Up-titrating heart failure medicines: A practical guide
Following the pathways and guidance in this article can make it more straightforward for GPs to up-titrate heart failure medicines, particularly for the large group of patients with HFrEF who are relatively well and usually do not experience adverse effects requiring a change to up-titration doses.

Key points
- People with HFrEF should be prescribed a combination of an ACE inhibitor (or ARB if not tolerated), a heart failure beta blocker and an MRA, up-titrated to target or maximum tolerated doses, to help improve quality of life, reduce hospitalisations and save lives.
- GPs play a vital role in starting and optimising doses of guideline medicines for patients with HFrEF.
- Up-titration guidance includes: start heart failure medicines at low doses, double the dose one medicine at a time every 2–4 weeks (except MRAs, up-titrated in 4–8 weeks) and add the next medicine before reaching target or maximum tolerated dose of the previous medicine.
- Monitor patients closely with a review 1–2 weeks after each medicine initiation and dose increase, and make variations to up-titration when required in response to adverse effects.
- Asymptomatic or mild changes in blood pressure, heart rate or renal function during up-titration do not usually require dose reduction.
A practical guide for up-titrating heart failure medicines
Australian heart failure guidelines are clear on the goal of pharmacological management for people who have heart failure with reduced ejection fraction (HFrEF).
That goal is to prescribe a combination of:1
- an angiotensin-converting enzyme (ACE) inhibitor, or if not tolerated, angiotensin receptor blocker (ARB) and
- a heart failure beta blocker and
- a mineralocorticoid receptor antagonist (MRA)
all at target or maximum tolerated doses.
The guidelines, developed by the National Heart Foundation of Australia and the Cardiac Society of Australia and New Zealand, also describe how to up-titrate these three medicines to reach the target or maximum tolerated doses.1
However GPs can find it challenging to put up-titration into practice.2,3
This article provides a practical guide to assist GPs with the up-titration of heart failure medicines for people with HFrEF.
Download and print
Evidence for a combination of heart failure medicines
The recommendation to prescribe a combination of an ACE inhibitor (or ARB if not tolerated), a heart failure beta blocker and an MRA at target or maximum tolerated doses is based on evidence that these medicines together can help improve quality of life, reduce hospitalisations and save lives for people with HFrEF.1,4
A 2017 meta-analysis found that people with HFrEF who were prescribed a combination of ACE inhibitor, heart failure beta blocker and MRA at target doses had a 56% reduction in all-cause mortality over 1–3 years, compared to placebo. This combination was much more effective than an ACE inhibitor or heart failure beta blocker alone, or a combination of two out of three medicines.6 See Figure 1.
Figure 1: Percentage reduction in all-cause mortality over 1–3 years for people with HFrEF on selected, initial heart failure medicines versus placebo6
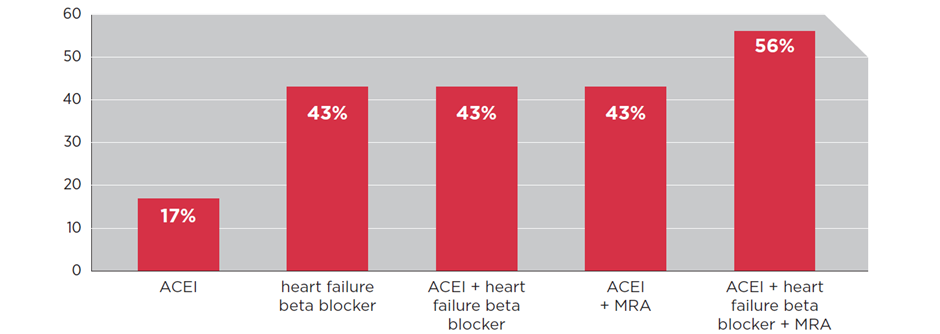
ACEI = angiotensin-converting enzyme inhibitor; ARB = angiotensin receptor blocker; ARNI = angiotensin receptor neprilysin inhibitor; MRA = mineralocorticoid receptor antagonist
Up-titration guidance
The process of up-titration recommended by Australian guidelines to reach the goal of a combination of heart failure medicines is to:1,4
- start each medicine at a low dose and
- gradually up-titrate them to target or maximum tolerated doses.
