SUMMARY CMI
Epclusa®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Epclusa?
Epclusa contains the active ingredients sofosbuvir and velpatasvir in a single tablet. Epclusa is used to treat hepatitis C virus infection in adults and children 12 years of age and older and weighing 30 kg or more.
For more information, see Section 1. Why am I using Epclusa? in the full CMI.
2. What should I know before I use Epclusa?
Do not use if you have ever had an allergic reaction to sofosbuvir, velpatasvir or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Epclusa? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Epclusa and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Epclusa?
The usual dose is one Epclusa tablet orally, once daily. Epclusa tablets can be taken with or without food.
More instructions can be found in Section 4. How do I use Epclusa? in the full CMI.
5. What should I know while using Epclusa?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using EPCLUSA? in the full CMI.
6. Are there any side effects?
Common side effects include headache, tiredness, feeling sick (nausea), inflammation of nose and throat (nasopharyngitis).
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
FULL CMI
Epclusa®
Active ingredient(s): sofosbuvir and velpatasvir
Consumer Medicine Information (CMI)
This leaflet provides important information about using Epclusa. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Epclusa.
Where to find information in this leaflet:
1. Why am I using Epclusa?
2. What should I know before I use Epclusa?
3. What if I am taking other medicines?
4. How do I use Epclusa?
5. What should I know while using Epclusa?
6. Are there any side effects?
7. Product details
1. Why am I using Epclusa?
Epclusa contains the active ingredients sofosbuvir and velpatasvir in a single tablet. Epclusa is a direct acting antiviral. This medicine works by lowering the amount of hepatitis C virus in your body and may lead to a cure of your HCV infection over a number of weeks.
Cure means the HCV virus is cleared from your blood (remains at an undetectable level) when measured 3 months after finishing all treatment.
Epclusa does not protect against re-infection with the HCV virus if cure has been achieved.
Epclusa is used to treat hepatitis C virus infection in adults and children 12 years of age and older and weighing 30 kg or more.
Hepatitis C is a virus that infects the liver.
2. What should I know before I use Epclusa?
Warnings
Do not use Epclusa if:
- you are allergic to ingredients sofosbuvir, velpatasvir or any of the ingredients listed at the end of this leaflet.
- you are taking any other medicine that also contains sofosbuvir or velpatasvir
Check with your doctor if you:
- Take any medicines for any other condition
- Have liver problems, other than hepatitis C
- Have a current or previous infection with the hepatitis B virus, since your doctor may want to monitor you more closely.
- Have kidney problems or if you are on haemodialysis.
- Have HIV infection
- Have diabetes
- Have any other medical condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant. Please speak to your doctor if you are unsure. Epclusa may be used with ribavirin. Ribavirin can damage your unborn baby. It is therefore absolutely essential that you (and your partner) take all precautions not to get pregnant if you are taking ribavirin. You and your partner must use an effective birth control method during ribavirin treatment and during the 6 months after completing ribavirin treatment. It is very important that you read the Pregnancy section in the ribavirin product information very carefully.
Talk to your doctor if you are breastfeeding or intend to breastfeed. It is not known whether velpatasvir or sofosbuvir, the two active ingredients of Epclusa, pass into human breast milk.
Use in Children
Epclusa is not recommended for children under 12 years of age or weighing less than 30 kg.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Tell your doctor if you take any of the following medicines:
- amiodarone used to treat heart conditions
- digoxin used to treat heart conditions
- rosuvastatin used to treat high cholesterol
- tenofovir disoproxil fumarate used to treat HIV infection
- efavirenz used to treat HIV infection
- rifampicin, rifapentine, rifabutin (antibiotics used to treat infections, including tuberculosis)
- St. John's Wort (Hypericum perforatum – herbal medicine used to treat depression)
- carbamazepine, phenytoin, phenobarbital (medicines used to treat epilepsy and prevent seizures)
- Warfarin or other similar medicines called vitamin K antagonists used to thin the blood
Epclusa may interact with these medicines. As a result, the amounts of Epclusa or other medicines in your blood may be affected. This may stop your medicines from working properly, or make any side effects worse. In some cases, your doctor may need to give you a different medicine or adjust the dose of medicine you are taking.
