What is in this leaflet?
This leaflet answers some common questions about ADRENALINE MYLAN Auto-Injector. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor or pharmacist has weighed the risks of you receiving ADRENALINE MYLAN against the benefits this medicine is expected to have for you.
If you have any concerns about receiving this medicine, talk to your doctor or pharmacist before there is a situation where ADRENALINE MYLAN is needed.
Please read this leaflet carefully before an emergency arises.
Keep this leaflet safely, with the ADRENALINE MYLAN Auto-Injector. You will probably want to read it again.
What ADRENALINE MYLAN is used for
ADRENALINE MYLAN is only to be used for the immediate EMERGENCY TREATMENT of a SEVERE ALLERGIC REACTION (also known as ANAPHYLAXIS).
Your doctor or pharmacist will explain what this is like, so that you know when you must use ADRENALINE MYLAN.
ADRENALINE MYLAN is used to treat people who have a severe allergic reaction caused by, for example, hypersensitivity to
- food
- medicines
- stinging insects
- insect bites
- latex or
- other allergens, possibly unidentified.
See Side effects for information regarding allergy to sodium metabisulfite.
Avoidance of known allergens is most important in preventing severe allergic reactions. A severe allergic reaction, otherwise known as anaphylaxis, is overwhelming and life-threatening.
It affects the whole body, in particular the
- heart and blood circulation.
This may cause, for example, low blood pressure and collapse, or abnormal heart beat or function. - air passages and lungs.
This causes difficulty in breathing, with noisy wheezing or gasping, possibly with sneezing and a very runny nose. - stomach and bowels.
This may cause nausea, vomiting, diarrhoea and abdominal cramps, often severe. - skin (very common).
This causes flushing, itching, skin rash, hives or swelling, which may be widespread or in one part of the body. - brain.
There may be fits or confusion due to lack of oxygen.
Usually several parts of the body are affected. The patient feels extremely unwell.
It is important to act quickly to treat a severe allergic reaction.
The adrenaline in ADRENALINE MYLAN treats allergic emergencies. It
- shrinks abnormally wide blood vessels and
- makes the heart beat strongly.
This helps improve the very low blood pressure and poor circulation that occur in a severe allergic reaction.
Adrenaline also
- relaxes the lungs. This eases breathing and lessens wheezing.
- helps stop
- swelling, for example, of the face and lips
- skin rash and/or
- itching.
The ADRENALINE MYLAN Auto-Injector is intended for use in people who have been assessed and advised by their doctor or pharmacist as needing an ADRENALINE MYLAN Auto-Injector to treat their severe allergic reaction and who
- weigh more than 30 kg, and
- are having a severe allergic reaction.
Note: Selection of the appropriate dosage is determined according to patient body weight.
For a person who weighs from 15 kg up to 30 kg, the doctor or pharmacist may assess their severe allergic reaction as needing the larger dosage of adrenaline as provided by an ADRENALINE MYLAN Auto-Injector. (The ADRENALINE JR MYLAN Auto-Injector delivers a smaller amount of adrenaline).
If you have any questions about this, talk to your doctor or pharmacist.
ADRENALINE MYLAN
- provides convenient first-aid
- may be self-administered or administered by a carer
- has a spring-activated, hidden needle
- automatically injects one pre-measured dose of adrenaline
- is for single use only.
ADRENALINE MYLAN may be prescribed by your doctor. You can also purchase it directly from a pharmacy.
Before you use ADRENALINE MYLAN
The ADRENALINE MYLAN Auto-Injector is simple to use. Please read and follow the step-by-step instructions.
Do not remove the blue safety release until you need to use ADRENALINE MYLAN and are ready to use it.
The safety release stops ADRENALINE MYLAN from "firing" or working.
There is no visible needle. This helps overcome fear of injection. The hidden needle is very fine so that the injection does not usually hurt.
When you must not use it
ADRENALINE MYLAN is a life-saving treatment in the emergency management of severe allergic reactions. Therefore, it should not be withheld from anyone who is considered to need it.
The ADRENALINE MYLAN Auto-Injector has a clear "viewing window" so you can see the contents.
Do not use ADRENALINE MYLAN if the contents
- are cloudy or
- are coloured or
- contain sediment or
- if the "viewing window" is obscured.
These changes indicate that the effectiveness of the drug product may be decreased.
