1 Name of Medicine
Trifarotene.
2 Qualitative and Quantitative Composition
One gram of cream contains 50 microgram of trifarotene.
Excipients with known effect.
Contains alcohol as 6.335% v/v ethanol. For the full list of excipients see Section 6.1 List of Excipients.3 Pharmaceutical Form
Cream.
White and homogenous cream.
4.1 Therapeutic Indications
Aklief is indicated for the topical treatment of Acne Vulgaris of the face and/or the trunk in patients from 12 years of age and older, when many comedones, papules and/or pustules are present.
4.2 Dose and Method of Administration
Dosage.
Apply a thin layer of Aklief cream to the affected areas of the face and/or trunk once a day, in the evening, on clean and dry skin.
It is recommended that the physician assesses the continued improvement of the patient after three months of treatment. The duration of treatment should be determined by the doctor based on the clinical response.
Special populations. Elderly patients.
The safety and efficacy of Aklief in geriatric patients aged 65 years and above have not been established.
Renal impairment and hepatic impairment.
Aklief has not been studied in patients with renal and hepatic impairment.
Paediatric population.
The safety and efficacy of Aklief in children below 12 years old have not been established.
Method of administration.
For topical use only.
Before using the pump for the first time, prime it by pressing down several times until a small amount of medicine is dispensed (up to 10 times maximum). The pump is now ready to use.
Apply a thin layer of Aklief cream to the affected areas of the face (forehead, nose, chin and right and left cheeks) and all affected areas of the trunk once a day, in the evening, on clean and dry skin:
One pump actuation should be enough to cover the face (i.e. forehead, cheeks, nose, and chin).
Two pump actuations should be enough to cover the upper trunk (i.e. reachable upper back, shoulders and chest). One additional pump actuation may be used for middle and lower back if acne is present.
Patients should be instructed to avoid contact with the eyes, eyelids, lips and mucous membranes and to wash their hands after applying the medicinal product.
The use of a moisturiser is recommended as needed from the initiation of treatment, while allowing sufficient time before and after the application of Aklief cream to allow the skin to dry.4.3 Contraindications
Pregnancy (see Section 4.6 Fertility, Pregnancy and Lactation).
Women planning a pregnancy.
Hypersensitivity to the active substance or to any of the excipients listed, see Section 6.1 List of Excipients.
4.4 Special Warnings and Precautions for Use
Erythema, scaling, dryness, and stinging/burning may be experienced with use of Aklief cream (see Section 4.8 Adverse Effects (Undesirable Effects)). To mitigate the risk of such reactions, patients should be instructed to use a moisturiser from the initiation of treatment, and, if appropriate, reduce the frequency of application of Aklief cream, or suspend use temporarily. Despite mitigation measures, if severe reactions persist the treatment may be discontinued.
The product should not be applied to cuts, abrasions, eczematous or sunburnt skin.
As with other retinoids, use of "waxing" as a depilatory method should be avoided on skin treated with Aklief.
If a reaction suggesting sensitivity to any component of the formula occurs, the use of Aklief should be discontinued. Caution should be exercised if cosmetics or acne medications with desquamative, irritant or drying effects are concomitantly used with the medicinal product, as they may produce additive irritant effects.
Aklief should not come into contact with the eyes, eyelids, lips, or mucous membranes. If the product enter the eye, rinse immediately with plenty of lukewarm water.
Excessive exposure to sunlight, including sunlamps or phototherapy should be avoided during the treatment. An increased risk of sunburn was reported in 1.7% of the children, compared to 0.7% of the adults. Use of a broad-spectrum, water-resistant sunscreen with a Sun Protection Factor (SPF) of 30 or higher and protective clothing over treated areas is recommended when exposure cannot be avoided.
This product contains propylene glycol that may cause skin irritation. Aklief also contains 50 mg alcohol (ethanol) in each gram which is equivalent to 5% w/w. It may cause burning sensation on damaged skin.
Use in the elderly.
No data available.
Paediatric use.
The safety of Aklief in children below 12 years old has not been established.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
Effect of Aklief cream on other medicinal products.
A clinical drug-drug interaction study has shown that topical application of trifarotene did not affect the circulating concentrations of hormonal contraceptives (ethinylestradiol and levonorgestrel) administered by oral route.
Effect of other medicinal products on Aklief cream.
No clinical drug-drug interaction studies were performed to assess effects of other drugs on trifarotene systemic levels (see Section 5.2 Pharmacokinetic Properties).
There is no data on the pharmacodynamic interaction potential of trifarotene. Caution should be exercised if cosmetics or acne medications with desquamative, irritant or drying effects are concomitantly used with the medicinal product, as they may produce additive irritant effects (see Section 4.4 Special Warnings and Precautions for Use).
Pharmacokinetic drug interaction potential.
In vitro studies show that Aklief cream at the concentrations achieved systemically after topical administration did not inhibit the CYP450 isoenzymes CYP1A2, 2B6, 2C8, 2C9, 2C19, 2D6 and 3A4, and did not induce CYP1A2, 2B6, or 3A4.
In vitro studies have shown that Aklief cream at the concentrations achieved systemically after topical administration did not inhibit either MATE, OATP, OAT or OCT uptake transporters or BCRP, PgP, BSEP or MRP efflux transporters.4.6 Fertility, Pregnancy and Lactation
Orally administered retinoids have been associated with congenital abnormalities. When used in accordance with the prescribing information, topically administered retinoids are generally assumed to result into low systemic exposure due to minimal dermal absorption. However, there could be individual factors (e.g. damaged skin barrier, excessive use) that contribute to an increased systemic exposure.
