What is in this leaflet
This leaflet answers some common questions about Actair Initiation Treatment and Actair Continuation Treatment sublingual tablets.
It does not contain all the available information.
It does not take the place of talking to your doctor.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking Actair against the expected benefits it will have for you.
If you have any concerns about taking this medicine, ask your doctor.
What is Actair used for
Actair contains a 50% mixture of allergen extracts from 2 species of house dust mite. The treatment with Actair is intended to increase the immunological tolerance towards house dust mites, and thereby reducing the allergic symptoms.
Actair is used for the treatment of house dust mite allergies that are characterised by rhinitis (sneezing, runny or itchy nose, nasal congestion) with or without conjunctivitis (itchy and watery eyes) in adults and adolescents over 12 years.
Before treatment, the allergy is confirmed by a doctor with adequate training and experience in allergic diseases, who will perform the appropriate skin and/or blood tests.
The house dust mites that Actair is used for are: European house dust mite (Dermatophagoides pteronyssinus) and American house dust mite (Dermatophagoides farinae).
How it works:
Treatment with Actair is intended to increase the immunological tolerance towards house dust mites, thereby building immunity to the specific allergens in house dust mites, thus helping to reduce the allergic symptoms.
Ask your doctor if you have any questions why Actair has been prescribed for you.
Actair is not addictive.
This medicine is available only with a doctor’s prescription.
Before you are given Actair
Do not take Actair:
- if you are hypersensitive (allergic) to any of the other ingredients of Actair;
- if you suffer from severe and/or unstable asthma;
- if your immune system is very weakened or if you suffer from a disease that attacks your own immune system;
- if you suffer from a malignant disease (for example cancer);
- if you have any inflammation in your mouth (for example mouth ulcers, bleeding gums).
If you are not sure if you should be taking Actair, talk to your doctor.
Before you start your treatment with Actair:
Take special care with Actair:
If you have to undergo surgery in the mouth or if you are having a tooth pulled, you should stop the treatment with Actair for 7 days to allow your oral cavity to heal. Thereafter, restart the treatment with the previous dosage. If you stopped the treatment for more than 7 days, please ask your doctor how you should restart the treatment.
Talk to your doctor if:
- you have any history of allergic inflammation of the oesophagus (eosinophilic oesophagitis). During treatment with Actair, if you have severe or persistent upper abdominal pain, swallowing difficulties or chest pain, please contact your doctor who may reconsider your treatment.
- you are taking a beta blocker (i.e., a class of drugs often prescribed for heart conditions and high blood pressure but also present in some eye drops and ointments), as this drug may decrease the effectiveness of adrenaline, a medicine used to treat serious allergic reactions.
Taking other medicines
Tell your doctor if you are taking or have recently taken any other medicines, including medicines obtained without a prescription.
Pregnancy and breast-feeding
Ask your doctor for advice before taking any medicine.
At present there is no experience for the use of Actair during pregnancy. Therefore, you should not start immunotherapy if you are pregnant. If you become pregnant, speak to your doctor about whether it is appropriate for you to continue treatment.
There is no experience for the use of Actair during breast-feeding as well. No effects on infants who are breast-fed during treatment are anticipated. However, you should not start immunotherapy if you are breast-feeding. If you wish to breastfeed while on treatment, speak to your doctor about whether it is appropriate for you to continue treatment.
Driving and using machines
No effect on the capacity to drive or use machines has been observed with Actair.
Important information about some of the ingredients of Actair
This medicine contains lactose. If you have been told by your doctor that you have intolerance to some sugars, contact your doctor before taking this medicinal product.
Your doctor will advise you.
Your doctor has more information on medicines to be careful with or avoid while taking Actair.
If necessary, your doctor may at the same time prescribe medications to treat the possible allergic reactions.
How Actair is given
Always take Actair exactly as your doctor has told you. Check with your doctor if you are not sure.
Actair is prescribed by doctors with adequate training and experience in treatment of allergy. With prescriptions for adolescents over 12 years, the doctor will have the relevant experience in the treatment of patients in this age group.