See Table 1 for start and target doses.
Table 1: Start and target doses for heart failure medicines for people with HFrEF7
Class | Medicine | Start dose | Target dose |
ACEI | captopril enalapril fosinopril lisinopril perindopril arginine perindopril erbumine quinapril ramipril trandolapril | 6.25 mg TDS 2.5 mg D 5 mg D 2.5 mg D 2.5 mg D 2 mg D 5 mg D 2.5 mg BD 0.5 mg D | 75 mg BD 20 mg D 40 mg D 50 mg D 10 mg D 8 mg D 20 mg D 5 mg BD 4 mg D |
ARB | candesartan eprosartan irbesartan losartan olmesartan telmisartan valsartan | 4 mg D 400 mg D 75 mg D 25 mg D 10 mg D 40 mg D 40 mg BD | 32 mg D 600 mg D 300 mg D 100 mg D 40 mg D 80 mg D 160 mg BD |
Heart failure beta blocker | bisoprolol carvedilol metoprolol succinate MR nebivolol | 1.25 mg D 3.125 mg BD 23.75 mg D 1.25 mg D | 10 mg D 50 mg BD 190 mg D 10 mg D |
MRA | eplerenone spironolactone | 25 mg D 25 mg D | 50 mg D 50 mg D |
ARNI | sacubitril/valsartan | 49/51 mg BD | 97/103 mg BD |
HFrEF = Heart failure with reduced ejection fraction; ACEI = angiotensin-converting enzyme inhibitor; ARB = angiotensin receptor blocker; ARNI = angiotensin receptor neprilysin inhibitor; MRA = mineralocorticoid receptor antagonist; D: daily; BD: twice daily; TDS: three times a day; MR: modified release
* From the National Heart Foundation of Australia 2018. Clinical factsheet: pharmacological management of chronic heart failure with reduced left ventricular ejection fraction (HFrEF). Copyright 2019 by National Heart Foundation of Australia. Reprinted with permission.
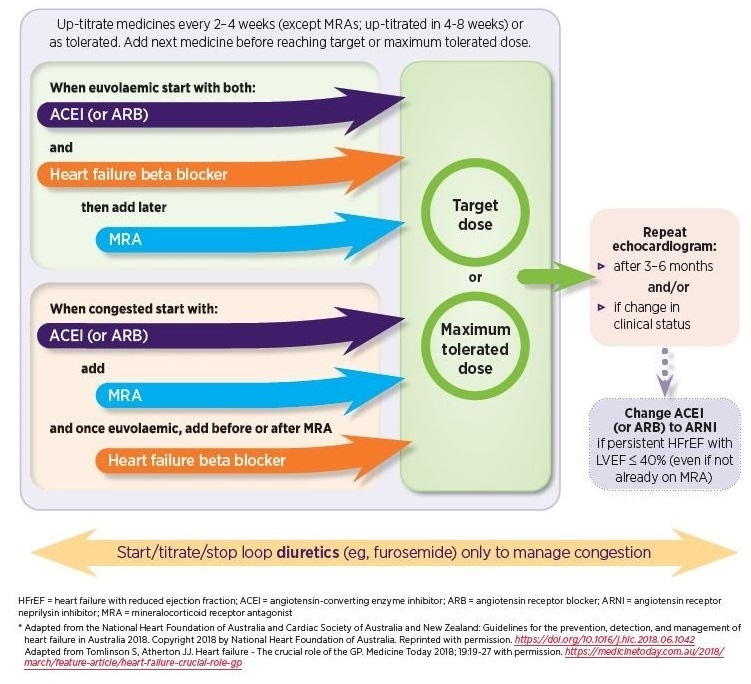
However rather than a single up-titration pathway to reach this goal, there are two pathways that are differentiated by the patient’s volume status when starting pharmacological management; euvolaemic or congested.1 See Figure 2.
