What is in this leaflet
This leaflet answers some common questions about ACARIZAX®. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking ACARIZAX® against the benefits this medicine is expected to have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What ACARIZAX® is used for
ACARIZAX® contains an allergen extract from house dust mites. It comes in a tablet form which is intended to be absorbed in the body by placing it under the tongue.
ACARIZAX® is for the treatment of house dust mite allergy that is characterised by rhinitis (sneezing, runny or itchy nose, nasal congestion) in adults and adolescents aged 12 years and above. It is also for the treatment of house dust mite related allergic asthma in adults.
ACARIZAX® is not recommended for use in children below 12 years of age.
ACARIZAX® works by increasing your immune tolerance to house dust mites leading to reduction in the severity of your allergic symptoms over time.
Before starting treatment, the doctor will check if ACARIZAX® is suitable for you by performing the appropriate blood and/or skin prick tests.
ACARIZAX® is only available with a prescription from the doctor.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is not expected to affect your ability to drive a car or operate machinery.
Before you take ACARIZAX®
When you must not take it
Do not take ACARIZAX® if:
- you have an allergy to any of the ‘Other ingredients’ listed at the end of this leaflet
- you have poor lung function (as assessed by your doctor)
- your asthma has worsened over the last 3 months (as assessed by your doctor)
- you have asthma as well as an ongoing airway infection such as a cold or chest infection on the day you are to take the first dose of ACARIZAX®. Your doctor may recommend delaying the start of your treatment until your cold or infection is better
- you have an illness which affects your immune system or have cancer
- you have recently lost or had a tooth taken out had other forms of mouth surgery, have ulcers, wounds, or infections in the mouth (refer to Before you start to take it).
Do not take the medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering.
Before you start to take it
Tell your doctor if you:
- have an allergy to any of the ‘Other ingredients’ listed at the end of this leaflet
- are being treated for depression, asthma, Parkinson’s disease or have a heart condition such as high blood pressure or an irregular heartbeat (refer to Taking other medicines)
- have previously been treated for house dust mite allergy and had a severe allergic reaction, have recently lost or had a tooth taken out, had other forms of mouth surgery, are planning to have any form of mouth surgery, have ulcers, wounds or infections in the mouth. Your doctor may delay or stop your treatment until your condition has resolved
- are recovering or are in remission from an illness which affects your immune system
- have an inflamed oesophagus
- are taking steroids
- are pregnant or intend to become pregnant during treatment
- are breast-feeding or intend to breast-feed during treatment.
Taking other medicines
Tell your doctor if you are taking any other medicines including any that you get without a prescription from your pharmacy, supermarket or health food shop. Some medicines and ACARIZAX® may interfere with each other.
Ask your doctor for more information on medicines to be careful of or avoid while taking ACARIZAX®.
How to take ACARIZAX®
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions in this leaflet, ask your doctor or pharmacist for help.
How much to take
The recommended dose of ACARIZAX® is one tablet per day. Each tablet contains 12 SQ-HDM standardised allergen extract from the house dust mites Dermatophagoides farinae and Dermatophagoides pteronyssinus.
However your doctor may recommend a different dose for you. Always follow the instructions given to you by your doctor or pharmacist.
How to take it
Make sure your hands are clean and dry before handling the medicine. Follow the instructions below when taking ACARIZAX®:
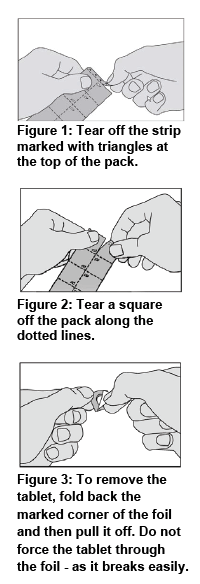
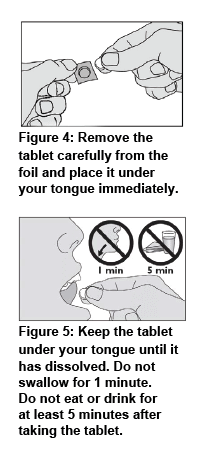
Your doctor should give you the first tablet under medical supervision and then monitor you for 30 minutes. This gives you the opportunity to discuss any possible side effects with your doctor (refer to Side effects).
When to take it
Take your medicine at about the same time each day. Taking it at the same time each day will help you get the best effect. It will also help you to remember when to take it.
Do not consume food and drink for 5 minutes after taking this medicine. Food can interfere with the absorption of this medicine.
How long to take it
Continue taking the medicine for as long as your doctor tells you to.
Do not stop taking the medicine until your doctor tells you - even if you feel better. If you do not take this medicine as prescribed, you may not get the full effect of the treatment.
If you forget to take it
If you forget to take a dose, take it later in the day.
Do not take a double dose to make up for the dose you missed.
If you have not taken ACARIZAX® for more than 7 days contact your doctor before taking ACARIZAX® again.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or Poisons Information Centre (telephone 13 1126) for advice, or go to Accident and Emergency at the nearest hospital if you think that you or anyone else may have taken too much ACARIZAX®. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are using ACARIZAX®
Things you must do
If you are about to be started on any new medicine, inform your doctor or pharmacist that you are taking ACARIZAX®.
Tell any other doctors, dentists, and pharmacists who treat you that you are taking this medicine.
If you become pregnant while taking this medicine, tell your doctor immediately.