You are advised to take the first tablet under medical supervision and be monitored for 30 minutes. This gives you the possibility of discussing possible side effects with your doctor.
Dosage
Adults and adolescents over 12 years
The therapy is composed of an initiation treatment (including a 3-day dose escalation) and a continuation treatment.
Initiation Treatment:
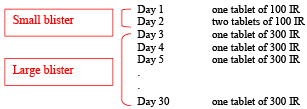
The pack corresponding to the initiation treatment contains two or four blisters:
- One small blister with 3 tablets of 100 IR
- One large blister with 28 tablets of 300 IR or three large blisters with respectively 28, 30 and 30 tablets of 300 IR
Method of administration
Take one 100 IR tablet on day one. Keep the tablet under your tongue until it is completely disintegrated before you swallow. On the second day of treatment, two 100 IR tablets must be placed under the tongue simultaneously and then swallowed.
It is recommended that the tablets be taken during the day. You should have no food or drink in your mouth at the same time you are taking the medicine.
Use the following dosing scheme:
Always start with the small blister:
Continuation Treatment:
The pack corresponding to the continuation treatment contains large blister strip with 30 or 90 tablets of 300IR sublingual tablets
The treatment must be continued with 1 tablet of 300 IR once a day from the Actair continuation treatment which contains 300 IR sublingual tablets only.
Do not halve the tablet. Tablet is to be taken whole.
Duration of treatment
Start your Actair treatment on the day determined by your doctor.
Take these tablets as prescribed by your doctor until the end of the treatment course.
After one year of treatment, efficacy has been shown to be maintained in the following year without treatment.
There is no experience with Actair in children younger than 5 years and in patients over 65 years of age.
How long should Actair be taken for?
Actair is started on the day determined by your doctor. It is recommended to maintain the treatment all year round.
Each year your doctor will assess if it is suitable for you to continue Actair treatment.
If you take more Actair than you should
Higher doses may be associated with reduced tolerability and may potentially increase the risk of serious allergic reactions.
In the event of overdose you may need medical treatment.
Contact the Poisons Information Centre on (13 11 26 in Australia and 0800 POISON (0800 764 766) in New Zealand) for advice on the management of overdose.
If you forget to take Actair
Do not take a double dose to make up for forgotten dose.
If you interrupted the treatment with Actair for less than one week, you can take up treatment where you left off.
If you stop taking Actair
If you stop taking Actair, you may not have an effect of the treatment.
If you interrupt treatment for longer than 7 days, you should ask your doctor before restarting treatment
If you have any further questions on the use of this product, ask your doctor.
While you are receiving Actair
Things you must do
Tell all doctors, dentists and pharmacists who are treating you, that you are receiving Actair.
Tell your doctor if you feel the treatment is not helping your condition.
If you experience any infection or inflammation in the mouth or if you experience an increase in asthma or allergy symptoms, ask your doctor for advice before continuing treatment with Actair.
Side effects
Actair helps people with allergies to the house dust mites allergen extracts of the product but they may have unwanted side effects in a few people.
All medicines can have side effects. Sometimes they may be serious, most of the time they are not. You may need medical treatment if you get some of these side effects.
Contact your doctor or nurse immediately if symptoms of an allergic reaction occur.
During treatment with Actair, you will be exposed to substances that may cause application site reactions and/or symptoms, which affect the whole body. You may experience application site reactions (such as itching of the mouth and throat irritation).
Stop taking ACTAIR and contact your doctor immediately if you develop or notice:
Allergic symptoms that affect the whole body (i.e. rapid onset of an illness associated with involvement of the skin and/or mucosa, breathing difficulty, abdominal pain or symptoms related to a drop in blood pressure).
