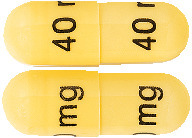WHAT IS IN THIS LEAFLET
This leaflet answers some of the common questions people ask about ESOMEPRAZOLE ACTAVIS. It does not contain all the information that is known about ESOMEPRAZOLE ACTAVIS.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor will have weighed the risks of you taking ESOMEPRAZOLE ACTAVIS against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine.
You may need to read it again.
WHAT ESOMEPRAZOLE ACTAVIS IS USED FOR
Reflux Oesophagitis
ESOMEPRAZOLE ACTAVIS is taken to treat reflux oesophagitis. This can be caused by "washing back" (reflux) of food and acid from the stomach into the food pipe (oesophagus).
Reflux can cause a burning sensation in the chest rising up to the throat, also known as heartburn.
ESOMEPRAZOLE ACTAVIS is also taken to help stop reflux oesophagitis coming back or relapsing.
Upper gastrointestinal symptoms associated with non-steroidal anti-inflammatory drugs (NSAIDs) therapy
ESOMEPRAZOLE ACTAVIS is taken to treat the symptoms of pain or discomfort, in the stomach caused by NSAIDs, a type of medicine for pain or inflammation.
ESOMEPRAZOLE ACTAVIS is also taken to help heal and prevent ulcers caused by NSAIDs.
Peptic Ulcers Associated with Helicobacter pylori Infection
Most people who have a peptic (gastric and duodenal) ulcer also have a bacterium called Helicobacter pylori in their stomach.
Depending on the position of the ulcer it is called a gastric or duodenal ulcer. A gastric ulcer occurs in the stomach. A duodenal ulcer occurs in the duodenum which is the tube leading out from the stomach.
If you have a peptic ulcer, your doctor will prescribe ESOMEPRAZOLE ACTAVIS with antibiotics. When ESOMEPRAZOLE ACTAVIS and antibiotics are taken together, they work to kill the bacterium and let your ulcer heal. You may need further treatment with antibiotics.
Zollinger-Ellison Syndrome
ESOMEPRAZOLE ACTAVIS is also used to treat a rare condition called Zollinger-Ellison syndrome, where the stomach produces large amounts of acid, much more than in ulcers or reflux disease.
Bleeding Peptic Ulcers
When peptic ulcers become severe enough, they start to bleed. You may receive treatment injected into your veins initially. This treatment may be followed with ESOMEPRAZOLE ACTAVIS capsules prescribed by your doctor for a longer period of time. This is to help your ulcer/s to heal.
How ESOMEPRAZOLE ACTAVIS works
ESOMEPRAZOLE ACTAVIS is a type of medicine called a proton-pump inhibitor. It works by decreasing the amount of acid made by the stomach, to give relief of symptoms and allow healing to take place. This does not stop food being digested in the normal way.
Follow all directions given to you by your doctor carefully.
They may differ from the information contained in this leaflet.
Ask your doctor if you want more information.
Your doctor may have prescribed ESOMEPRAZOLE ACTAVIS for another reason. There is no evidence that ESOMEPRAZOLE ACTAVIS is addictive
BEFORE YOU TAKE ESOMEPRAZOLE ACTAVIS
When you must not take it
Do not take ESOMEPRAZOLE ACTAVIS if you have allergies to:
- esomeprazole or any ingredient listed at the end of this leaflet.
- any medicines containing a protonpump inhibitor.
Some of the symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty breathing; swelling of the face, lips, tongue or other parts of the body; rash, itching or hives on the skin.
Do not take ESOMEPRAZOLE ACTAVIS if you are also taking atazanavir or cilostazol.
Please check with your doctor or pharmacist if you are taking these medicines. These medicines will be affected by ESOMEPRAZOLE ACTAVIS.
ESOMEPRAZOLE ACTAVIS is not approved for use in children younger than 1 year of age.
There is no specific information about use in children younger than 1 year of age, so ESOMEPRAZOLE ACTAVIS is not recommended in these patients.
Do not take ESOMEPRAZOLE ACTAVIS after the use by (expiry) date printed on the pack or if the packaging is torn or shows signs of tampering.
If it has expired or is damaged, return it to your pharmacist for disposal.
Before you start to take it
You must tell your doctor if you have:
- allergies to any other medicines, foods, dyes or preservatives.
- any problems with your liver
- severe kidney problems
- any other medical conditions
Do not take ESOMEPRAZOLE ACTAVIS if you are pregnant or breastfeeding unless your doctor says so. Ask your doctor about the risks and benefits involved.