You can expect some mild to moderate localised allergic reactions during your treatment. In most cases these reactions are only temporary. However, if you think these are severe, talk to your doctor to see if you need any anti-allergic medicines such as antihistamines (refer to Side effects).
If you have not taken ACARIZAX® for more than 7 days you should contact your doctor before taking ACARIZAX® again.
Tell your doctor if you feel the treatment is not helping your condition.
Things you must not do
Do not use ACARIZAX® to treat any other conditions unless your doctor tells you to.
Do not give this medicine to anyone else even if they have the same conditions as you.
Do not take this medicine if you are below 12 years of age.
Do not stop taking your medicine without checking with your doctor. If you stop taking it suddenly, your condition may worsen or you may have unwanted side effects.
Side effects
Tell your doctor or pharmacist if you have any unpleasant effects while taking ACARIZAX® even if you do not think the effect is connected with this medicine.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need to seek medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- Irritation of the throat
- Dryness or redness of the mouth
- Itching of the mouth, tongue, lips, eyes or ears
- Inflammation of the nose and throat
- Taste disturbance or loss of taste
- Runny nose, blocked nose or sneezing
Tell your doctor as soon as possible if you notice any of the following:
- Prickling sensation or numbness of the mouth or tongue
- Itching, redness, watering of the eyes
- Swelling of the mouth, throat, tongue or lips
- Inflammation, discomfort or burning sensation in the mouth
- Stomach pain or discomfort
- Diarrhoea
- Feeling sick (nausea) or vomiting
- Pain when swallowing or difficulty in swallowing
- Indigestion or heartburn
- Hoarseness
- Blistering of the mouth and mouth ulcers
- Fast or irregular heartbeat
- Sensation of something stuck in the throat
- Tightness in the chest
- Feeling tired or dizzy
- Itching or redness of the skin, hives
- Ear pain
In most cases these reactions are only temporary.
If any of the following happen, stop taking ACARIZAX® and tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- Worsening of existing asthma
- Rapid swelling of face, mouth, throat or skin
- Difficulties in swallowing or breathing
- Voice changing
- Start to feel faint
- Feeling of fullness in the throat (like swelling)
- Tightness in the throat.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
After using ACARIZAX®
Storage
Store the blister pack in the carton to protect the tablets from light.
Keep it in a cool dry place where the temperature stays below 25°C.
Do not store ACARIZAX®, or any other medicine, in the bathroom or near a sink. Do not leave it in the car on hot days. Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking the tablets, or the tablets have passed their expiry date, ask your pharmacist what to do with any ACARIZAX® that is left over.
Product description
What ACARIZAX® looks like
ACARIZAX® tablets are white to off-white with an imprint on one side. The tablets are individually packed in blister foils.
The following pack sizes are available: 10, 30 or 90 tablets.
Not all pack sizes may be marketed.
Ingredients
Active ingredients
Each ACARIZAX® tablet contains 12 SQ-HDM standardised allergen extract from the house dust mites Dermatophagoides farinae and Dermatophagoides pteronyssinus as the active ingredient.
Other ingredients
The tablet also contains:
- gelatin (from fish)
- mannitol
- sodium hydroxide.
This medicine does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
Sponsor
ACARIZAX® is sponsored in Australia by:
Seqirus Pty Ltd
ABN: 26 160 735 035
63 Poplar Road Parkville
VIC 3052 Australia
Telephone: 1800 642 865
www.seqirus.com.au
Australian Registration Number
AUST R 250392
This leaflet was prepared on:
13 May 2020
ACARIZAX® is a registered trademark of ALK- Abelló A/S used under license
Published by MIMS July 2020


 In the pooled phase II/III adult studies, time to onset from first administration for oral pruritus, throat irritation and oedema mouth was typically fast (median onset 2 minutes, 2 minutes and 1 minute after first administration, respectively). Also see Section 4.4 Special Warnings and Precautions for Use.
In the pooled phase II/III adult studies, time to onset from first administration for oral pruritus, throat irritation and oedema mouth was typically fast (median onset 2 minutes, 2 minutes and 1 minute after first administration, respectively). Also see Section 4.4 Special Warnings and Precautions for Use.


 Subjects were randomised to receive Acarizax 12 SQ-HDM, 6 SQ-HDM or placebo once daily for 12 months. Use of nasal steroids (budesonide 64 microgram/dose), oral antihistamines (desloratadine tablets, 5 mg), and antihistamine eye drops (azelastine 0.05%) was permitted as needed.
Subjects were randomised to receive Acarizax 12 SQ-HDM, 6 SQ-HDM or placebo once daily for 12 months. Use of nasal steroids (budesonide 64 microgram/dose), oral antihistamines (desloratadine tablets, 5 mg), and antihistamine eye drops (azelastine 0.05%) was permitted as needed.
 In TO-203-3-2, 278 adolescent subjects were randomised to receive Acarizax 12 SQ-HDM, 6 SQ-HDM and placebo once daily for 12 months. Use of nasal steroids (fluticasone propionate 25-50 microgram/dose), oral antihistamines (loratadine tablet 10 mg) and antihistamine eye drops (olopatadine hydrochloride 0.1%) were permitted as needed.
In TO-203-3-2, 278 adolescent subjects were randomised to receive Acarizax 12 SQ-HDM, 6 SQ-HDM and placebo once daily for 12 months. Use of nasal steroids (fluticasone propionate 25-50 microgram/dose), oral antihistamines (loratadine tablet 10 mg) and antihistamine eye drops (olopatadine hydrochloride 0.1%) were permitted as needed.
