What is in this leaflet
This leaflet answers some common questions about ABRAXANE.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you being given ABRAXANE against the benefits they expect it will have for you.
If you have any concerns about being given this medicine, ask your doctor. Your doctor or pharmacist can provide you with more information about ABRAXANE.
Keep this leaflet You may need to read it again.
What ABRAXANE is used for
This medicine is used to treat:
- Breast cancer
- Lung Cancer
- Pancreatic Cancer
This medicine belongs to a group of medicines called antineoplastic or cytotoxic medicines. You may also hear of these being called chemotherapy medicines.
It works by killing cancer cells and stopping them from multiplying.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is not addictive.
This medicine is available only with a doctor’s prescription.
Before you are given ABRAXANE
When you must not be given it
You must not be given ABRAXANE if you have an allergy to:
- any medicine containing paclitaxel
- any of the ingredients listed at the end of this leaflet
Some of the symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty breathing; swelling of the face, lips, tongue or other parts of the body; rash, itching or hives on the skin.
You must not be given this medicine if you have a very low white blood cell (WBC) count.
Tell your doctor if you have an infection or high temperature. Your doctor may decide to delay your treatment until the infection is gone. A mild illness, such as a cold, is not usually a reason to delay treatment.
You must not be given this medicine if you have severe liver damage. Tell your doctor you have liver disease or damage.
You must not be given this medicine if you are pregnant or plan to become pregnant. Like most cytotoxic medicines ABRAXANE is not recommended for use during pregnancy. If there is any need to consider this medicine during your pregnancy, your doctor will discuss with you the benefits and risks of using it.
Males: tell your doctor if your partner plans to become pregnant while you are being treated with this medicine or shortly after you have stopped treatment with it. ABRAXANE may cause birth defects if either the male or female is being treated with it at the time of conception. It is recommended that you use some kind of birth control while you are being treated with ABRAXANE and for at least 12 weeks after you stop using it. Your doctor will discuss this with you.
Do not breast-feed if you are taking this medicine. The active ingredient in ABRAXANE passes into breast milk and there is a possibility that your baby may be affected.
If you are not sure whether you should be given this medicine, talk to your doctor.
The safety and effectiveness of ABRAXANE in children (under 18 years) has not been established.
Before you are given it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- kidney disease
- liver disease
- heart problems
- any blood disorder with a reduced number of blood cells, or platelets
- any disease of the nerves
- lowered immunity due to diseases such as HIV/AIDS
- lowered immunity due to treatment with medicines such as cyclosporine, or other medicines used to treat cancer (including radiation therapy)
If you have not told your doctor about any of the above, tell him/her before you start taking ABRAXANE.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and ABRAXANE may interfere with each other.
Some medicines may be affected by ABRAXANE or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How to take ABRAXANE
How much is given
Your doctor will decide what dose of ABRAXANE you will receive. This depends on your condition and other factors, such as your weight, kidney function and other chemotherapy medicines you are being given.
ABRAXANE may be given alone or in combination with other drugs.
Several courses of ABRAXANE therapy may be needed depending on your response to treatment.
Additional treatment may not be repeated until your blood cell numbers return to acceptable levels and any uncontrolled effects have been controlled.
Ask your doctor if you want to know more about the dose of ABRAXANE you receive.
How it is given
ABRAXANE is usually given as an infusion (drip) into a vein over 30 minutes.
How long it will be given for
Breast Cancer:
ABRAXANE is usually given once every three weeks (i.e. on day 1 of a 21-day cycle). Each infusion is called one 'cycle' of chemotherapy. Your doctor will decide how many of these cycles you will need.
Lung Cancer:
ABRAXANE is given every week (i.e. on days 1, 8 and 15 of each 21-day cycle). Another medicine called carboplatin is also given on day 1 only of each 21-day cycle.
Your doctor will decide how many of these cycles you will need.
Pancreatic Cancer:
ABRAXANE is given every week (i.e. on days 1, 8 and 15 of each 28-day cycle). Every 28 days is called one ‘cycle’ of chemotherapy.
