WHAT IS IN THIS LEAFLET
This leaflet answers some common questions about IDAPREX. It does not contain all the available information. It does not take the place of talking to your doctor, pharmacist or nurse. If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet.
All medicines have risks and benefits. Your doctor has weighed the risks of taking IDAPREX against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor, pharmacist or nurse. Keep this leaflet with the medicine. You may need to read it again.
WHAT IDAPREX IS
The name of your medicine is IDAPREX. The medicine contains the active ingredient perindopril erbumine. Perindopril belongs to a group of medicines called angiotensin converting enzyme (ACE) inhibitors.
WHAT IDAPREX IS USED FOR
IDAPREX is available only with a doctor's prescription.
IDAPREX lowers high blood pressure, a condition which doctors call hypertension.
Everyone has blood pressure. This pressure helps get your blood all around the body. Your blood pressure may be different at different times of the day, depending on how busy or worried you are. You have high blood pressure when your blood pressure stays higher than is needed, even when you are calm or relaxed.
There are usually no symptoms of high blood pressure. The only way of knowing that you have it is to have your blood pressure checked on a regular basis. If high blood pressure is not treated it can lead to serious health problems. You may feel fine and have no symptoms, but eventually it can cause stroke, heart disease and kidney failure. IDAPREX helps lower your blood pressure.
You may be prescribed IDAPREX for heart failure. Heart failure means that the heart muscle cannot pump blood strongly enough to supply all the blood needed throughout the body. Heart failure is not the same as heart attack and does not mean that the heart stops working.
Some people develop heart failure after having had a heart attack. However there are also other causes of heart failure.
Heart failure may start off with no symptoms, but as the condition progresses, you may feel short of breath or may get tired easily after light physical activity such as walking. You may wake up short of breath at night. Fluid may collect in different parts of the body, often first noticed as swollen ankles and feet. In severe heart failure, symptoms may occur even at rest.
IDAPREX helps to treat heart failure. If you follow your doctor's advice, your ability to perform daily activities may improve. You may breathe more easily, feel less tired, and have less swelling.
You may also have been prescribed IDAPREX if you have coronary artery disease. Coronary artery disease is narrowing of the vessels carrying blood to the heart. In patients with coronary artery disease, IDAPREX has been shown to reduce some of the risks, including heart attacks.
BEFORE YOU TAKE IDAPREX
There are some people who shouldn't take IDAPREX. Please read the list below. If you think any of these situations apply to you or you have any questions, please consult your doctor.
Do not take IDAPREX if:
- You are allergic to perindopril, any other ACE inhibitor or any of the ingredients listed at the end of this leaflet.
- You have experienced symptoms such as wheezing, swelling of the face, tongue, lips or throat, intense itching or severe skin rashes with previous ACE inhibitor treatment or if you or a member of your family have had these symptoms either spontaneously or, in response to another medicine in the past (a rare condition called angioedema).
- You are pregnant or trying to become pregnant.
- You are breastfeeding or plan to breast-feed.
- You undergo treatments where your blood is treated outside of the body (also known as extracorporeal treatments) that may increase your risk of allergic reactions, treatments such as:
- renal dialysis or haemofiltration using polyacrylonitrile membranes.
- low-density lipoprotein (LDL) apheresis, a technique where LDL is 'filtered' out of the blood, using dextran sulphate. - You are treated with a blood pressure lowering medicine containing aliskiren and have diabetes or impaired kidney function.
- You have kidney problems where the blood supply to your kidneys is reduced (renal artery stenosis).
- You are treated with sacubitril/ valsartan a medicine used to treat long-term heart failure as the risk of angioedema (rapid swelling under the skin in an area such as the throat) is increased (see also 'Tell Your Doctor Straight Away' and ‘Taking Other Medicines’ sections).
- The packaging is damaged or shows sign of tampering.
- The expiry date (EXP) on the pack has passed.
Tell your doctor straight away if:
- You are pregnant or become pregnant while taking IDAPREX, as it may cause serious harm to your baby.
- You are undergoing desensitisation treatment or have had an allergic reaction during previous desensitisation treatment (e.g. treatments using bee, wasp or ant venom).
- You are undergoing, or you are intending to undergo, treatments where your blood is treated outside of the body (also known as extracorporeal treatments).
- You are to undergo anaesthesia and/or surgery.
- You have recently suffered from diarrhoea or vomiting or are dehydrated.
- You are on a salt restricted diet or use salt substitutes which contain potassium.
- You have an intolerance to some sugars as IDAPREX contains lactose.
