What is in this leaflet
Read this leaflet carefully before you start taking TENOFOVIR DISOPROXIL VIATRIS. Also, read it each time you get your TENOFOVIR DISOPROXIL VIATRIS prescription refilled, in case something has changed.
This information does not take the place of talking with your doctor or pharmacist when you start this medicine and at check-ups. You should stay under a doctor's care when taking TENOFOVIR DISOPROXIL VIATRIS.
It is extremely important that you do not change or stop your medicine without first talking with your doctor or pharmacist (see HOW SHOULD I TAKE TENOFOVIR DISOPROXIL VIATRIS and WHAT ARE THE POSSIBLE SIDE EFFECTS OF TENOFOVIR DISOPROXIL VIATRIS).
Talk to your doctor or pharmacist if you have any questions about TENOFOVIR DISOPROXIL VIATRIS.
What TENOFOVIR DISOPROXIL VIATRIS is used for
Tenofovir disoproxil maleate is an antiviral medication used to treat two different viruses;
- Chronic Hepatitis B (CHB) and
- Human Immunodeficiency Virus (HIV) infection.
TENOFOVIR DISOPROXIL VIATRIS is a type of medicine called HBV polymerase inhibitor and a nucleotide analogue reverse transcriptase inhibitor (NRTI).
How TENOFOVIR DISOPROXIL VIATRIS works in the Treatment of CHB:
TENOFOVIR DISOPROXIL VIATRIS is used to treat CHB (an infection with hepatitis B virus [HBV]) in adults and paediatric patients aged 12 years and older and weighing at least 35 kg.
TENOFOVIR DISOPROXIL VIATRIS works by interfering with the normal working of enzymes (HBV DNA polymerase) that are essential for HBV to reproduce itself. TENOFOVIR DISOPROXIL VIATRIS may help lower the amount of hepatitis B virus in your body by lowering the ability of the virus to multiply and infect new liver cells and can improve the inflammation and scar tissue caused by the hepatitis B virus in your liver.
Lowering the amount of virus in your body may reduce the chance of developing cirrhosis, liver failure and liver cancer.
We do not know how long TENOFOVIR DISOPROXIL VIATRIS may help treat your hepatitis.
Sometimes viruses change in your body and medicines no longer work. This is called drug resistance.
How TENOFOVIR DISOPROXIL VIATRIS works in the Treatment of HIV Infection:
TENOFOVIR DISOPROXIL VIATRIS is also used to treat HIV infection in adults and paediatric patients aged 12 years and older and weighing at least 35 kg.
TENOFOVIR DISOPROXIL VIATRIS is always used in combination with other anti-HIV medicines to treat people with HIV-1 infection.
HIV infection destroys CD4 (T) cells, which are important to the immune system. After a large number of T cells are destroyed, acquired immune deficiency syndrome (AIDS) develops.
TENOFOVIR DISOPROXIL VIATRIS helps to block HIV-1 reverse transcriptase, a chemical (enzyme) in your body that is needed for HIV-1 to multiply. TENOFOVIR DISOPROXIL VIATRIS lowers the amount of HIV-1 in the blood (called viral load) and may help to increase the number of T cells (called CD4 cells). Lowering the amount of HIV-1 in the blood lowers the chance of death or infections that happen when your immune system is weak (opportunistic infections).
You do not have to have HIV infection to be treated with TENOFOVIR DISOPROXIL VIATRIS for HBV and vice versa.
Use in Children
TENOFOVIR DISOPROXIL VIATRIS is for adults and paediatric patients aged 12 years and older and weighing at least 35 kg.
Does TENOFOVIR DISOPROXIL VIATRIS cure HIV or AIDS?
TENOFOVIR DISOPROXIL VIATRIS does not cure HIV infection or AIDS.
The long-term effects of TENOFOVIR DISOPROXIL VIATRIS are not known at this time. People taking TENOFOVIR DISOPROXIL VIATRIS may still get opportunistic infections or other conditions that happen with HIV-1 infection.
Opportunistic infections are infections that develop because the immune system is weak.