When the "viewing window" is obscured, the auto-injector has already "fired" and cannot be used again.
Check your ADRENALINE MYLAN regularly (see Things you must do).
Do not use ADRENALINE MYLAN
- after the expiry date on the ADRENALINE MYLAN Auto-Injector and carton or
- if the packaging is torn, doesn't look quite right or shows signs of tampering.
If the ADRENALINE MYLAN packaging is damaged or it is past the expiry date, return it to your pharmacist for disposal.
Help in using ADRENALINE MYLAN
It is important to know how to use an ADRENALINE MYLAN Auto-Injector BEFORE a severe allergic reaction causes a medical emergency.
Ask your doctor or pharmacist to show you how to use an ADRENALINE MYLAN Auto-Injector.
It is important to regularly ask your doctor or pharmacist how to use an ADRENALINE MYLAN Auto-Injector.
ADRENALINE MYLAN is effective as emergency treatment of a severe allergic reaction ONLY when used according to the directions in this leaflet.
Give WITH CAUTION to those who have
- high blood pressure
- thyroid problems
- high pressure in the eye (narrow-angle glaucoma).
Take care also when giving ADRENALINE MYLAN to those
- having an anaesthetic
or who have:
- diabetes
- heart disease
- allergy to ADRENALINE MYLAN
- brain damage.
You should tell your doctor or pharmacist if you have:
- depression, or are on medication for depression
- thyroid problems, or are on thyroid medication
- heart disease or high blood pressure
- diabetes
- high pressure in the eye (narrow angle glaucoma)
- asthma
- are pregnant or planning to become pregnant
- ever had an allergic reaction to sodium metabisulfite.
Taking other medicines
Tell your doctor or pharmacist if you are taking other medicines, including any medicines that you buy without a prescription from your pharmacy, supermarket or health food shop. Some medicines may interfere with ADRENALINE MYLAN including:
- medicines for high blood pressure, especially those known as "beta-blockers", and other medicines for heart problems
- medicines for depression, especially "tricyclic antidepressants"
- thyroid hormone
- medicines for diabetes
- antihistamines.
These medicines may affect how well the adrenaline in ADRENALINE MYLAN works. Your doctor or pharmacist can tell you what to do if you are taking any of these medicines.
You should discuss any concerns you have with your doctor or pharmacist.
How to use ADRENALINE MYLAN
It is important to know how to use your ADRENALINE MYLAN in an emergency (see Help in using ADRENALINE MYLAN).
Follow carefully all directions given to you by your doctor or pharmacist. They may differ from the information contained in this leaflet.
If you do not understand the instructions in this leaflet or on the ADRENALINE MYLAN Auto-Injector, ask your doctor or pharmacist for help.
When to use it
Use ADRENALINE MYLAN immediately if you have signs and symptoms of a severe allergic reaction, as described by your doctor or pharmacist. Symptoms do vary. However, the symptoms are likely to include:
- collapse
- difficulty in breathing
- wheezing
- swelling, especially of the face, lips, tongue or throat
- flushing of the face
- irregular or faint pulse
- vomiting, diarrhoea, stomach cramps
- skin rash, hives
- itching.
How much to use
- ADRENALINE MYLAN Auto-Injector delivers one measured dose of adrenaline. This dose is intended for a person who weighs more than 30 kg and has been assessed by their doctor or pharmacist as needing ADRENALINE MYLAN.
- Your doctor or pharmacist may also advise ADRENALINE MYLAN Auto-Injector for a child who weighs from 15 kg up to 30 kg.
NOTE: Unless otherwise advised by the doctor or pharmacist, ADRENALINE JR MYLAN Auto-Injector should be used for children who weigh from 15 kg up to 30 kg.
See also "What ADRENALINE MYLAN is used for" above.
How to use it
General
- Grip only the mid-section of the ADRENALINE MYLAN Auto-Injector. Avoid touching either end, except when you remove the blue safety release. The blue safety release stops the ADRENALINE MYLAN from "firing" or working.
- Never place thumb, fingers or hand over the orange end as this contains the needle.
- Always point the orange end (which holds the needle) away from any part of any body, except the planned injection site. This is usually the thigh of the person who has the severe allergic reaction.
- Always hold the leg firmly during injection especially in people who are uncooperative and kick or move during an injection. This will reduce the risk of injection related injuries.