- You are taking lithium (used to treat mania or depression).
- You are taking any of the following medicines used to treat high blood pressure:
- an 'angiotensin II receptor blocker' (also known as ARBs or sartans - for example valsartan, telmisartan, irbesartan), in particular if you have diabetes-related kidney problems
- aliskiren. - You have any other health problems, including:
- kidney disease, or if you are on renal dialysis
- aortic stenosis (narrowing of the main blood vessel leading from the heart)
- liver disease
- high or low levels of potassium, or other problems with salt balance
- diabetes
- low blood pressure
- heart disease, including hypertrophic cardiomyopathy (heart muscle disease)
- systemic lupus erythematous or scleroderma (a disease affecting the skin, joints and kidneys)
- are of African origin since you may have a higher risk of angioedema and this medicine is less effective in lowering your blood pressure
- have abnormally increased levels of a hormone called aldosterone in your blood (primary aldosternism).
If you think any of these situations apply to you, or you have any doubts or questions about taking IDAPREX consult your doctor or pharmacist.
Taking other medicines
Taking IDAPREX may change the effect of some medicines, and some medicines may affect how well IDAPREX works. You may need different amounts of your medication or to take different medicines. The medicines that may interact with IDAPREX include the following:
- Some medications used to treat high blood pressure (including angiotensin receptor blockers), aliskiren (see also 'Do Not Take IDAPREX If' and 'Tell Your Doctor Straight Away' sections), diuretics (sometimes called "fluid" or "water" tablets because they increase the amount of urine passed each day)
- Some treatments where your blood is treated outside of the body, also known as extracorporeal treatments (see also 'Do Not Take IDAPREX If' and 'Tell Your Doctor Straight Away' sections)
- Some antibiotics and medicines used to treat infections
- Some anti-inflammatory drugs (including high dose aspirin, ibuprofen) for pain relief
- Medicines used to treat mood swings and some types of depression (lithium, tricyclic antidepressants, antipsychotics)
- Potassium-sparing diuretics, sources of potassium, like potassium tablet and salt substitutes containing potassium, other drugs which can increase potassium in your body (such as heparin, a medicine used to thin blood to prevent clots; co-trimoxazole also known as trimethoprim/sulfamethoxazole for infections caused by bacteria; and ciclosporin, an immunosuppressant medicine used to prevent organ transplant rejection)
- Immunosuppressants (medicines which reduces the activity of the body's natural defences)
- Vasodilators including nitrates
- Medicines used to treat diabetes (tablets and insulin)
- Medicines which may affect the blood cells, such as allopurinol, procainamide
- Baclofen (a medicine used to treat muscle stiffness in diseases such as multiple sclerosis)
- Medicines used for the treatment of low blood pressure, shock or asthma (e.g. ephedrine, noradrenaline or adrenaline (epinephrine))
- Gold salts, especially with intravenous administration (used to treat symptoms of rheumatoid arthritis)
- Medicines which may increase the risk of angioedema (a severe allergic reaction) such as:
- mammalian target of rapamycin (mTOR) inhibitors used to avoid rejection of transplanted organs (e.g. temsirolimus, sirolimus, everolimus)
- sacubitril (available as fixed-dose combination with valsartan), used to treat long-term heart failure (see also 'Do Not Take IDAPREX If' and 'Tell Your Doctor Straight Away' sections)
- gliptins used to treat diabetes (e.g. linagliptin, saxagliptin, sitagliptin, vildagliptin, alogliptin).
It is a good idea to remind your doctor of all other medicines you take. Your doctor or pharmacist has more information on medicines to be careful with or avoid while taking IDAPREX.
For older people or children
IDAPREX can generally be used safely by elderly people. However reduced kidney function is often found in elderly people and in this case, the starting dose should always be 2mg.
IDAPREX is not recommended for children.
HOW TO TAKE IDAPREX
The dose of IDAPREX you may need each day will be decided and adjusted by your doctor. This will normally be 2 mg (IDAPREX 2), 4 mg (IDAPREX 4) or 8 mg (IDAPREX 8) once daily for high blood pressure and for people with coronary artery disease or 2 mg (IDAPREX 2) to 4 mg (IDAPREX 4) once daily for heart failure.
Swallow your tablet(s) with water, preferably in the morning before a meal.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to. Otherwise, take it as soon as you remember, and then go back to taking it as you would normally.
Do not take a double dose to make up for the dose that you missed.
How long to take it
IDAPREX helps control your blood pressure, or heart failure or coronary artery disease but does not cure it. Continue taking the tablets for as long as your doctor tells you.