Some of these conditions are:
- pneumonia,
- herpes virus infections, and
- Mycobacterium avium complex (MAC) infections.
Does TENOFOVIR DISOPROXIL VIATRIS reduce the risk of passing HIV-1 or HBV to others?
TENOFOVIR DISOPROXIL VIATRIS does not reduce the risk of passing HIV-1 or HBV to others through sexual contact or blood contamination.
Continue to practice safe sex and do not use or share dirty needles.
Before you take TENOFOVIR DISOPROXIL VIATRIS
Who must not take it
Together with your doctor, you need to decide whether TENOFOVIR DISOPROXIL VIATRIS is right for you.
Do not take TENOFOVIR DISOPROXIL VIATRIS if:
- you are allergic to tenofovir disoproxil maleate, tenofovir disoproxil fumarate or any of the ingredients listed at the end of this leaflet under the heading PRODUCT DESCRIPTION.
- you are already taking any other medicines containing tenofovir disoproxil maleate or tenofovir disoproxil fumarate.
- you are already taking adefovir dipivoxil to treat hepatitis B virus (HBV) infection.
- you are already taking tenofovir alafenamide to treat HIV or hepatitis B virus (HBV) infection.
Before you start to take it:
Tell your doctor if you are pregnant or planning to become pregnant. The effects of TENOFOVIR DISOPROXIL VIATRIS on pregnant women or their unborn babies are not known.
Tell your doctor if you are breastfeeding. Do not breast-feed if you are taking TENOFOVIR DISOPROXIL VIATRIS. The active substance in this medicine (tenofovir disoproxil maleate) has been found in breast milk at low concentrations.
Do not breast-feed if you have HIV or HBV.
If you are a woman who has or will have a baby, talk with your doctor or pharmacist about the best way to feed your baby. If your baby does not already have HIV or HBV, there is a chance that the baby can get HIV or HBV through breast-feeding.
Tell your doctor if you have kidney problems.
Tell your doctor if you have bone problems.
Tell your doctor if you have liver problems, including HBV and hepatitis C virus (HCV).
Tell your doctor if you have HIV infection.
Tell your doctor or pharmacist about all your medical conditions.
Taking other medicines
Tell your doctor or pharmacist about all the medicines you take, including prescription and non- prescription medicines and dietary supplements.
Especially tell your doctor or pharmacist if you take:
- VOSEVI (sofosbuvir/ velpatasvir/ voxilaprevir), HARVONI (ledipasvir/sofosbuvir) or EPCLUSA (sofosbuvir/ velpatasvir) to treat your HCV infection. These medicines may increase the amount of TENOFOVIR DISOPROXIL VIATRIS in your blood, which could result in additional or more intense side effects (see SIDE EFFECTS).
- VIDEX, VIDEX EC (didanosine); TENOFOVIR DISOPROXIL VIATRIS may increase the amount of VIDEX in your blood. You may need to be followed more carefully if you are taking VIDEX and TENOFOVIR DISOPROXIL VIATRIS together. If you are taking VIDEX and TENOFOVIR DISOPROXIL VIATRIS together, your doctor may need to reduce your dose of VIDEX.
- REYATAZ (atazanavir sulfate) or KALETRA (lopinavir/ritonavir); These medicines may increase the amount of TENOFOVIR DISOPROXIL VIATRIS in your blood, which could result in more side effects (see SIDE EFFECTS). You may need to be followed more carefully if you are taking TENOFOVIR DISOPROXIL VIATRIS and REYATAZ or KALETRA together. TENOFOVIR DISOPROXIL VIATRIS may decrease the amount of REYATAZ in your blood. If you are taking TENOFOVIR DISOPROXIL VIATRIS and REYATAZ together you should also be taking NORVIR (ritonavir).
It is a good idea to keep a complete list of all the medicines that you take.
Make a new list when medicines are added or stopped. Give copies of this list to your doctor or pharmacist every time you visit your doctor or fill a prescription.
How to take TENOFOVIR DISOPROXIL VIATRIS
Stay under a doctor's care when taking TENOFOVIR DISOPROXIL VIATRIS.