It is not known if it is safe for you to take ESOMEPRAZOLE ACTAVIS while you are pregnant. It may affect your baby.
It is not known if your baby can take in ESOMEPRAZOLE ACTAVIS from breast milk if you are breastfeeding.
Taking other medicines
Do not take ESOMEPRAZOLE ACTAVIS if you are taking the following medicines:
- atazanavir, a medicine used to treat Human Immunodeficiency Virus (HIV)
- cilostazol, a medicine used to treat intermittent claudication.
Tell your doctor if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with ESOMEPRAZOLE ACTAVIS. These include:
- medicines used to treat fungal infections such as ketoconazole, itraconazole and voriconazole
- diazepam, a medicine used to treat anxiety and some other conditions
- phenytoin, a medicine used to treat epilepsy
- medicines used to treat depression such as citalopram, clomipramine and imipramine
- St John's Wort, a herbal remedy used to treat mood disorders
- medicines used to treat bacterial infections such as clarithromycin and rifampicin
- warfarin, a medicine used to prevent blood clots
- digoxin, a medicine used to treat heart conditions
- methotrexate, a medicine used to treat arthritis and some types of cancer
- tacrolimus – a medicine used to assist in organ transplants
- erlotinib or related medicines used to treat cancer.
These medicines may be affected by ESOMEPRAZOLE ACTAVIS or may affect how well it works. You may need different amounts of your medicine or you may need to take different medicines.
Your doctor can tell you what to do if you are taking any other medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking ESOMEPRAZOLE ACTAVIS.
If you have not told your doctor about any of these things, tell them before you take ESOMEPRAZOLE ACTAVIS.
HOW TO TAKE ESOMEPRAZOLE ACTAVIS
Follow all directions given to you by your doctor or pharmacist carefully.
These instructions may differ from the information contained in this leaflet.
If you do not understand the instructions, ask your doctor or pharmacist for help.
Children 1-11 years
Alternative preparations (oral suspensions) will be required for children in this age range.
How to take it
ESOMEPRAZOLE ACTAVIS is available as capsules
Take one ESOMEPRAZOLE ACTAVIS capsule each day, unless your doctor has told you otherwise.
How much to take
The dose of ESOMEPRAZOLE ACTAVIS capsules is usually 20 mg or 40 mg a day depending on what condition you are being treated for and how severe it is.
Swallow ESOMEPRAZOLE ACTAVIS capsules whole with a glass of water. Do not crush or chew the capsules.
If the capsules are chewed or crushed they will not work properly.
If you have difficulty swallowing the capsules:
- Place the contents of the capsule in half a glass of non-carbonated water. Mineral water or other liquids are not suitable.
- Gently mix the contents of the capsule in water by stirring, taking care not to crush the pellets.
- Drink the liquid with the pellets immediately, or within 30 minutes. Do not chew the pellets.
- Rinse the glass with half a glass of water and drink.
If you cannot swallow at all, follow steps 1-3 above to disperse the pellets and administer the liquid and pellets through a gastric tube.
Keeping a regular time for taking ESOMEPRAZOLE ACTAVIS will help to remind you to take it.
Keep taking ESOMEPRAZOLE ACTAVIS for as long as your doctor recommends.
In most patients, ESOMEPRAZOLE ACTAVIS relieves symptoms rapidly and healing is usually complete within 4 weeks.
Continue taking ESOMEPRAZOLE ACTAVIS for as long as your doctor tells you to.
ESOMEPRAZOLE ACTAVIS can be taken with food or on an empty stomach.
If you forget to take it
If you forget to take a dose, take it as soon as you remember, and then go back to taking it as you would normally.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to. Do not take a double dose to make up for the dose that you missed.
If you have trouble remembering when to take your medicine, ask your pharmacist for some hints.
Taking too much (overdose)
Telephone your doctor or the Poisons Information Centre (13 11 26) or go to Accident and Emergency at your nearest hospital immediately if you think that you or anyone else may have taken too many ESOMEPRAZOLE ACTAVIS. Do this even if there are no signs of discomfort or poisoning
WHILE YOU ARE TAKING ESOMEPRAZOLE ACTAVIS
Things you must do
Take ESOMEPRAZOLE ACTAVIS exactly as your doctor has prescribed.
If you are about to be started on any new medicine, tell your doctor, dentist or pharmacist that you are taking ESOMEPRAZOLE ACTAVIS.
Tell all doctors, dentists and pharmacists who are treating you that you are taking ESOMEPRAZOLE ACTAVIS.
Tell your doctor if you become pregnant while you are taking ESOMEPRAZOLE ACTAVIS.
Tell your doctor if your symptoms return.