Another medicine called gemcitabine is also given on days 1, 8 and 15 after ABRAXANE has been given. Your doctor will decide how many of these cycles you will need.
Overdose
As ABRAXANE is given to you under the supervision of your doctor, it is very unlikely that you will receive too much. However, if you experience severe side effects after being given this medicine, tell your doctor or nurse immediately. You may need urgent medical attention.
Symptoms of an ABRAXANE overdose include the side effects listed below in the 'Side Effects' section, but are usually of a more severe nature.
While you are being given ABRAXANE
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are being treated with ABRAXANE.
Tell any other doctors, dentists, and pharmacists who are treating you that you are being given this medicine.
If you are going to have surgery, tell the surgeon or anaesthetist that you are being treated with this medicine. It may affect other medicines used during surgery.
If you become pregnant while you are being treated with this medicine, tell your doctor immediately.
Keep all your doctor's appointments so your progress can be checked. Your doctor may want to check your blood pressure and do some blood and other tests from time to time to check on your progress and detect any unwanted side effects.
Keep follow-up appointments with your doctor. It is important to have your follow-up cycles of ABRAXANE at the appropriate times to get the best effects from your treatments.
This medicine can lower the number of white blood cells and platelets in your blood. This means that you have an increased chance of getting an infection or bleeding.
The following precautions should be taken to reduce your risk of infection or bleeding:
- Avoid people who have infections. Check with your doctor immediately if you think you may be getting an infection, or if you get a fever, chills, cough, hoarse throat, lower back or side pain or find it painful or difficult to urinate.
- Be careful when using a toothbrush, toothpick or dental floss. Your doctor, dentist, nurse or pharmacist may recommend other ways to clean your teeth and gums. Check with your doctor before having any dental work.
- Be careful not to cut yourself when you are using sharp objects such as a razor or nail cutters.
- Avoid contact sports or other situations where you may bruise or get injured.
Your body breaks down ABRAXANE and uses it to fight cancer. The breakdown products may be excreted in body fluids and waste, including blood, urine, faeces, vomitus and semen.
In general, precautions to protect other people should be taken while you are receiving chemotherapy and for one week after the treatment period by:
- Flushing the toilet twice to dispose of any body fluids and waste.
- Wearing gloves to clean any spill of body fluid or waste. Use paper towels or old rags, a strong solution of non-bleaching detergent and large amounts of water to mop up the spill. Discard the towels or rags into a separate waste bag and dispose of fluids in the toilet.
- Wash linen or clothing that is heavily contaminated by body fluids or waste separately from other items. Use a strong solution of non-bleaching detergent and large amounts of water.
- Place soiled disposable nappies and other pads in a plastic bag, seal and dispose into the garbage.
- For sexual intercourse, use a barrier method such as a condom.
Things to be careful of
Be careful driving or operating machinery until you know how ABRAXANE affects you. There is no known reason why you cannot continue driving or using machines between courses of ABRAXANE unless you feel tired or dizzy.
If you are given other medication as part of your treatment, you should ask your doctor for advice on driving and using machines.
Side effects
Tell your doctor, nurse or pharmacist as soon as possible if you do not feel well while you are being given ABRAXANE.
Like other medicines that treat cancer, ABRAXANE may have unwanted side effects, some of which may be serious. You may need medical treatment if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- muscle or joint pain on the arms and legs
- nausea and vomiting
- hair loss
- diarrhoea
- changes in skin or nail appearance
- soreness or ulceration of the mouth
- tiredness
The above list includes the more common side effects of your medicine.
Tell your doctor or nurse as soon as possible if you notice any of the following:
- pain, swelling, irritation and redness at the injection site
- flushing
- light-headedness, dizziness or fainting (due to low blood pressure)
- numbness or tingling in the fingers and/or toes
- changes in vision
- abdominal pain
The above list includes serious side effects which may require medical attention.