Do not change your treatment or stop treatment without first talking with your doctor.
Take TENOFOVIR DISOPROXIL VIATRIS exactly as your doctor prescribed it.
Follow the directions from your doctor or pharmacist, exactly as written on the label.
Set up a dosing schedule and follow it carefully.
How much to take
The usual dose of TENOFOVIR DISOPROXIL VIATRIS is one tablet once a day.
If you have kidney problems, your doctor may recommend that you take TENOFOVIR DISOPROXIL VIATRIS less frequently.
How to take it
TENOFOVIR DISOPROXIL VIATRIS is best taken with a meal or just afterwards, however taking it without food should not reduce the effectiveness of the medicine.
If you are taking TENOFOVIR DISOPROXIL VIATRIS to treat HIV or if you have HIV and HBV coinfection and are taking TENOFOVIR DISOPROXIL VIATRIS, always take TENOFOVIR DISOPROXIL VIATRIS in combination with other anti-HIV medicines. TENOFOVIR DISOPROXIL VIATRIS and other medicines like TENOFOVIR DISOPROXIL VIATRIS, may be less likely to work in the future if you are not taking TENOFOVIR DISOPROXIL VIATRIS with other anti-HIV medicines because you may develop resistance to those medicines. If you have any questions about what medicines you should or should not be taking, please see your doctor or pharmacist.
If you have been given TENOFOVIR DISOPROXIL VIATRIS to treat CHB, you are advised to get a HIV test before you start taking TENOFOVIR DISOPROXIL VIATRIS and at any time after that when there is a chance you were exposed to HIV.
When your TENOFOVIR DISOPROXIL VIATRIS supply starts to run low, get more from your doctor or pharmacist. This is very important because the amount of virus in your blood may increase if the medicine is stopped for even a short time. The virus may develop resistance to TENOFOVIR DISOPROXIL VIATRIS, and may become harder to treat. If you are taking TENOFOVIR DISOPROXIL VIATRIS to treat CHB, stopping treatment may result in very severe hepatitis and serious liver problems (see SIDE EFFECTS).
Only take medicine that has been prescribed specifically for you.
Do not give TENOFOVIR DISOPROXIL VIATRIS to others or take medicine prescribed for someone else.
If you forget to take TENOFOVIR DISOPROXIL VIATRIS?
It is important that you do not miss any doses.
If you miss a dose of TENOFOVIR DISOPROXIL VIATRIS, take it as soon as possible and then take your next scheduled dose at its regular time.
If it is almost time for your next dose, do not take the missed dose. Wait and take the next dose at the regular time. Do not double the next dose.
If you take too much (overdose)?
Immediately telephone your doctor or Poisons Information Centre (telephone 13 11 26), or in New Zealand the Poisons Centre (telephone 0800 POISON or 0800 764 766) or go to the accident and emergency department at your nearest hospital if you think you or anyone else may have taken too many TENOFOVIR DISOPROXIL VIATRIS tablets. Do this even if there are no signs of discomfort or poisoning. This may need urgent medical attention.
While you are taking TENOFOVIR DISOPROXIL VIATRIS
Things you must not do
Do not breast-feed. See "Before you start to take it".
Things to be careful of
Some patients taking TENOFOVIR DISOPROXIL VIATRIS have experienced dizziness. Make sure you know how you react to TENOFOVIR DISOPROXIL VIATRIS before you drive a car, operate machinery or do anything else that could be dangerous if you are dizzy.
SIDE EFFECTS
In clinical studies in patients with HIV: The most common side effects of TENOFOVIR DISOPROXIL VIATRIS are:
- diarrhoea
- nausea
- vomiting
- dizziness
Less common side effects include:
- flatulence (intestinal gas)
In clinical studies in patients with CHB, the only common side effect of TENOFOVIR DISOPROXIL VIATRIS is:
- nausea
Marketing experience has shown other side effects reported since tenofovir disoproxil has been marketed include:
- low blood phosphate
- shortness of breath
- increased liver enzymes
- increased amylase
- inflammation of the liver
- stomach pain
- inflammation of the pancreas
- rash and
- weakness
TENOFOVIR DISOPROXIL VIATRIS may cause the following other side effects
Kidney Problems
Some patients treated with tenofovir disoproxil have had kidney problems. If you have had kidney problems in the past or need to take another drug that can cause kidney problems, your doctor may need to perform additional blood tests. Kidney problems may be associated with muscle problems and softening of the bones.