Other possible side effects include the following:
Very common (affects more than 1 in 10 people)
- Throat irritation, itchy mouth, swelling mouth, itchy ears
Common (affects less than 1 in 10 people):
- Inflammation in the eyes, itchy eyes, watery eyes
- Swelling tongue, itchy tongue, inflammation of the tongue
- Swelling or itching or blistering of lips,
- Formation of blisters in the mouth, uncomfortable feeling in the mouth or throat, tingling or numbness in the mouth, Decreased sensitivity in the mouth, Inflammation in the mouth with or without ulcers, pain in the mouth,
- Pain in the throat, swelling in the throat, sensation of dryness in throat.
- Difficulty in swallowing
- Ear pain
- Sinus congestion
- Cough
- Breathing difficulty, chest discomfort, asthma, bronchospasm
- Nausea, abdominal pain, diarrhoea
- Itching, dry skin, abnormal skin sensations
Uncommon (affects less than 1 in 100 people):
- Eyelid spasm
- Rhinitis (stuffy nose, runny nose, sneezing, , nasal discomfort ),
- Dry mouth
- Inflammation of the nose and throat, Throat tightness
- Gum swelling, gum pain, swelling of palate
- Salivary gland enlargement, increased production of saliva
- Wheezing
- Chest pain,
- Serious allergic reaction with swelling of the face and throat (angioedema)
- Headache
- Tiredness, malaise
- Bad breath, heartburn, vomiting, bowel disorders
- Increased heartbeat rhythm
- Anxiety, Sleepiness
- Muscle spasms
- Rash, urticaria, blisters
- Abnormalities in morphology or count of blood cells or in biological parameters
Side effects in children and adolescents
In children and adolescents, malaise was reported at a higher incidence than in adults. In patients of this age group, oral disorder, bronchitis, enterocolitis, candida infection, seborrhoea and ear disorder were also reported as uncommon (affects less than 1 in 100 people).
Additional side effects experienced with similar products
Cases of oesophageal inflammation have been reported with other sublingual immunotherapy products.
These do not represent all possible adverse drug reactions. For a full list of all reported adverse drug reactions refer to the Australian Product Information, New Zealand Data Sheet or Doctor.
After using Actair
Storage
- Keep it where children cannot reach it
- Keep it in the original pack in order to protect from moisture
- Keep it below 30ºC.
- Do not use if past the expiry date which is stamped on the carton side panel
Product Description
Actair Initiation Treatment sublingual tablets 100 IR and 300 IR contain a small blister with 3 tablets of 100 IR concentration and a large blister with 28 tablets of 300 IR concentration
OR
Actair Initiation Treatment sublingual tablets 100 IR and 300 IR contain a small blister with 3 tablets of 100 IR concentration and a three large blisters with respectively 28, 30 and 30 tablets of 300 IR concentration
OR
Actair Initiation Treatment sublingual tablets 100 IR and 300 IR contain a small blister with 3 tablets of 100 IR concentration and a large blister with 7 tablets of 300 IR concentration
Actair Continuation Treatment sublingual tablets 300 IR contain blisters with 30 tablets of 300 IR concentration. The continuation treatment is available in packs of 30 or 90 tablets.
The tablets are round, biconvex, white to beige, brown speckled. The 100 IR tablets are engraved “SAC” on one side and “100” on the other and the 300 IR tablets are engraved SAC” on one side and “300” on the other.