For each pathway, it’s recommended to:
- double the dose of heart failure medicines, one at a time, every 2–4 weeks (except MRAs; up-titrated in 4–8 weeks), or as tolerated.1,7,8
- add the next medicine before reaching target or maximum tolerated dose, eg, if the patient is euvolaemic, a heart failure beta blocker may be started before achieving target or maximum tolerated dose of an ACE inhibitor.7
- review every 1–2 weeks after each medicine initiation and dose increase, including a clinical review and checking blood pressure, heart rate, renal function.1,7
In addition, variations during up-titration may be necessary for patients who experience certain adverse effects, particularly those that are symptomatic.1,7
These variations include reducing dosage and pausing up-titration of a medicine with the aim of reducing or stopping the adverse effects to enable up-titration to target dose to be restarted. If adverse effects don’t improve sufficiently, the patient may be considered to have reached maximum tolerated dose.7
See guidance on blood pressure Table 2, heart rate Table 3, renal function Table 4. volume status Table 5, and miscellaneous Table 6.
Further down the pathways, for people with persistent HFrEF with left ventricular ejection fraction (LVEF) ≤ 40%, it’s recommended to change the ACE inhibitor (or ARB) to an angiotensin receptor-neprilysin inhibitor (ARNI).1 See Figure 2.
ACE inhibitors, ARBs and ARNIs may be regarded as a single group of medicines for the purposes of up-titration and adverse effects.1
Figure 2: Initial pharmacological management for people with HFrEF1,7,8
Up-titration is straightforward for most patients
Standard approach and variations that may be required
GPs and cardiologists working together
GPs can play a vital role with initiating and implementing up-titration of heart failure medicines. At the same time patients can benefit from a referral to a cardiologist specialising in heart failure.9
'A shared care approach, where a cardiologist who specialises in heart failure reviews your patient once a year, is a good thing,' says Professor Audehm.
'We wouldn't think twice about an annual review for a patient with, for example, kidney disease. Heart failure has dreadful mortality and morbidity that we can approach with the same intent,' he says.
Indeed, only 50% of people with heart failure are alive 5 years after diagnosis.7 The all-cause readmission rate is 20% and all-cause mortality is 8% 30 days after hospitalisation with heart failure.10
Once the relationship with a cardiologist is established, GPs gain access to the cardiologist’s guidance and management when urgently needed, says Professor Audehm.
‘You can call the cardiologist if a patient is deteriorating and ask them to see the patient. You can receive advice, such as when up-titration is more difficult or challenging, and this will help build your confidence and skills.’
How a GP became confident with up-titration
How to help patients with heart failure medicines
Up-titration can also feel complex for patients who have to ensure they are correctly taking their medicines and self-managing their condition.
A patient-centred approach and keeping information and guidance as simple as possible is recommended by Professor Driscoll.
At the first visit she always includes guidance on daily weighing if the patient is congested, an action plan for when to see the GP again and when to seek emergency care and an explanation of ‘the bigger picture’ of their condition.
Patients find this approach helpful and comforting, and it helps them to achieve the best possible outcomes, says Professor Driscoll.