Changes in Bone Mineral Density
Laboratory tests show changes in the bones of patients treated with tenofovir disoproxil. It is not known whether long-term use of TENOFOVIR DISOPROXIL VIATRIS will cause damage to your bones. If you have had bone problems in the past, your doctor may need to perform additional tests or may suggest additional medication.
Lactic Acidosis
Some patients taking antiviral drugs like TENOFOVIR DISOPROXIL VIATRIS have developed a condition called lactic acidosis (a build-up in the blood of lactic acid, the same substance that causes your muscles to burn during heavy exercise). Symptoms of lactic acidosis include nausea, vomiting, unusual or unexpected stomach discomfort, and weakness.
If you notice these symptoms or if your medical condition changes suddenly, call your doctor right away.
Hepatic Flares
It is extremely important that you do not stop taking TENOFOVIR DISOPROXIL VIATRIS without your doctor's advice. If you have Hepatitis B infection or HIV and HBV infection together, you may have a "flare-up" of Hepatitis B if you stop taking TENOFOVIR DISOPROXIL VIATRIS, where the disease suddenly returns in a worse way than before. This flare- up may lead to liver failure and possibly liver transplantation or death.
After stopping TENOFOVIR DISOPROXIL VIATRIS, tell your doctor immediately about any new, unusual, or worsening symptoms that you notice after stopping treatment. After you stop taking TENOFOVIR DISOPROXIL VIATRIS, your doctor will still need to check your health and take blood tests to check your liver for several months.
There have been other side effects in patients taking tenofovir disoproxil. However, these side effects may have been due to other medicines that patients were taking or to the illness itself.
Some of these side effects can be serious.
This list of side effects is not complete. If you have questions about side effects, ask your doctor or pharmacist.
You should report any new or continuing symptoms to your doctor or pharmacist right away. Your doctor or pharmacist may be able to help you manage these side effects.
After taking TENOFOVIR DISOPROXIL VIATRIS
Storage
Keep TENOFOVIR DISOPROXIL VIATRIS and all other medications out of reach of children.
Store TENOFOVIR DISOPROXIL VIATRIS at room temperature (below 25°C).
It should remain stable until the expiration date printed on the label.
Do not keep your medicine in places that are too hot or cold.
Do not leave TENOFOVIR DISOPROXIL VIATRIS in the car or on a window sill. Heat and dampness can destroy some medicines.
Keep your TENOFOVIR DISOPROXIL VIATRIS tablets in the bottle with the cap tightly closed until you take them.
If you take TENOFOVIR DISOPROXIL VIATRIS tablets out of their pack they may not keep well.
Do not keep medicine that is out of date or that you no longer need.
If you throw any medicines away make sure that children will not find them.
General Advice
Talk to your doctor or pharmacist if you have any questions about this medicine or your condition. Medicines are sometimes prescribed for purposes other than those listed in a Consumer Medicine Information.
If you have any concerns about this medicine, ask your doctor or pharmacist. Your doctor or pharmacist can give you information about this medicine that was written for doctors or pharmacists (Product Information/Data Sheet).
Do not use this medicine for a condition for which it was not prescribed.
Do not share this medicine with other people.
Do not use if seal over bottle opening is broken or missing.
Product description
What TENOFOVIR DISOPROXIL VIATRIS tablets look like
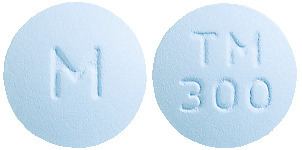
TENOFOVIR DISOPROXIL VIATRIS 300 mg film coated tablets are light blue coloured, 12 mm, round, biconvex, film coated tablets debossed with 'TM300' on one side of the tablet and 'M' on other side.