- Keep ADRENALINE MYLAN away from the face and eyes.
- Never inject ADRENALINE MYLAN into the buttock.
Carefully follow the step-by-step directions to make the ADRENALINE MYLAN Auto-Injector work properly.
Step 1
Flip open the yellow cap on the protective carry tube. Carefully tip and slide the ADRENALINE MYLAN Auto-Injector out of the carry tube. Never place thumb, fingers or hand over the orange endas this contains the needle. Check the contents of the ADRENALINE MYLAN Auto-Injector through the"viewing window". Make sure the solution is clear, colourless and sediment-free. If the "viewing window" is obscured, the ADRENALINE MYLAN Auto-Injector has already "fired" and cannot be used again.
Step 2
Make a fist around the ADRENALINE MYLAN Auto-Injector, with your thumb nearest to the blue safety release (see FIGURE 1).

Step 3
Activate the ADRENALINE MYLAN Auto-Injector by removing the blue safety release with your other hand (also shown in FIGURE 1). This "triggers" the ADRENALINE MYLAN Auto-Injector. It is now ready to
- "fire" the needle and
- inject the medicine through the needle into the thigh muscle.
After you remove the blue safety release do not touch or place thumb, fingers or hand over the orange end as this contains the needle.
Step 4
Hold the ADRENALINE MYLAN Auto-Injector at a 90° angle (right angle) to the outer part of the thigh. Hold the orange end about 5cm away from the thigh (see FIGURE 2).

Step 5
If you are administering the ADRENALINE MYLAN Auto-Injector to another person, hold their leg firmly, especially if they are uncooperative.
Push the orange end FIRMLY into the outer mid-thigh until a "click" is heard or felt. Keep pressing the ADRENALINE MYLAN Auto-Injector firmly against the thigh for approximately 3 seconds (see FIGURE 3).
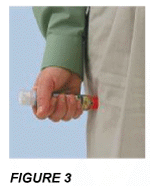
NOTE: ADRENALINE MYLAN may be used either
- through clothing, as shown in FIGURE 3, or
- directly on to skin.
When you press the orange end of the ADRENALINE MYLAN Auto-Injector firmly against the thigh, the hidden needle "fires". It injects the measured dose of adrenaline into the thigh muscle.
Step 6
Carefully take the ADRENALINE MYLAN Auto-Injector away from the thigh.
The orange needle cover will extend to cover the needle (see FIGURE 4).

Check the "viewing window" – it should now be obscured. If it isn't, the ADRENALINE MYLAN Auto-Injector has not "fired" or worked. If the "viewing window" is not obscured, repeat steps 2-6 above.
Step 7
Record the time ADRENALINE MYLAN was given.
Step 8
Seek further medical attention for yourself immediately. Although you have received adrenaline from ADRENALINE MYLAN, you may need more medical treatment.
Either
- Contact your doctor or
- Call an ambulance (telephone 000 in Australia). State that adrenaline treatment may be needed.
Step 9
With severe, persistent allergic reactions, repeat injections with an additional ADRENALINE MYLAN may be necessary. The ADRENALINE MYLAN dose may be repeated every 5 to 15 minutes if symptoms recur or have not subsided.
Do not inject more than two sequential doses of ADRENALINE MYLAN unless under medical supervision.
Step 10
Explain to the doctor that you have had ADRENALINE MYLAN (intramuscular adrenaline).
Take your used ADRENALINE MYLAN Auto-Injector with you.
To avoid injury, follow the used ADRENALINE MYLAN disposal instructions below (see Disposal).
Additional information about treatment of insect sting allergy
Use ADRENALINE MYLAN immediately if you have been stung or bitten by the insect and experience the signs and symptoms as described by your doctor or pharmacist.
If the insect's sting is easily accessible, remove the sting with your fingernails. Do not squeeze, pinch or push the sting deeper into the skin. If available, ice packs or sodium bicarbonate soaks may then be applied to the stung area.
Keep warm, calm, and as still as possible.
If you use too much(overdose)
Because each ADRENALINE MYLAN Auto-Injector contains only one dose, overdosage is unlikely. Overdosage may lead to a rise in blood pressure. This may cause bleeding in the brain or fluid in the lung.