Although ESOMEPRAZOLE ACTAVIS can heal ulcers successfully, it may not prevent them recurring at a later date.
If you need to have any medical tests while you are taking ESOMEPRAZOLE ACTAVIS, tell your doctor.
It may affect the results of some tests.
Things you must not do
Do not take it to treat any other complaints unless your doctor tells you to.
Do not give this medicine to anyone else, even if their symptoms seem similar to yours.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking ESOMEPRAZOLE ACTAVIS.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- nausea or vomiting
- constipation
- diarrhoea
- headache
- wind
- stomach pain
- skin rash, itchy skin
- dizziness
- dry mouth.
These side effects are usually mild.
Tell your doctor immediately if you notice any of the following:
- blurred vision
- mood changes, confusion or depression
- muscle pain or weakness, joint pain
- increase in breast size (males)
- increased sweating
- changes in sleep patterns
- fever
- increased bruising
- "pins and needles"
- hair loss
- tremor
- blood in the urine
These side effects may require medical attention.
If you notice any of the following happen, tell your doctor immediately or go to Accident and Emergency at the nearest hospital:
- shortness of breath, wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- severe skin reaction which may include rash, itching, redness, blistering or peeling of the skin
- signs of liver inflammation including yellowing of the skin or eyes, feeling generally unwell, nausea, vomiting, loss of appetite.
These are very serious side effects. You may need urgent medical treatment or hospitalisation.
Occasionally, ESOMEPRAZOLE ACTAVIS may be associated with changes in your liver or blood, which may require your doctor to do certain blood tests.
Tell your doctor if you think you have any of these effects or notice anything else that is making you feel unwell.
Some people may get other side effects while taking ESOMEPRAZOLE ACTAVIS.
Other problems are more likely to arise from the condition itself rather than the treatment.
For this reason, contact your doctor immediately if you notice any of the following:
- pain or indigestion during treatment with ESOMEPRAZOLE ACTAVIS
- you begin to vomit blood or food • you pass black (blood- stained) motions.
After taking it
Storage
Keep your ESOMEPRAZOLE ACTAVIS in the blister pack or bottle until it is time to take them.
If you take ESOMEPRAZOLE ACTAVIS out of the blister pack or bottle, it will not keep well.
Keep it in a cool, dry place where the temperature stays below 25°C.
Do not store it or any other medicine in the bathroom or near a sink.
Do not leave it in the car or on a window sill.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
A locked cupboard at least one-and a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking ESOMEPRAZOLE ACTAVIS or the capsules have passed their expiry date, ask your pharmacist what to do with any capsules you have left over.
PRODUCT DESCRIPTION
What it looks like
ESOMEPRAZOLE ACTAVIS 20 mg, gastro-resistant hard capsule, are size 4 capsules containing off-white to greyish spherical microgranules. The capsules have opaque yellow cap and opaque white body printed with a black inscription “20 mg” on the cap and on the body.
ESOMEPRAZOLE ACTAVIS 40 mg, gastro-resistant hard capsule, are size 3 capsules containing off-white to greyish spherical microgranules. The capsules have opaque yellow cap and body printed with a black inscription “40 mg” on the cap and on the body
Active ingredients:
Each capsule contains esomeprazole magnesium dihydrate equal to 20mg or 40 mg base.
Inactive ingredients:
sucrose, maize starch, hypromellose, dimethicone emulsion, polysorbate 80, mannitol, diacetylated monoglycerides, talc - purified, methacrylic acid-ethyl acrylate copolymer, triethyl citrate, and stearoyl macrogolglycerides.
Capsule shell: gelatin, titanium dioxide (E171), yellow iron oxide (E172) and imprinting Tekprint SW-9008 black ink (PI No. 2328)
This medicine does not contain lactose or gluten.
ESOMEPRAZOLE ACTAVIS capsules are available in blister packs of 7, 15, 30, or 100 capsules, or bottles containing desiccant and closed with a child resistant cap – bottle top is sealed with an aluminium induction seal for 30 or 98 capsules presentation.
Note: All pack sizes may not be marketed
Sponsor
Medis Pharma Pty Ltd
1002/53 Walker Street
North Sydney, NSW 2060
Australia
Australian Register Number(s)
ESOMEPRAZOLE ACTAVIS 20mg blister packs: AUST R 206009
ESOMEPRAZOLE ACTAVIS 20mg bottles: AUST R 206006
ESOMEPRAZOLE ACTAVIS 40mg blister packs: AUST R 206014
ESOMEPRAZOLE ACTAVIS 40mg bottles: AUST R 206002
This leaflet was prepared in February 2016