Ingredients
Each TENOFOVIR DISOPROXIL VIATRIS tablet contains the following active ingredient
- tenofovir disoproxil maleate
Each TENOFOVIR DISOPROXIL VIATRIS tablet contains the following inactive ingredients
- microcrystalline cellulose
- lactose monohydrate
- hyprolose
- colloidal anhydrous silica
- magnesium stearate
- OPADRY II complete film coating system 32K505018 BLUE (ARTG PI No: 109808)
TENOFOVIR DISOPROXIL VIATRIS also contains sugars as lactose.
Sponsor
Alphapharm Pty Ltd trading as Viatris
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point NSW 2000
www.viatris.com.au
Phone: 1800 274 276
This leaflet was prepared in September 2022.
Australian registration numbers:
TENOFOVIR DISOPROXIL VIATRIS 300 mg film coated tablet bottle:
AUST R 261093
TENOFOVIR DISOPROXIL VIATRIS_cmi\Sep22/00
Published by MIMS November 2022

 The pharmacokinetics of tenofovir have not been evaluated in non-haemodialysis patients with creatinine clearance < 10 mL/min; therefore, no dosing recommendation is available for these patients.
The pharmacokinetics of tenofovir have not been evaluated in non-haemodialysis patients with creatinine clearance < 10 mL/min; therefore, no dosing recommendation is available for these patients. Following multiple dosing to HIV- and HBV-negative subjects receiving either chronic methadone maintenance therapy or oral contraceptives, steady state tenofovir pharmacokinetics were similar to those observed in previous studies, indicating lack of clinically significant drug interactions between these agents and tenofovir. In a study conducted in healthy volunteers dosed with a single 600 mg dose of ribavirin, no clinically significant drug interactions were observed between tenofovir disoproxil (as fumarate) and ribavirin.
Following multiple dosing to HIV- and HBV-negative subjects receiving either chronic methadone maintenance therapy or oral contraceptives, steady state tenofovir pharmacokinetics were similar to those observed in previous studies, indicating lack of clinically significant drug interactions between these agents and tenofovir. In a study conducted in healthy volunteers dosed with a single 600 mg dose of ribavirin, no clinically significant drug interactions were observed between tenofovir disoproxil (as fumarate) and ribavirin.









 The percent of patients with HIV-1 RNA < 400 copies/mL and outcomes of patients through 48 weeks are summarised in Table 13.
The percent of patients with HIV-1 RNA < 400 copies/mL and outcomes of patients through 48 weeks are summarised in Table 13. At 24 weeks of therapy, there was a higher proportion of patients in the tenofovir arm compared to the placebo arm with HIV-1 RNA < 50 copies/mL (19% and 1% respectively). Mean change in absolute CD4 counts by week 24 was +11 cells/mm3 for tenofovir group and -5 cells/mm3 for the placebo group. Mean change in absolute CD4 counts by week 48 was +4 cells/mm3 for the tenofovir group.
At 24 weeks of therapy, there was a higher proportion of patients in the tenofovir arm compared to the placebo arm with HIV-1 RNA < 50 copies/mL (19% and 1% respectively). Mean change in absolute CD4 counts by week 24 was +11 cells/mm3 for tenofovir group and -5 cells/mm3 for the placebo group. Mean change in absolute CD4 counts by week 48 was +4 cells/mm3 for the tenofovir group. Achievement of plasma HIV-1 RNA concentrations of less than 400 copies/mL at week 144 was similar between the two treatment groups for the population stratified at baseline on the basis of HIV-1 RNA concentration (≤ or > 100,000 copies/mL) and CD4 cell count (< or ≥ 200 cells/mm3). Through 144 weeks of therapy, 62% and 58% of patients in the tenofovir and d4T arms, respectively achieved and maintained confirmed HIV-1 RNA < 50 copies/mL. The mean increase from baseline in CD4 cell count was 263 cells/mm3 for the tenofovir arm and 283 cells/mm3 for the d4T arm.