If you think you or anyone else may have used too much of this medicine,
- consult your doctor immediately or
- telephone the Poisons Information Centre (telephone 13 11 26 in Australia) for advice, or
- go to Accident and Emergency at your nearest hospital. Do this even if there are no signs of discomfort or poisoning. Urgent medical attention may be required.
Things to note about ADRENALINE MYLAN
Things you must do
Check what the expiry date is on your ADRENALINE MYLAN Auto-Injector.
Mark this expiry date on your calendar or diary.
Replace your ADRENALINE MYLAN Auto-Injector prior to this expiry date.
Also note on your calendar or diary to check the contents of your ADRENALINE MYLAN Auto-Injector each month. Do this through the "viewing window".
Make sure the solution is clear, colourless and sediment-free. If not, obtain a new ADRENALINE MYLAN Auto-Injector immediately.
If the "viewing window" is obscured this means the ADRENALINE MYLAN Auto-Injector has already "fired" or worked. It cannot be used again. Return it to your doctor or pharmacist for disposal. Obtain a new ADRENALINE MYLAN Auto-Injector immediately.
The ADRENALINE MYLAN Auto-Injector can be damaged and made unusable if it is mishandled.
ADRENALINE MYLAN is NOT a toy. Keep it out of reach of children.
Things you must not do
Do not use ADRENALINE MYLAN
- to treat any other medical conditions including other types of shock unless your doctor tells you to.
- if the solution is brown or contains any particles or cloudiness.
- in a child who weighs 15 kg to 30 kg unless advised by your doctor or pharmacist. ADRENALINE JR MYLAN is generally recommended for children who weigh 15 kg to 30 kg. (See also "What ADRENALINE MYLAN is used for" above).
- in a child who weighs less than 15 kg. Talk to your doctor or pharmacist if this is the case so that a careful assessment can be made and the appropriate dose selected based on the child's weight and the life-threatening nature of the reaction(s) for which ADRENALINE MYLAN is being prescribed.
Do not take off the blue safety release until you need to use ADRENALINE MYLAN and are ready to use it.
Remember that the ADRENALINE MYLAN Auto-Injector is activated ("triggered", ready to "fire") as soon as you take off the blue safety release.
Do not touch the orange end on any person or object while you are taking off the blue safety release and/or after you have taken off the blue safety release.
The orange end holds the hidden needle, ready to "fire" and inject the adrenaline.
After you take off the blue safety release
- Do not "click" on either end of the ADRENALINE MYLAN Auto-Injector. The ADRENALINE MYLAN Auto-Injector does not work like a retractable ball-point pen (see How to use it step 3).
- Do not bump the ADRENALINE MYLAN Auto-Injector until you intentionally push the orange end firmly into the thigh (see How to use it step 4).
- This will help prevent accidental activation of the ADRENALINE MYLAN Auto-Injector prior to use.
Do not inject into hands, feet, ears, nose, buttocks, genital area or into a blood vessel. If this does happen accidentally you must immediately seek medical attention.
Do not inject into the buttock. Rare cases of serious skin infections have been reported following injection into the buttock. If you develop signs or symptoms of infection, such as persistent redness, warmth, swelling, or tenderness, at the adrenaline injection site you must seek medical attention immediately.
Side effects
Like all medicines, ADRENALINE MYLAN can cause some side effects. Side effects not listed in this leaflet may occur in some patients.
Tell your doctor or pharmacist if you experience any unpleasant side effects after using ADRENALINE MYLAN, even if you do not think the effect is connected with the medicine or is listed in this leaflet. Ask your doctor or pharmacist to answer any questions you may have.
Do not be alarmed by the following list of side effects. You may not experience any of them.
Adrenaline
Tell your doctor or pharmacist if you have any of the following:
- fast or noticeable heart beat
- difficulty breathing
- chest pain
- shortness of breath
- dizziness
- pale skin colour
- weakness
- tremor
- headache
- throbbing
- sweating
- nausea or vomiting
- sleeplessness
- coldness
- hallucinations
- restlessness
- anxiety
- fear
- flushing or redness of face and skin.
These side effects are minor and short lasting.
Accidental injection into the hands, fingers or feet may result in the following:
- fast or noticeable heart beat
- pale skin colour at the site of accidental injection
- feeling of coldness at the site of accidental injection
- bruising or discolouration at the site of accidental injection
- bleeding
If an accidental injection occurs at one of these areas, seek medical attention immediately.