If you take too much
Taking too much IDAPREX (an overdose) may cause low blood pressure (also known as hypotension). The most likely effect in case of overdose is low blood pressure which can make you feel dizzy or faint. If this happens, lying down with the legs elevated can help. Other effects like sickness, cramps, sleepiness, confusion, kidney problems, salt and water disturbances are possible. You may require urgent medical attention.
If you think that you or anyone else may have taken too much IDAPREX then act immediately:
Telephone your doctor or the Poisons Information Centre (13 11 26 in Australia), or go to the Accident and Emergency department at your nearest hospital. Do this even if there are no signs of discomfort or poisoning.
WHILE YOU ARE TAKING IDAPREX
Things you must do
Take IDAPREX exactly as your doctor has prescribed. Otherwise you may not get the benefits from treatment. Tell all doctors, dentists and pharmacists who are involved with your treatment that you are taking IDAPREX.
Make sure you drink enough water during exercise and hot weather especially if you sweat a lot. This will help you avoid any dizziness or light-headedness caused by a sudden drop in blood pressure.
Tell your doctor straight away if you have excessive vomiting or diarrhoea while taking IDAPREX.
Things you must not do
- Do not give IDAPREX to anyone else, even if they have the same condition as you.
- Do not use IDAPREX to treat other complaints unless your doctor tells you to.
- Do not stop taking IDAPREX or change the dosage, without checking with your doctor.
- Do not stop taking your tablets because you are feeling better, unless advised by your doctor.
Things to be careful of
Take care when driving or operating machinery until you know how IDAPREX affects you.
Dizziness or weakness due to low blood pressure may occur in certain patients. If you have any of these symptoms do not drive or operate machinery.
SIDE EFFECTS
If you do not feel well while you are taking IDAPREX then tell your doctor or pharmacist as soon as possible.
All medicines can have side effects. Sometimes they are serious, most of the time they are not.
IDAPREX helps most people with high blood pressure, heart failure or coronary artery disease, but it may have unwanted side effects in a few people. While these side effects when they occur are usually mild, they can be serious.
Angioedema (a severe allergic reaction) has been reported in patients treated with ACE inhibitors, including IDAPREX. This may occur at any time during treatment. If you develop such symptoms described below you should tell your doctor immediately or go to the Accident and Emergency department at your nearest hospital. These side effects are extremely rare but can become serious:
- Swelling of your extremities (limbs, hands or feet), lips, face, mouth, tongue or throat
- A fast and irregular heartbeat
- Purple spots with occasional blisters on the front of your arms and legs and/or around your neck and ears (a rare condition known as Stevens-Johnson Syndrome)
- Difficulty in breathing
- Severe blisters, skin rash, itching, erythema multiforme or other allergic reactions.
Talk to your doctor or pharmacist or nurse if you notice any of the following side effects:
Common (may affect up to 1 in 10 people) side effects can include:
- Cough, often described as dry and irritating, shortness of breath, discomfort on exertion
- Headache, dizziness, vertigo, pins and needles
- Changes in the rhythm or rate of the heartbeat, fast or irregular heartbeat
- Feeling tired, lethargic or weak.
- Tinnitus (persistent noise in the ears), vision disturbances
- Hypotension, flushing, impaired peripheral circulation, vasculitis, nose bleeds
- Nausea, vomiting, taste disturbances, indigestion, diarrhoea, constipation, stomach pain or discomfort
- Muscle cramps
- Rash, pruritus (itching).
Uncommon (may affect up to 1 in 100 people) side effects can include:
- High levels in the blood of potassium, urea and/or creatine, low sodium levels in the blood
- Depression
- Mood disturbance, sleep disturbances (difficulty sleeping, abnormal dreams), feeling sleepy or drowsy, fainting
- Difficulty breathing or wheezing
- Dry mouth
- Swelling of the face, lips, mouth, tongue or throat which may cause difficulty in swallowing or breathing
- Excessive sweating
- Increased sensitivity of the skin to sun, skin rash or inflammation of the skin often including blisters that weep and become crusted
- Increase in some white blood cells
- Erectile dysfunction
- Fever or high temperature
- Chest pain
- Kidney problems
- Decreased blood sugar levels
- Aching muscles, muscle tenderness or weakness, not caused by exercise
- Generally feeling unwell
- Falls.
Rare (may affect up to 1 in 1,000 people) side effects can include:
- Elevation of bilirubin levels in the blood, increases in liver enzymes
- Worsening of psoriasis
- Kidney disease
- Problems with production or passing of urine
- Dark urine, feeling sick (nausea) or being sick (vomiting), muscle cramps, confusion and seizures.