Blood pressure guidance
Table 2: Guidance for managing blood pressure adverse effects
Blood pressure (BP) – including orthostatic BP (postural drop):11,13 Review 1–2 weeks after each medicine initiation / each medicine dose increase1,7
|
Adverse effects |
Actions a |
||
|
ACEI / ARB / ARNI |
Heart failure beta blocker |
MRA |
|
|
Asymptomatic hypotension7,11 |
Continue therapy |
Continue therapy |
Continue therapy |
|
Symptomatic hypotension eg dizziness, light-headedness and/or confusion1,7,11 |
1) Assess volume status, consider reducing or stopping diuretic if there are no signs or symptoms of congestion 2) Review other medicines that can reduce blood pressure (eg calcium channel blockers, nitrates, diuretics) 3) If still symptomatic: a) temporarily decrease dose of either ACEI/ARB/ARNI or heart failure beta blocker b) review patient within 1 week and if still symptomatic continue dose reduction (or cease) and seek specialist advice |
Continue therapy Only consider decreasing dose if, after implementing actions for ACEI/ARB/ARNI and/or heart failure beta blocker to address symptomatic hypotension, the patient is still symptomatic. |
|
|
Severe symptomatic hypotension / cardiogenic shock eg, cold and sweaty skin, dyspnoea, blue skin tone or weak and rapid pulse1,11,12 |
Immediate referral to an emergency department |
||
a Diuretic dose may be reduced at any time if euvolaemic (unless this has previously exacerbated symptoms).
Expert advice
- Hypotension is common because all of the heart failure medicines tend to lower blood pressure.
- Most patients only experience 2–3 seconds of dizziness when they stand up and, if warned (eg stop and wait for dizziness to subside before start walking), are safe and tolerate it well.
- Advice to reduce risk of falls due to dizziness may also be considered, eg, if experiencing nocturia, turn on the light when going to the toilet.
- Measures to help reduce risk of hypotension include: take medicines at night, avoid dehydration, and if dehydrated and taking diuretic, reduce dosage.
- Symptoms are more important than the actual number (mm Hg) of the systolic blood pressure.
Heart rate guidance
Table 3: Guidance for managing heart rate adverse effects
Heart rate: Review 1–2 weeks after each medicine initiation / each medicine dose increase1,7
|
Adverse effects |
Actions a |
||
|
Heart failure beta blocker |
ACEI/ ARB / ARNI |
MRA |
|
|
Asymptomatic bradycardia (50–60 bpm)1,14,15 |
Continue therapy |
Continue therapy |
Continue therapy |
|
Symptomatic bradycardia (< 50 bpm) eg marked fatigue, dizziness, light-headedness1,11,14 |
1) Arrange ECG to document rhythm 2) Review need for other medicines that can lower heart rate (eg digoxin, amiodarone) 3) If above not successful, may need to decrease dose and seek specialist advice |
Continue therapy |
Continue therapy |
a Diuretic dose may be reduced at any time if euvolaemic (unless this has previously exacerbated symptoms).
Expert advice
- Symptoms are more important than the actual heart rate.
- Symptomatic bradycardia will almost only occur when the heart rate is < 50 bpm.
- Symptomatic bradycardia at 50–60 bpm is usually due to other reasons (eg, hypothyroidism, arrhythmias) and should be investigated before considering an adjustment to the dose of beta blocker.
Renal function guidance
Table 4: Guidance for managing renal function adverse effects
Renal function: Review 1–2 weeks after each medicine initiation / each medicine dose increase1,7
|
Results /Adverse effects |
Actions a |
||
|
MRA |
ACEI / ARB / ARNI |
Heart failure beta blocker |
|
|
eGFR decrease ≤ 30%7 |
Continue therapy |
Continue therapy |
Continue therapy |
|
eGFR decrease > 30%1,7,11 |
1. Assess volume status 2. Review need for other medicines that impact on renal function (eg NSAIDs, diuretics) 3. If above not successful: |
Continue therapy |
|
|
♦ for MRA; decrease dose |
♦ for ACEI/ARB/ARNI: may need to: |
||
|
Hyperkalaemia Serum K+ (potassium) > 5.5 mmol/L1,7 |
1. Assess volume status 2. Review need for other medicines that impact on serum K+ (eg potassium supplements) 3. If above not successful: |
Continue therapy |
|
|
♦ for MRA: decrease dose |
♦ for ACEI/ARB/ARNI: may need to: |
||
|
Hyperkalaemia Serum K+ (potassium) > 6.0 mmol/L1,7 |
♦ for MRA: stop and seek specialist advice |
♦ for ACEI/ARB/ARNI: follow above steps 1, 2, 3 |
|
|
Creatinine increase ≤ 30%1 |
Continue therapy |
Continue therapy |
Continue therapy |
a Diuretic dose may be reduced at any time if euvolaemic (unless this has previously exacerbated symptoms).