Get advice from a doctor or pharmacist if you take medicines used to treat stomach ulcers, heartburn or acid reflux. These medicines include:
- antacids (e.g. aluminium hydroxide or magnesium hydroxide)
- Proton pump inhibitors (e.g. omeprazole)
- H2-antagonists (e.g. famotidine)
These medicines can decrease the amount of velpatasvir in your blood. If you are taking one of these medicines your doctor will either give you a different medicine for stomach ulcers, heartburn, or acid reflux, or recommend how and when you take that medicine.
- If you are taking an antacid, take it at least 4 hours before or at least 4 hours after Epclusa.
- If you are taking a proton pump inhibitor, take Epclusa with food. Your doctor may give you a different medicine or adjust the dose of medicine you are taking.
- If you are taking an H2-antagonist, your doctor may give you a different medicine or adjust the dose of the medicine you are taking.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Epclusa.
4. How do I use Epclusa?
How much to take
- The usual dose is one Epclusa tablet orally, once daily.
- Epclusa tablets can be taken with or without food.
- Follow the instructions provided and use Epclusa until your doctor tells you to stop.
When to take Epclusa
- Epclusa should be taken at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
If you forget to use Epclusa
Epclusa should be used regularly at the same time each day. If you miss your dose at the usual time, take your missed dose right away unless it is almost time for your next dose.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed. Continue with your regular dosing schedule.
Do not stop taking Epclusa unless your doctor tells you to. It is very important that you complete the full course of treatment to give the medicine the best chance to cure your hepatitis C virus infection.
If you use too much Epclusa
If you think that you have used too much Epclusa, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
by calling 13 11 26 (Australia), or 0800 764 766 (New Zealand) - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Epclusa?
Things you should do
Remind any doctor, dentist or pharmacist you visit that you are using Epclusa.
Tell your doctor as soon as possible if there is any worsening of your condition
Things you should not do
- Do not give Epclusa to anyone else, even if they have the same condition as you.
- Do not stop using this medicine without checking with your doctor
- Do not breastfeed.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Epclusa affects you.
Looking after your medicine
- Keep your Epclusa tablets in the bottle with the cap tightly closed until you take them. If you take Epclusa tablets out of their pack they may not keep well.
- Store Epclusa in a cool, dry place where it stays below 30°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight.
Do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Signs of allergic reaction such as:
| Do not take any more Epclusa. Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Epclusa contains
| Active ingredient (main ingredient) | sofosbuvir velpatasvir |
| Other ingredients (inactive ingredients) | copovidone microcrystalline cellulose croscarmellose sodium magnesium stearate polyvinyl alcohol macrogol 3350 titanium dioxide talc-purified iron oxide red |
| Potential allergens | n/a |
Do not take this medicine if you are allergic to any of these ingredients.
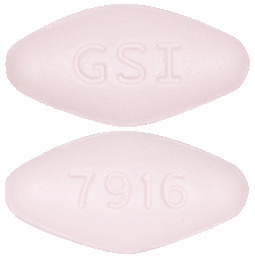
What Epclusa looks like
Epclusa tablets are diamond-shaped and pink in colour. Each tablet has “GSI” on one side and “7916” on the other side of the tablet.
Epclusa tablets are supplied in bottles containing 28 tablets.
AUST R 266823
Who distributes Epclusa
Australia
Gilead Sciences Pty Ltd
Level 6, 417 St Kilda Road Melbourne, Victoria 3004
New Zealand
c/- Grant Thornton New Zealand Limited,
L4, 152 Fanshawe Street
Auckland 1010
This leaflet was prepared in February 2022.
Epclusa, 7916, and GSI are trademarks of Gilead Sciences, Inc. or one of its related companies. Other brands listed are trademarks of their respective owners and are not trademarks of Gilead Sciences, Inc.