Achievement of plasma HIV-1 RNA concentrations of less than 400 copies/mL at week 144 was similar between the two treatment groups for the population stratified at baseline on the basis of HIV-1 RNA concentration (≤ or > 100,000 copies/mL) and CD4 cell count (< or ≥ 200 cells/mm3). Through 144 weeks of therapy, 62% and 58% of patients in the tenofovir and d4T arms, respectively achieved and maintained confirmed HIV-1 RNA < 50 copies/mL. The mean increase from baseline in CD4 cell count was 263 cells/mm3 for the tenofovir arm and 283 cells/mm3 for the d4T arm.
 In this study, tenofovir disoproxil + Emtriva in combination with efavirenz was statistically significantly superior to Combivir in combination with efavirenz with regards to the primary and secondary endpoints: achieving and maintaining HIV-1 RNA < 400 copies/mL through 48 and 144 weeks (Table 15). The difference in the proportions of responders between the tenofovir disoproxil + Emtriva group and the Combivir group was 11.4%, and the 95% CI was 4.3% to 18.6% (p=0.002) at week 48 and a difference of 12.9% (95% CI was 4.2% to 21.6%, p=0.004) at week 144.
In this study, tenofovir disoproxil + Emtriva in combination with efavirenz was statistically significantly superior to Combivir in combination with efavirenz with regards to the primary and secondary endpoints: achieving and maintaining HIV-1 RNA < 400 copies/mL through 48 and 144 weeks (Table 15). The difference in the proportions of responders between the tenofovir disoproxil + Emtriva group and the Combivir group was 11.4%, and the 95% CI was 4.3% to 18.6% (p=0.002) at week 48 and a difference of 12.9% (95% CI was 4.2% to 21.6%, p=0.004) at week 144. In the protocol defined analyses, virologic response to tenofovir disoproxil was not reduced in patients with HIV that expressed the lamivudine/abacavir-associated M184V mutation. In the absence of zidovudine-associated mutations, patients with the M184V mutation receiving tenofovir disoproxil showed a -0.84 log10 copies/mL decrease in their HIV RNA relative to placebo. In the presence of zidovudine-associated mutations, the M184V mutation did not affect the mean HIV RNA responses to tenofovir treatment. HIV-1 RNA responses among these patients were durable through week 48.
In the protocol defined analyses, virologic response to tenofovir disoproxil was not reduced in patients with HIV that expressed the lamivudine/abacavir-associated M184V mutation. In the absence of zidovudine-associated mutations, patients with the M184V mutation receiving tenofovir disoproxil showed a -0.84 log10 copies/mL decrease in their HIV RNA relative to placebo. In the presence of zidovudine-associated mutations, the M184V mutation did not affect the mean HIV RNA responses to tenofovir treatment. HIV-1 RNA responses among these patients were durable through week 48.
 Tenofovir disoproxil was associated with statistically significantly greater proportions of patients with undetectable HBV DNA (< 169 copies/mL [< 29 IU/mL]; the limit of quantification of the Roche Cobas TaqMan HBV assay), when compared to Hepsera (study 0102; 91%, and 56% and study 0103; 69% and 9%), respectively.
Tenofovir disoproxil was associated with statistically significantly greater proportions of patients with undetectable HBV DNA (< 169 copies/mL [< 29 IU/mL]; the limit of quantification of the Roche Cobas TaqMan HBV assay), when compared to Hepsera (study 0102; 91%, and 56% and study 0103; 69% and 9%), respectively.
 Paired baseline and week 240 liver biopsy data were available for 331/489 patients who remained in studies 0102 and 0103 (see Table 21). Ninety-five percent (225/237) of patients without cirrhosis at baseline and 99% (93/94) of patients with cirrhosis at baseline had either no change or an improvement in fibrosis (Ishak fibrosis score). Of the 94 patients with cirrhosis at baseline (Ishak fibrosis score 5-6), 26% (24) experienced no change in Ishak fibrosis score and 72% (68) experienced reversal of cirrhosis by week 240 with a reduction in Ishak fibrosis score of at least 2 points except for one patient with an initial Ishak score of five.