Expert advice
- Up-titration does not have to stop because of a change to renal function.
- While MRAs can lead to hyperkalaemia and ACE inhibitors can increase serum potassium and creatinine levels, it is the degree of the change that determines if actions are recommended.
- The main challenge is when there are multiple renal function adverse effects or established renal impairment.
- If unsure how to manage multiple changes to renal function, consider referral to a cardiologist.
- A small rise in creatinine is common after starting an ACE inhibitor; the level will usually return to baseline after around 4 weeks.
Volume status guidance
Table 5: Guidance for managing volume status adverse effects
Volume status: Review 1–2 weeks after each medicine initiation / each medicine dose increase1,14
|
Adverse effects |
Actions |
|||
|
Diuretic |
Heart failure beta blocker |
ACEI / ARB / ARNI |
MRA |
|
|
Congestion (fluid overload, wet) Signs and symptoms include: dyspnoea, peripheral/ sacral oedema, increased jugular venous pressure, weight gain; ≥ 2 kg over 2 days1,16,17 |
If not on a diuretic; start at low dose (eg furosemide 20–40 mg daily) and adjust according to clinical response If on a diuretic; increase dose by 50%–100% with goal of reducing weight by 0.5–1 kg a day If weight continues to increase, seek specialist advice |
If increasing congestion, consider: a) decreasing dose, or b) temporarily stopping if recently started |
Continue therapy |
Continue therapy |
|
Dehydration (over-diuresis, dry) Signs and symptoms include: weight loss; ≥ 2 kg over 2 days, dizziness, thirst, fatigue, reduced urine output, increased urine concentration, orthostatic BP (postural drop)1,16,17 |
If on a diuretic; decrease dose (eg furosemide, reduce by 40 mg) until weight returns to baseline If weight continues to decrease, seek specialist advice |
Continue therapy Closely monitor symptoms Review renal function |
Continue therapy Closely monitor symptoms Review renal function |
Continue therapy Closely monitor symptoms Review renal function |
Expert advice
- Use of diuretics shouldn’t be prioritised over the medicines that decrease mortality; ACE inhibitor/ARB/ARNI, heart failure beta blocker and MRA.
- Diuretic dose may be reduced at any time if euvolaemic (unless this has previously exacerbated symptoms).
- When using diuretics to treat congestion, be sure to use an effective dose, eg, if there is no response or inadequate diuresis (weight reduction) the dose should be doubled (not given BD).
Miscellaneous adverse effects guidance
Table 6: Guidance for managing miscellaneous adverse effects
|
Clinical indicator |
Adverse effects |
Actions |
||
|
ACEI / ARB / ARNI |
Heart failure beta blocker |
MRA |
||
|
Respiratory As part of clinical review after maicine initiation and at each dose up-titration |
Cough dry, non-productive, interfering with quality of life1 |
May change ACEI to ARB |
Continue therapy |
Continue therapy |
|
Allergic reactions As part of clinical review at each dose increase |
Angioedema1 |
Manage the angioedema, stop ACEI, ARB or ARNI and seek specialist advice |
Continue therapy |
Continue therapy |
Conclusion
A combination of an ACE inhibitor (or ARB if not tolerated), a heart failure beta blocker and an MRA up-titrated to target or maximum tolerated doses, can help improve quality of life, reduce hospitalisations and save lives for people with HFrEF.1,4 However GPs can find it challenging to put up-titration into practice.2,3
Following the pathways and guidance described in this article can make it more straightforward for GPs to up-titrate heart failure medicines, particularly for the large group of patients with HFrEF who are relatively well and who usually do not experience adverse effects requiring a change to up-titration doses.