Injection related injuries, such as needle cuts and bent needles, have been reported in people who are uncooperative and kick or move during the injection.
Rare cases of serious skin infections have been reported following injection into the buttock.
More severe side effects can occur occasionally. They are caused by adrenaline stimulating the heart and increasing the blood pressure. Rarely, these side effects can cause, for example, a stroke, lung problems or severe irregular heartbeat.
Adrenaline may also cause disorientation and impaired memory.
Sodium metabisulfite
ADRENALINE MYLAN contains a very small quantity of sodium metabisulfite as a preservative. In people who are allergic to sodium metabisulfite, this could cause, for example, severe allergic reactions (hypersensitivity) or breathing difficulty (bronchospasm) in certain groups of people, especially those with history of asthma. However, the active ingredient, adrenaline, in ADRENALINE MYLAN is expected to overcome any allergic reaction to sodium metabisulfite. In an emergency, the risk of exposure to the very small amount of sodium metabisulfite is generally outweighed by the benefits of ADRENALINE MYLAN.
Storing ADRENALINE MYLAN before use
Always handle the ADRENALINE MYLAN Auto-Injector carefully, so as not to accidentally activate it or make it "fire" or work.
Storage
Keep ADRENALINE MYLAN available with the person for whom it is intended.
Keep ADRENALINE MYLAN in a cool dry place below 25°C. Temperature excursions between 15°C to 25°C permitted.
Do not place ADRENALINE MYLAN in the fridge. Do not leave ADRENALINE MYLAN in your car. Extreme temperatures may cause the auto-injector unit to malfunction.
Protect ADRENALINE MYLAN from light. Keep ADRENALINE MYLAN in the protective carry tube until required.
Do not store ADRENALINE MYLAN or any other medicine in the bathroom or near a sink. Do not leave it on a windowsill or in the car. Heat and dampness can destroy some medicines.
Keep ADRENALINE MYLAN where children cannot accidentally reach it.
After using ADRENALINE MYLAN
Disposal
After the ADRENALINE MYLAN Auto-Injector has "fired" or worked, the orange needle cover will extend to cover the exposed needle. After use, continue to handle the ADRENALINE MYLAN Auto-Injector safely and with care. Do this even if you think the ADRENALINE MYLAN Auto-Injector has not "triggered", "fired" or worked properly.
You CANNOT RE-USE the ADRENALINE MYLAN Auto-Injector even though some adrenaline is left inside it.
After use, do not just throw away the ADRENALINE MYLAN Auto-Injector.
- Do not tamper with the orange needle cover.
- Do not replace the blue safety release.
- The used auto-injector with extended needle cover will not fit back into the carry tube.
- Give your used ADRENALINE MYLAN Auto-Injector, to the doctor's office or to a hospital. This is for inspection, if required, and also for safe disposal. The used ADRENALINE MYLAN Auto-Injector, should be placed in a rigid sharps disposal unit.
Product description
Ingredients
ADRENALINE MYLAN delivers one x 0.3 mL dose of adrenaline (epinephrine). This ADRENALINE MYLAN dose provides 300 mcg of adrenaline (epinephrine).
The ADRENALINE MYLAN Auto-Injector holds 2 mL of adrenaline (epinephrine) 1:1,000 solution.
The injection also contains small amounts of
- sodium chloride
- sodium metabisulfite
- hydrochloric acid.
The ADRENALINE MYLAN Auto-Injector is packed in a non-waterproof protective plastic carry tube. This carry tube has a yellow cap. The carry tube is supplied in a cardboard pack.
The Australian Registration Number is AUST R 296842.
This is not all the information available on ADRENALINE MYLAN. If you have any more questions or are unsure about anything, ask your doctor or pharmacist.
Supplier
ADRENALINE MYLAN is distributed in Australia by:
Alphapharm Pty Ltd
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point NSW 2000
www.mylan.com.au
Medical Information
Phone: 1800 274 276
This leaflet was prepared in July 2021.
Made in USA
ADRENALINE MYLAN_CMI/Jul21\00
Published by MIMS March 2022

 Molecular formula: C9H13NO3. Molecular weight: 183.2.
Molecular formula: C9H13NO3. Molecular weight: 183.2.