become pregnant while taking AURORIX.
- for any reason, you have not taken your medicine exactly as prescribed. Otherwise, your doctor may think that it was not effective and change your treatment unnecessarily.
- feel the tablets are not helping your condition.
Remind any doctor, dentist or pharmacist you visit that you are using AURORIX.
Things you should not do
- Do not take any other medicines whether they require a prescription or not without first telling your doctor.
- Do not stop taking AURORIX or lower the dose without first checking with your doctor.
- Do not let yourself run out of medicine over the weekend or on holidays.
- Do not give this medicine to anyone else even if their symptoms seem similar to yours.
- Do not use AURORIX to treat other complaints unless your doctor says to.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how AURORIX affects you.
AURORIX causes dizziness in some people at first.
Drinking alcohol
Tell your doctor if you drink alcohol.
Although drinking alcohol is unlikely to affect your response to AURORIX, your doctor may suggest avoiding alcohol while you are being treated for depression.
Looking after your medicine
- Keep AURORIX in a cool dry place where the temperature stays below 30°C.
- A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Follow the instructions in the carton on how to take care of your medicine properly.
Keep your tablets in the blister pack until it is time to take them.
If you take the tablets out of the blister, they may not keep well.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
When to discard your medicine (as relevant)
If your doctor tells you to stop taking AURORIX, or the tablets have passed their expiry date, ask your pharmacist what to do with any tablets that are left over.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking AURORIX.
In the first week or two you may experience the following:
- sleep disturbances
- dizziness
- nausea
- headache
- dry mouth
Occasionally, the symptoms of depression may include thoughts of suicide or self-harm. These symptoms may continue or get worse during the first one to two months of treatment until the full antidepressant effect of the medicine becomes apparent. This is more likely to occur if you are a young adult i.e. under 24 years of age.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Warning signs of suicide while taking AURORIX:
| Call your doctor or a mental health professional straight away, or go straight to the Emergency Department at your nearest hospital if you or someone you know notice any of these serious side effects. All thoughts or talk of suicide or violence must be taken seriously. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Ask your doctor or pharmacist if you don't understand anything in this list.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What AURORIX contains
| Active ingredient (main ingredient) | AURORIX 150 mg - 150 mg moclobemide. AURORIX 300 mg - 300 mg moclobemide. |
| Other ingredients (inactive ingredients) | Lactose monohydrate Maize starch Povidone Sodium starch glycollate Magnesium stearate Aquacoat ECD Ethylcellulose Aqueous Dispersion NF (ARTG PI No: 1973) Macrogol 6000 Hypromellose Purified talc Titanium dioxide Yellow iron oxide (only present in AURORIX 150 mg) |
| Potential allergens | AURORIX 150 mg - Contains sugars as lactose. AURORIX 300 mg - Contains lactose. |
Do not take this medicine if you are allergic to any of these ingredients.
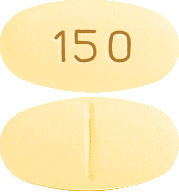
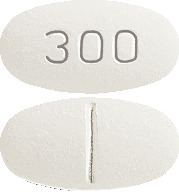
What AURORIX looks like
AURORIX comes in two strengths of tablets.
AURORIX 150 mg is an oval, cylindrical, biconvex, pale yellow tablets marked "150" on one side with a break bar on the other side. Blister packs of 60 (AUST R 9987).
AURORIX 300 mg is an oval, cylindrical, biconvex, white to yellow-white tablets marked "300" on one side with a break bar on the other side. Blister packs of 60 (AUST R 51626).
Who distributes AURORIX
Viatris Pty Ltd
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point NSW 2000
www.viatris.com.au
Phone: 1800 274 276
This leaflet was prepared in March 2022.
AURORIX® is a Viatris company trade mark
AURORIX_cmi\Mar22/00
Published by MIMS May 2022


 Other adverse events with an incidence of < 1% in clinical studies, or reported in postmarketing surveillance are as follows:
Other adverse events with an incidence of < 1% in clinical studies, or reported in postmarketing surveillance are as follows: The chemical name of moclobemide is p-chloro-N- (2-morpholinoethyl) benzamide. Its empirical formula is C13H17O2N2Cl with a molecular weight of 268.74.
The chemical name of moclobemide is p-chloro-N- (2-morpholinoethyl) benzamide. Its empirical formula is C13H17O2N2Cl with a molecular weight of 268.74.