If any of the following happen, tell your doctor or nurse immediately, or if you are not in hospital, go to an Accident and Emergency Room at your nearest hospital:
- shortness of breath, coughing, wheezing or difficulty breathing
- swelling of the face, lips, tongue, or other parts of the body
- rash, itching or hives on the skin
- extreme weakness or tiredness
- seizures (fits)
- fast, slow or irregular heart beat
- chest pain
- yellowing of the skin or eyes
- unusual bleeding or bruising (including blood in your stools or urine)
- fever, sore throat or other signs of infection
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation.
Tell your doctor, nurse or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
The benefits and side effects of ABRAXANE may take some time to occur. Therefore, even after you have finished receiving your treatment, you should tell your doctor immediately if you notice any of the side effects listed in this section.
After being given ABRAXANE
Storage
ABRAXANE will be stored in the pharmacy or on the ward. The injection is kept in a cool, dry place, protected from light, where the temperature stays below 25°C.
Product description
What it looks like:
ABRAXANE is a white to yellow solid powder cake in a clear glass vial.
When it is reconstituted it will be a milky liquid.
Ingredients
Each vial of ABRAXANE contains either 100 mg or 250 mg of paclitaxel as the active ingredient, and human albumin solution (which contains sodium, sodium octanoate and sodium acetyltryptophanate).
This medicine does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
Sponsor
In Australia:
Abraxis BioScience Australia Pty Ltd
Level 2, 4 Nexus Court
Mulgrave, VIC 3170
Australia
Telephone: 1800 CELGENE (1800 235 4363)
Distributed by Specialised Therapeutics Australia Pty Ltd
In New Zealand:
Celgene Limited
PO Box 3035
Wellington,
New Zealand
Telephone: 0800 526 529
100 mg vial AUST R 133500
250 mg vial AUST R 297973
The 250 mg presentation is not approved by Medsafe and is therefore not marketed in New Zealand.
Abraxis BioScience Australia Pty Ltd is an affiliate of Celgene Corporation
This leaflet was prepared in May 2023
Published by MIMS July 2023



 Dose recommendation and modifications for neutropenia and thrombocytopenia at the start of a cycle or within a cycle for patients with metastatic adenocarcinoma of the pancreas are provided in Table 4.
Dose recommendation and modifications for neutropenia and thrombocytopenia at the start of a cycle or within a cycle for patients with metastatic adenocarcinoma of the pancreas are provided in Table 4. Dose modifications for other adverse drug reactions in patients with metastatic adenocarcinoma of the pancreas are provided in Table 5.
Dose modifications for other adverse drug reactions in patients with metastatic adenocarcinoma of the pancreas are provided in Table 5.
 1.
1. 

 Additional clinically relevant adverse reactions that were reported in ≥ 1% and < 5% of the non-small cell lung cancer patients who received Abraxane and carboplatin included:
Additional clinically relevant adverse reactions that were reported in ≥ 1% and < 5% of the non-small cell lung cancer patients who received Abraxane and carboplatin included: Table 11 provides the frequency and severity of adverse reactions by system organ class/preferred term that have been reported in ≥ 10% of patients with metastatic adenocarcinoma of the pancreas who received Abraxane and gemcitabine or gemcitabine monotherapy. Within each system organ class grouping, adverse reactions are presented in order of decreasing frequency.
Table 11 provides the frequency and severity of adverse reactions by system organ class/preferred term that have been reported in ≥ 10% of patients with metastatic adenocarcinoma of the pancreas who received Abraxane and gemcitabine or gemcitabine monotherapy. Within each system organ class grouping, adverse reactions are presented in order of decreasing frequency.



 Patients received a median of 6 cycles of treatment in both study arms. For the treated population, the median cumulative paclitaxel dose and the median average paclitaxel dose intensity were higher with Abraxane administered weekly (1325.0 mg/m2 and 81.9 mg/m2/week, respectively) relative to solvent-based paclitaxel administered every 3 weeks (1125.0 mg/m2 and 65.1 mg/m2/week, respectively). The median cumulative carboplatin dose and the median average carboplatin dose intensity were lower for the Abraxane and carboplatin regimen (3140.5 mg and 166.1 mg/week, respectively) relative to the solvent based paclitaxel and carboplatin regimen (3315.0 mg and 203.6 mg/week, respectively).