Published by MIMS April 2022





 The presence of 40% human serum had no effect on the anti-HCV activity of sofosbuvir, but reduced the anti-HCV activity of velpatasvir by 13-fold against genotype 1a HCV replicons.
The presence of 40% human serum had no effect on the anti-HCV activity of sofosbuvir, but reduced the anti-HCV activity of velpatasvir by 13-fold against genotype 1a HCV replicons. Among the 75 genotype 1 patients who had baseline NS5A RAVs, SVR12 was 97% (67/69) and 100% (6/6) in patients with baseline NS5A RAVs that confer ≤ 100-fold and > 100-fold reduced susceptibility to velpatasvir, respectively. Among the 43 genotype 3 patients who had baseline NS5A RAVs, SVR12 was 94% (15/16) and 85% (23/27) in patients with NS5A RAVS that confer ≤ 100-fold and > 100-fold reduced susceptibility to velpatasvir, respectively. The 4 genotype 3 patients who had baseline NS5A RAVs conferring > 100-fold reduced susceptibility to velpatasvir and failed to achieve SVR12, all had NS5A substitution Y93H at baseline. Twenty one of 25 (84%) genotype 3 patients with baseline NS5A substitution Y93H achieved SVR12.
Among the 75 genotype 1 patients who had baseline NS5A RAVs, SVR12 was 97% (67/69) and 100% (6/6) in patients with baseline NS5A RAVs that confer ≤ 100-fold and > 100-fold reduced susceptibility to velpatasvir, respectively. Among the 43 genotype 3 patients who had baseline NS5A RAVs, SVR12 was 94% (15/16) and 85% (23/27) in patients with NS5A RAVS that confer ≤ 100-fold and > 100-fold reduced susceptibility to velpatasvir, respectively. The 4 genotype 3 patients who had baseline NS5A RAVs conferring > 100-fold reduced susceptibility to velpatasvir and failed to achieve SVR12, all had NS5A substitution Y93H at baseline. Twenty one of 25 (84%) genotype 3 patients with baseline NS5A substitution Y93H achieved SVR12. The single genotype 3 patient who had baseline NS5A RAVs and failed to achieve SVR12 had NS5A substitution Y93H at baseline; pharmacokinetic data from this patient was consistent with nonadherence.
The single genotype 3 patient who had baseline NS5A RAVs and failed to achieve SVR12 had NS5A substitution Y93H at baseline; pharmacokinetic data from this patient was consistent with nonadherence. The ribavirin dose was weight based (1000 mg daily administered in two divided doses for patients < 75 kg and 1200 mg for those ≥ 75 kg) and administered in two divided doses when used in combination with sofosbuvir in the ASTRAL-2 and ASTRAL-3 trials or in combination with Epclusa in the ASTRAL-4 trial. RBV dose adjustments were performed according to the RBV product information. Serum HCV RNA values were measured during the clinical trials using the COBAS AmpliPrep/ COBAS Taqman HCV test (version 2.0) with a lower limit of quantification (LLOQ) of 15 IU per mL. Sustained virologic response (SVR12), defined as HCV RNA less than LLOQ at 12 weeks after the cessation of treatment, was the primary endpoint to determine the HCV cure rate.
The ribavirin dose was weight based (1000 mg daily administered in two divided doses for patients < 75 kg and 1200 mg for those ≥ 75 kg) and administered in two divided doses when used in combination with sofosbuvir in the ASTRAL-2 and ASTRAL-3 trials or in combination with Epclusa in the ASTRAL-4 trial. RBV dose adjustments were performed according to the RBV product information. Serum HCV RNA values were measured during the clinical trials using the COBAS AmpliPrep/ COBAS Taqman HCV test (version 2.0) with a lower limit of quantification (LLOQ) of 15 IU per mL. Sustained virologic response (SVR12), defined as HCV RNA less than LLOQ at 12 weeks after the cessation of treatment, was the primary endpoint to determine the HCV cure rate.
 Treatment with Epclusa for 12 weeks demonstrated statistical superiority (p = 0.018) compared to treatment with SOF + RBV for 12 weeks (treatment difference +5.2%; 95% confidence interval: +0.2% to +10.3%).