Presentation
The following pack sizes are available:
Actair Initiation Treatment sublingual tablets 100 IR and 300 IR (AUST R 233470)
- Pack with 3 tablets 100 IR and 28 tablets 300 IR
- Pack with 3 tablets 100 IR and 88 tablets 300 IR
- Pack with 3 tablets 100 IR and 7 tablets 300 IR
Actair Continuation Treatment sublingual tablets 300 IR (AUST R 233471)
- Pack with 30 tablets
- Pack with 90 tablets
NAME AND ADDRESS OF THE SPONSOR IN AUSTRALIA:
Stallergenes Australia Pty Ltd
Suite 2408,
4 Daydream St.,
Warriewood, NSW 2102
Ph: 1800 824 166
Fax: 1800 824 199
NAME AND ADDRESS OF THE SPONSOR IN NEW ZEALAND:
Stallergenes Greer New Zealand Limited
Level 1, 24 Manukau Road,
Epsom, Auckland 1023
New Zealand
Ph: 0800 824 166
Date Prepared: 30 May 2019
Published by MIMS August 2019



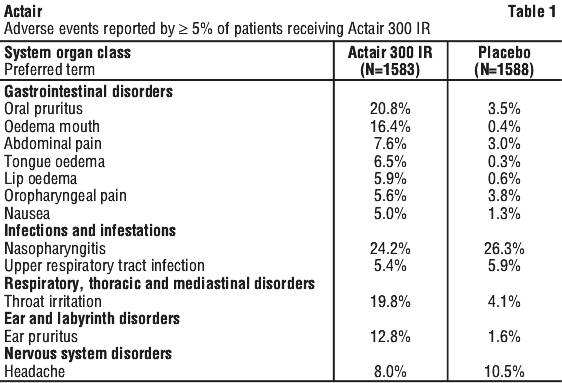 Adverse events with an incidence < 5% and with causality assigned to Actair are listed below according to the MedDRA convention by system organ class and by frequency common: ≥ 1/100, < 1/10, uncommon: ≥ 1/1,000, < 1/100, rare: ≥ 1/10,000, < 1/1,000.
Adverse events with an incidence < 5% and with causality assigned to Actair are listed below according to the MedDRA convention by system organ class and by frequency common: ≥ 1/100, < 1/10, uncommon: ≥ 1/1,000, < 1/100, rare: ≥ 1/10,000, < 1/1,000.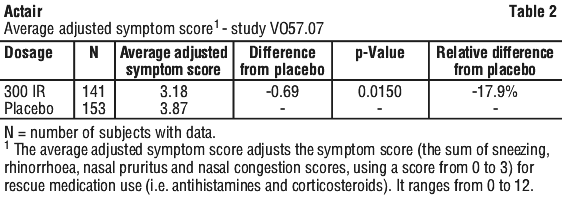 Patients receiving the 300 IR dose had a lower average rhinitis total symptom score by 18.5% than those receiving placebo. There was no difference in the average rescue medication score between the active and placebo treatment groups.
Patients receiving the 300 IR dose had a lower average rhinitis total symptom score by 18.5% than those receiving placebo. There was no difference in the average rescue medication score between the active and placebo treatment groups.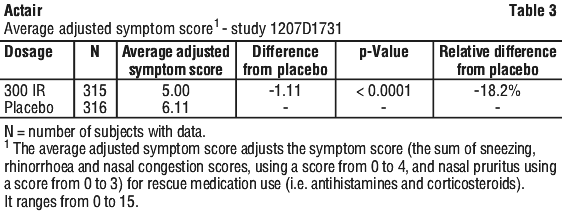 In the subset of adolescents (aged 12 to 17 years), patients receiving the 300 IR dose had a lower average adjusted symptom score by 26.9% than those receiving placebo.
In the subset of adolescents (aged 12 to 17 years), patients receiving the 300 IR dose had a lower average adjusted symptom score by 26.9% than those receiving placebo.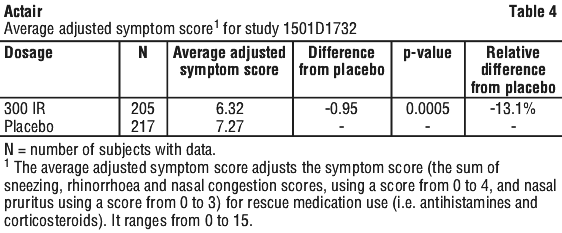 Patients receiving the 300 IR dose had a lower Average Rhinitis Total Symptom Score by 12.7% and a lower Average Rescue Medication Score by 8.5% than those receiving placebo.
Patients receiving the 300 IR dose had a lower Average Rhinitis Total Symptom Score by 12.7% and a lower Average Rescue Medication Score by 8.5% than those receiving placebo.