What is in this leaflet
This leaflet answers some common questions about Addaven. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking Addaven against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet. You may need to read it again.
What Addaven is used for
Addaven is a medicine that contains trace elements. Trace elements are tiny amounts of chemicals that your body needs to function normally. Addaven is given intravenously (as a drip into a vein) when you cannot eat normally. This medicine is usually used as part of a balanced intravenous diet, together with proteins, fat, carbohydrates, salts and vitamins.
Before you are given Addaven
When you must not take it
- if you are allergic to any of the ingredients of this medicine. If you develop a rash or other allergic reactions (like itching, swollen lips or face, or shortness of breath), inform your doctor immediately.
- if your bile excretion is blocked.
- if you have Wilson’s disease (a genetic disorder in which copper builds up in the body) or hemochromatosis (accumulation of iron in the body).
Addaven must not be given to children under 12 years of age.
Before you start to take it
Talk to your doctor if you have problems with the way your liver and/or kidney work.
Your doctor may want to do regular blood tests to check your condition. If you are taking iron orally in parallel with the infusion your doctor will check that iron is not accumulating in your body.
Iron and iodine may cause allergic reactions on rare occasions when given as a drip. Tell your doctor or nurse if you get any allergic reaction when receiving Addaven.
Pregnancy and breast-feeding
If you are pregnant or breast-feeding, think you may be pregnant or are planning to have a baby, ask your doctor for advice before using this medicine.
Taking other medicines
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines.
How to take Addaven
How much to take
Your doctor will decide on the correct dose for you to receive.
The recommended dose for adults is 10 millilitres (mL) each day. If you have problems with your liver or kidneys you may receive a lower dose.
How to take it
This medicine will be administered to you by a healthcare professional. You will receive your medicine by infusion (drip) into a vein.
Addaven should be mixed with another solution before it is given to you. Your doctor or nurse will make sure it is prepared correctly.
If you take too much (overdose)
It is very unlikely that you will receive more medicine than you should as your doctor or nurse will monitor you during the treatment.
However if you think that you have received too much Addaven, inform your doctor or nurse immediately.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
While you are using Addaven
Driving and using machines
Addaven has no effect on driving or using machines.
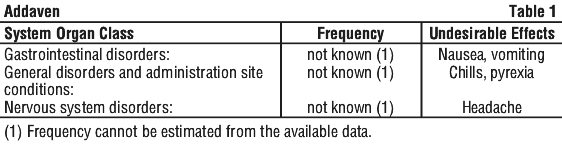
Adverse Effects
No known undesirable effects have been reported with the use of Addaven according to recommendation. If you get any adverse effects, talk to your doctor or nurse.
Storage
Storage
Store below 30°C.
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the carton label and embossed on the ampoule. The expiry date refers to the last day of that month.
Your doctor and hospital pharmacist are responsible for the correct storage, use and disposal of Addaven.
After dilution
The addition of Addaven should be performed immediately before the start of the infusion and should be used within 24 hours.
Disposal
Any unused medicinal product or waste material should be disposed of in accordance with local requirements.
Product Description
What it looks like
Addaven is a clear and colourless to slightly yellow solution of trace elements.
Addaven is available in a polypropylene ampoule containing 10 mL of concentrated solution. Each carton contains 20 ampoules of 10 mL.
Ingredients
The active substances in one ampoule (10 mL) are:
- Chromic chloride
53.3 µg - Cupric chloride dihydrate
1.02 mg - Ferric chloride hexahydrate
5.40 mg - Manganese chloride
198 µg - Potassium iodide
166 µg - Sodium fluoride
2.10 mg - Sodium molybdate dihydrate
48.5 µg - Sodium selenite
173 µg - Zinc chloride
10.5 mg
The other ingredients are: xylitol, hydrochloric acid and water for injections.
This product contains 0.052 mmol of sodium (1.2 mg) per 10 mL dose.
Supplier
Addaven is supplied in Australia by:
Fresenius Kabi Australia Pty Limited
Level 2, 2 Woodland Way
Mount Kuring-gai NSW 2080
Telephone: 1300 361 004
Addaven is supplied in New Zealand by:
Fresenius Kabi New Zealand Limited
60 Pavilion Drive
Airport Oaks, Auckland 2022
New Zealand
Freecall: 0800 144 892
Australian Registration Number
AUST R 244493
® = Registered Trademark
This leaflet was prepared on 29th July 2016.
Published by MIMS November 2016