Treatment with Epclusa for 12 weeks demonstrated statistical superiority (p = 0.018) compared to treatment with SOF + RBV for 12 weeks (treatment difference +5.2%; 95% confidence interval: +0.2% to +10.3%). Treatment with Epclusa for 12 weeks demonstrated statistical superiority (p < 0.001) compared to treatment with SOF + RBV for 24 weeks (treatment difference +14.8%; 95% confidence interval: +9.6% to +20.0%).
Treatment with Epclusa for 12 weeks demonstrated statistical superiority (p < 0.001) compared to treatment with SOF + RBV for 24 weeks (treatment difference +14.8%; 95% confidence interval: +9.6% to +20.0%).
 Table 14 presents the virologic outcome for patients with genotype 1 or 3 HCV infection in the ASTRAL-4 trial.
Table 14 presents the virologic outcome for patients with genotype 1 or 3 HCV infection in the ASTRAL-4 trial. Changes in MELD and CPT score from baseline to post-treatment week 12 were analysed for patients who achieved SVR12 to assess the effect of SVR12 on hepatic function. Of the 82 patients treated with Epclusa + RBV for 12 weeks who achieved SVR12, 81 had MELD and CPT assessments at baseline and post-treatment week 12.
Changes in MELD and CPT score from baseline to post-treatment week 12 were analysed for patients who achieved SVR12 to assess the effect of SVR12 on hepatic function. Of the 82 patients treated with Epclusa + RBV for 12 weeks who achieved SVR12, 81 had MELD and CPT assessments at baseline and post-treatment week 12. No patient had HIV-1 rebound during the study, and CD4+ counts were stable during treatment.
No patient had HIV-1 rebound during the study, and CD4+ counts were stable during treatment. The pharmacokinetics of sofosbuvir was studied in HCV negative adult patients with mild (eGFR ≥ 50 and < 80 mL/min/1.73 m2), moderate (eGFR ≥ 30 and < 50 mL/min/1.73 m2), severe renal impairment (eGFR < 30 mL/min/1.73 m2) and patients with ESRD requiring haemodialysis following a single 400 mg dose of sofosbuvir, relative to patients with normal renal function (eGFR > 80 mL/min/1.73 m2). GS-331007 is efficiently removed by haemodialysis with an extraction coefficient of approximately 53%. Following a single 400 mg dose of sofosbuvir, a 4 hour haemodialysis removed 18% of administered dose.
The pharmacokinetics of sofosbuvir was studied in HCV negative adult patients with mild (eGFR ≥ 50 and < 80 mL/min/1.73 m2), moderate (eGFR ≥ 30 and < 50 mL/min/1.73 m2), severe renal impairment (eGFR < 30 mL/min/1.73 m2) and patients with ESRD requiring haemodialysis following a single 400 mg dose of sofosbuvir, relative to patients with normal renal function (eGFR > 80 mL/min/1.73 m2). GS-331007 is efficiently removed by haemodialysis with an extraction coefficient of approximately 53%. Following a single 400 mg dose of sofosbuvir, a 4 hour haemodialysis removed 18% of administered dose.

 Sofosbuvir is a white to off white powder with a solubility of ≥ 2 mg/mL across the pH range of 2-7.7 at 37°C. The partition coefficient (log P) for sofosbuvir is 1.62 and the pKa is 9.3.
Sofosbuvir is a white to off white powder with a solubility of ≥ 2 mg/mL across the pH range of 2-7.7 at 37°C. The partition coefficient (log P) for sofosbuvir is 1.62 and the pKa is 9.3. Velpatasvir is practically insoluble (less than 0.1 mg/mL) above pH 5, slightly soluble (3.6 mg/mL) at pH 2, and soluble (greater than 36 mg/mL) at pH 1.2.
Velpatasvir is practically insoluble (less than 0.1 mg/mL) above pH 5, slightly soluble (3.6 mg/mL) at pH 2, and soluble (greater than 36 mg/mL) at pH 1.2.