Patients received a median of 6 cycles of treatment in both study arms. For the treated population, the median cumulative paclitaxel dose and the median average paclitaxel dose intensity were higher with Abraxane administered weekly (1325.0 mg/m2 and 81.9 mg/m2/week, respectively) relative to solvent-based paclitaxel administered every 3 weeks (1125.0 mg/m2 and 65.1 mg/m2/week, respectively). The median cumulative carboplatin dose and the median average carboplatin dose intensity were lower for the Abraxane and carboplatin regimen (3140.5 mg and 166.1 mg/week, respectively) relative to the solvent based paclitaxel and carboplatin regimen (3315.0 mg and 203.6 mg/week, respectively). The effect of prognostic factors on the primary efficacy endpoint of overall response rate was pre-specified. Two prognostic factors showed a significant interaction (defined as p ≤ 0.10) with treatment effect on overall response rate: (1) time interval from primary diagnosis to randomisation and (2) histology (see Table 17). There was no interaction between the variable and the treatment effect as measured by overall response rate for the following baseline factors: region, gender, race, age, smoking status, baseline ECOG status, stage at primary diagnosis, time from date of first documented metastasis/relapse to date of study entry, stage at current diagnosis, and number of lesions.
The effect of prognostic factors on the primary efficacy endpoint of overall response rate was pre-specified. Two prognostic factors showed a significant interaction (defined as p ≤ 0.10) with treatment effect on overall response rate: (1) time interval from primary diagnosis to randomisation and (2) histology (see Table 17). There was no interaction between the variable and the treatment effect as measured by overall response rate for the following baseline factors: region, gender, race, age, smoking status, baseline ECOG status, stage at primary diagnosis, time from date of first documented metastasis/relapse to date of study entry, stage at current diagnosis, and number of lesions.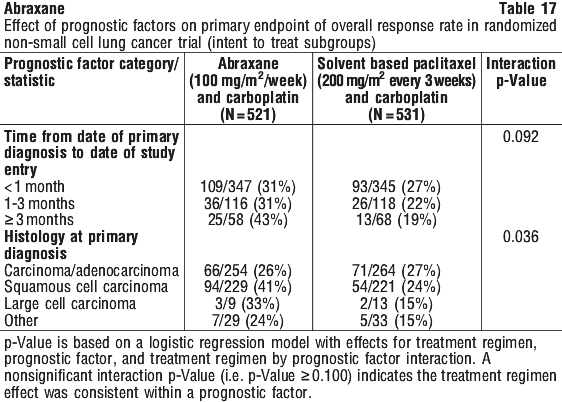
 There was a statistically significant improvement in OS for patients treated with Abraxane/gemcitabine versus gemcitabine alone, with 1.8 months increase in median OS, 28% overall reduction in risk of death, 59% improvement in 1-year survival, and 125% improvement in 2-year survival rates.
There was a statistically significant improvement in OS for patients treated with Abraxane/gemcitabine versus gemcitabine alone, with 1.8 months increase in median OS, 28% overall reduction in risk of death, 59% improvement in 1-year survival, and 125% improvement in 2-year survival rates. There was a statistically significant improvement in PFS for patients treated with Abraxane/gemcitabine versus gemcitabine alone, with 1.8 months increase in median PFS, 31% overall reduction in risk of progression or death, 76% improvement in 6 month risk of progression or death, and 78% improvement in 12-month risk of progression or death.
There was a statistically significant improvement in PFS for patients treated with Abraxane/gemcitabine versus gemcitabine alone, with 1.8 months increase in median PFS, 31% overall reduction in risk of progression or death, 76% improvement in 6 month risk of progression or death, and 78% improvement in 12-month risk of progression or death.