Paired baseline and week 240 liver biopsy data were available for 331/489 patients who remained in studies 0102 and 0103 (see Table 21). Ninety-five percent (225/237) of patients without cirrhosis at baseline and 99% (93/94) of patients with cirrhosis at baseline had either no change or an improvement in fibrosis (Ishak fibrosis score). Of the 94 patients with cirrhosis at baseline (Ishak fibrosis score 5-6), 26% (24) experienced no change in Ishak fibrosis score and 72% (68) experienced reversal of cirrhosis by week 240 with a reduction in Ishak fibrosis score of at least 2 points except for one patient with an initial Ishak score of five. When the data were evaluated including only patients that completed 384 weeks of therapy (observed (missing data is excluded) and data after the addition of emtricitabine included; on therapy analysis), in the group of patients who received 48 weeks of double-blind treatment with tenofovir disoproxil followed by open-label treatment with tenofovir disoproxil; 99% (173 of 174) and 100% (88 of 88) of patients had HBV DNA < 400 copies/mL and 88% (141 of 160) and 81% (70 of 86) of patients had ALT normalisation at week 384, in studies 0102 and 0103 respectively. In study 0103, HBeAg loss was reported for 44% (31 of 70) of patients and 28% (19 of 68) of patients experienced HBeAg seroconversion. 14% of patients experienced HBsAg loss and 12% of patients experienced HBsAg seroconversion by week 384. In study 102, HBsAg loss and seroconversion were 1% in both treatment group.
When the data were evaluated including only patients that completed 384 weeks of therapy (observed (missing data is excluded) and data after the addition of emtricitabine included; on therapy analysis), in the group of patients who received 48 weeks of double-blind treatment with tenofovir disoproxil followed by open-label treatment with tenofovir disoproxil; 99% (173 of 174) and 100% (88 of 88) of patients had HBV DNA < 400 copies/mL and 88% (141 of 160) and 81% (70 of 86) of patients had ALT normalisation at week 384, in studies 0102 and 0103 respectively. In study 0103, HBeAg loss was reported for 44% (31 of 70) of patients and 28% (19 of 68) of patients experienced HBeAg seroconversion. 14% of patients experienced HBsAg loss and 12% of patients experienced HBsAg seroconversion by week 384. In study 102, HBsAg loss and seroconversion were 1% in both treatment group.

 At week 48, no patient with lamivudine- or adefovir-resistant mutations at baseline, had HBeAg/HBsAg loss and/or seroconversion.
At week 48, no patient with lamivudine- or adefovir-resistant mutations at baseline, had HBeAg/HBsAg loss and/or seroconversion. Tenofovir disoproxil maleate is a water soluble ester prodrug of the active ingredient tenofovir. Tenofovir is converted intracellularly to tenofovir monophosphate and tenofovir diphosphate. The pharmacokinetics of tenofovir disoproxil (as fumarate) have been evaluated in healthy volunteers and HIV-1 infected individuals. Tenofovir pharmacokinetics are similar between these populations.
Tenofovir disoproxil maleate is a water soluble ester prodrug of the active ingredient tenofovir. Tenofovir is converted intracellularly to tenofovir monophosphate and tenofovir diphosphate. The pharmacokinetics of tenofovir disoproxil (as fumarate) have been evaluated in healthy volunteers and HIV-1 infected individuals. Tenofovir pharmacokinetics are similar between these populations. Tenofovir is efficiently removed by haemodialysis with an extraction coefficient of approximately 54%. Following a single 300 mg dose of tenofovir disoproxil (as fumarate), a four-hour haemodialysis session removed approximately 10% of the administered tenofovir dose.
Tenofovir is efficiently removed by haemodialysis with an extraction coefficient of approximately 54%. Following a single 300 mg dose of tenofovir disoproxil (as fumarate), a four-hour haemodialysis session removed approximately 10% of the administered tenofovir dose. Molecular formula: C19H30N5O10P.C4H4O4. Molecular weight: 635.51.
Molecular formula: C19H30N5O10P.C4H4O4. Molecular weight: 635.51.