Useful resources
For health professionals
Heart Foundation
Medicine Today
How to optimise therapy for heart failure with reduced ejection fraction
Largely based on the Guidelines for the Prevention, Detection, and Management of Heart Failure in Australia 2018. This article is accessible via a subscription or once-only payment.
For your patients
NPS MedicineWise
Introducing medicines for heart failure
Heart failure: more than just your heart; an action plan for people with heart failure.
How can I take an active role in managing my heart failure?
Heart Foundation
Living well with heart failure:this booklet provides information about what heart failure is, its symptoms, self-management strategies and pharmacological management.
Heart failure: this video series provides information about what is heart failure, pharmacological management, psychological health and self-management.
References
- Atherton JJ, Sindone A, De Pasquale CG, et al. National Heart Foundation of Australia and Cardiac Society of Australia and New Zealand: Guidelines for the prevention, detection, and management of heart failure in Australia 2018. Heart Lung Circ 2018;27:1123-208.
- Howlett J, Comin-Colet J, Dickstein K, et al. Clinical practices and attitudes regarding the diagnosis and management of heart failure: findings from the CORE Needs Assessment Survey. ESC Heart Fail 2018;5:172-83.
- Guirguis K. Prescribed heart failure pharmacotherapy: How closely do GPs adhere to treatment guidelines? Res Social Adm Pharm 2020;16:935-40.
- Cardiovascular Expert Group. Therapeutic Guidelines: Heart failure. East Melbourne: Therapeutic Guidelines Ltd 2018 (accessed 29 October 2020).
- Chan YK, Tuttle C, Ball J, et al. Current and projected burden of heart failure in the Australian adult population: a substantive but still ill-defined major health issue. BMC Health Serv Res 2016;16:501.
- Burnett H, Earley A, Voors AA, et al. Thirty years of evidence on the efficacy of drug treatments for chronic heart failure with reduced ejection fraction: a network meta-analysis. Circ Heart Fail 2017;10:e003529.
- National Heart Foundation of Australia. Pharmacological management of chronic heart failure with reduced left ventricular ejection fraction (HFrEF). Melbourne: NHF, 2019 (accessed 28 October 2020).
- Australian Medicines Handbook. Heart failure. Adelaide: AMH Pty Ltd, 2020 (accessed 27 October 2020).
- Audehm R, Sindone A. Rural Health Webinar Series - Heart Failure. East Melbourne: Royal Australian College of General Practitioners, 2020 (accessed 26 November 2020).
- Al-Omary MS, Davies AJ, Evans TJ, et al. Mortality and readmission following hospitalisation for heart failure in Australia: a systematic review and meta-analysis. Heart Lung Circ 2018;27:917-27.
- Queensland Government Department of Health. Heart Failure Medication Titration Plan. Brisbane: QLD Govt DoH, 2017 (accessed 16 March 2021).
- National Heart Lung and Blood Institute. Low blood pressure. Bethesda, Maryland, USA: US Department of Health & Human Services, 2021 (accessed 9 April 2021).
- Chisholm P, Anpalahan M. Orthostatic hypotension: pathophysiology, assessment, treatment and the paradox of supine hypertension. Intern Med J 2017;47:370-9.
- Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Circulation 2013;128:e240-327.
- Sidhu S, Marine JE. Evaluating and managing bradycardia. Trends Cardiovasc Med 2020;30:265-72.
- National Heart Foundation of Australia. Fluid management algorithm. Melbourne: NHF, 2014 (accessed 19 March 2021).
- Sindone AP, Driscoll A. How to optimise therapy for heart failure with reduced ejection fraction. Medicine Today 2019;20:22-7.