These may be symptoms of a condition called SIADH (inappropriate antidiuretic hormone secretion).
Very rare (may affect up to 1 in 10,000 people) side effects can include:
- Hepatitis
- Inflammation of the pancreas (pancreatitis)
- Eosinophilic pneumonia
- Runny or blocked nose, sneezing, facial pressure or pain
- Red, often itchy spots, similar to the rash of measles, which starts on the limbs and sometimes on the face and the rest of the body.
- Joint pain
- Swelling of hands, ankles or feet
- Bleeding or bruising more easily than normal caused by a low blood platelet count, frequent infections such as fever, severe chills, sore throat or mouth ulcers caused by a lack of white blood cells, pancytopenia (a rare type of anaemia)
- Illnesses resulting from a lack of red blood cells
- Stroke, myocardial infarction, angina pectoris (a feeling of tightness, pressure or heaviness in the chest)
- Changes in the rhythm or rate of the heartbeat
- Confusion, depression or hallucinations.
Not known (frequency cannot be estimated from the data available):
- Discolouration, numbness and pain in fingers or toes (Raynaud's phenomenon).
Other:
Concentrated urine (dark in colour), feel or are sick, have muscle cramps, confusion and fits which may be due to inappropriate anti-diuretic hormone (ADH) secretion can occur with ACE inhibitors. If you have these symptoms contact your doctor as soon as possible.
Consult your doctor, pharmacist or nurse if you experience any of these or notice anything else that is making you feel unwell.
Do not be alarmed by this list of possible side effects. You may not experience any of them. Other uncommon side effects have been reported and you should ask your doctor, pharmacist or nurse if you want to know more.
AFTER TAKING IDAPREX
Storage
Heat and dampness can destroy some medicines. Keep your tablets in the pack until it is time to take them. Keep them in a cool, dry place where it stays below 30°C. Do not store medicines in a bathroom or near a sink. Do not leave them in a car or on a windowsill. Keep them where children cannot reach them. Keep the container tightly closed.
Disposal
If your doctor tells you to stop taking IDAPREX, or the tablets have passed their expiry date, return any leftover tablets to your pharmacist for disposal.
WHAT IDAPREX LOOKS LIKE
IDAPREX 2 are round biconvex white tablets containing 2 mg of perindopril erbumine.
IDAPREX 4 are rod-shaped biconvex scored white tablets containing 4 mg of perindopril erbumine.
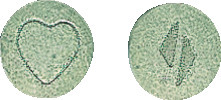
IDAPREX 8 tablets are round biconvex green tablets, with a heart on one face and a logo on the other face containing 8 mg of perindopril erbumine.
IDAPREX 2, IDAPREX 4 and IDAPREX 8 are contained in a blister strip that is itself enclosed in an aluminium foil envelope containing a desiccant sachet. Each box contains 30 tablets. A desiccant is a substance that absorbs moisture and helps to protect the tablets, especially when it is very humid.
Ingredients
Each tablet of IDAPREX 2 contains 2 mg of perindopril erbumine as the active ingredient and a number of inactive ingredients.
Each tablet of IDAPREX 4 contains 4 mg of perindopril erbumine as the active ingredient and a number of inactive ingredients.
Each tablet of IDAPREX 8 contains 8 mg of perindopril erbumine as the active ingredient and a number of inactive ingredients.
The inactive ingredients include microcrystalline cellulose, lactose monohydrate, magnesium stearate, colloidal anhydrous silica, and (8mg tablet only) aluminium copper complexes of chlorophyllins (E141ii) lake.
IDAPREX is registered on the Australian Register of Therapeutic Goods.
Australian Register Numbers:
IDAPREX 2: AUST R 34266
IDAPREX 4: AUST R 34267
IDAPREX 8: AUST R 96738
DISTRIBUTOR
IDAPREX is distributed in Australia by:
Sandoz Pty Ltd
54 Waterloo Road
Macquarie Park NSW 2113
This document was last revised in March 2022
Published by MIMS April 2022





 The reduction in the primary composite endpoint was mainly due to a reduction in the number of nonfatal myocardial infarctions. There was no significant reduction in the rate of cardiovascular mortality or total mortality in patients taking perindopril compared to those taking placebo.
The reduction in the primary composite endpoint was mainly due to a reduction in the number of nonfatal myocardial infarctions. There was no significant reduction in the rate of cardiovascular mortality or total mortality in patients taking perindopril compared to those taking placebo.