It is difficult to determine the exact time frame in which a patient's system would be completely free of trifarotene. The clinical pharmacology results demonstrated that trifarotene 50 microgram/g cream under maximal use conditions for 4 weeks in patient with acne vulgaris had a low systemic exposure, being non-quantifiable in most of the subjects. However, the daily application of 100 microgram/g cream for 4 weeks resulted in quantifiable drug levels, but still with a low systemic exposure, with a short terminal half-life ranging from 2.4 to 9.1 hours and without accumulation after repeated topical application. One could assume that nearly all the drug would be gone after approximately 3.5 days following the last application. In three and a half days, over 9 half-lives will have been completed. It takes about 5 half-lives for 97% of a drug to be eliminated and 7 half-lives for about 99% of a drug to be eliminated.
Effects on fertility.
No human fertility studies were conducted with Aklief.
Trifarotene showed no adverse effects on functional fertility in rats administered orally at exposures of approximately 1755 (males) and 1726 (females) times the 2 g dose in humans. However, after oral administration to dogs, Germ cell degeneration with pyknotic/apoptotic germ cells was evident from the lowest dose tested of 0.2 mg/kg/day corresponding to a systemic exposure 1368 times higher than those observed in humans. All animals with this finding also showed hypospermatogenesis and debris in the epididymides. The findings did not completely recover after 8 weeks, suggesting an extended and possibly chronic effect. As these effects were noted also at the lowest dose tested, the relevance of the findings for lower doses is unknown.
(Category D)
Aklief is contraindicated (see Section 4.3 Contraindications) during pregnancy or in women planning a pregnancy. If the product is used during pregnancy, or if the patient becomes pregnant while taking this drug, treatment should be discontinued.
In animal reproduction studies, oral administration of trifarotene in pregnant rats and rabbits during organogenesis was teratogenic and embryotoxic at exposures (AUC) that were > 800-times those observed in humans at the maximum recommended human dose (MRHD) of 2 g. Trifarotene was not teratogenic in rats and rabbits at systemic exposures corresponding to approximately 500 and 90-times, respectively, those observed in humans.
It is unknown whether trifarotene or its metabolites are excreted in human milk.
Available data in animals have shown excretion of trifarotene and/or its metabolites in milk.
A risk to the suckling child cannot be excluded.
A decision must be made whether to discontinue breast-feeding or to discontinue/abstain from Aklief therapy taking into account the benefit of breast-feeding for the child and the benefit of therapy for the woman.
To avoid the risk of ingestion by, and/or contact exposure of, an infant, nursing women should not apply trifarotene cream to the chest or breast area.4.7 Effects on Ability to Drive and Use Machines
The effects of trifarotene on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Summary of safety profile.
In the pivotal Phase 3 clinical studies (18251 and 18252), 331 (27.1%) subjects in the Aklief cream group reported 587 treatment-emergent adverse events (TEAEs) compared with 240 (20.0%) subjects in the Vehicle Cream group who reported 338 TEAEs.
Most common TEAEs (i.e. TEAEs reported in at least 1% of subjects in any treatment group were reported by 206 (16.9%) subjects in the Aklief cream group (total of 297 TEAEs) and in 116 (9.7%) subjects in the Vehicle Cream group (total of 140 TEAEs), as shown in Table 1.
 Local cutaneous reactions such as erythema, scaling, dryness, and stinging/burning) were collected separately from other adverse events as a measure of local tolerance. These cutaneous reactions are very common and of mild, moderate and severe intensity for up to 39%, 29.7% and 6.2% of patients, respectively on the face. On the trunk, up to 32.9%, 18.9%, 5.2% of patients had mild, moderate and severe reactions respectively. The maximum severity typically occurred at Week 1 for the face, and at Week 2 to 4 for the trunk, and decreased with continued use of the medication (see Section 4.4 Special Warnings and Precautions for Use).
Local cutaneous reactions such as erythema, scaling, dryness, and stinging/burning) were collected separately from other adverse events as a measure of local tolerance. These cutaneous reactions are very common and of mild, moderate and severe intensity for up to 39%, 29.7% and 6.2% of patients, respectively on the face. On the trunk, up to 32.9%, 18.9%, 5.2% of patients had mild, moderate and severe reactions respectively. The maximum severity typically occurred at Week 1 for the face, and at Week 2 to 4 for the trunk, and decreased with continued use of the medication (see Section 4.4 Special Warnings and Precautions for Use).
The most "commonly" reported adverse reactions as described in Table 2 are application site irritation, application site pruritus and sunburn, occurring in 1.2% to 6.5% of patients treated with Aklief cream in clinical studies.
Tabulated summary of adverse reactions.
Adverse reactions reported in the 12-week vehicle-controlled Phase 3 studies in 1220 patients treated with Aklief cream (and for which the rate for Aklief cream exceeds the rate for vehicle cream) are presented in Table 2.
The adverse reactions are classified by System Organ Class and frequency, using the following convention: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to 1 < 100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000), not known (cannot be estimated from the available data).

Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
Aklief is for once-daily topical use only.
If the medication is applied excessively, no more rapid or better results will be obtained and marked redness, scaling, or skin discomfort may occur. In this event, discontinue use and wait until the skin has recovered.
In case of accidental ingestion, appropriate symptomatic measures should be taken. Chronic ingestion of the drug may lead to the same side effects as those associated with excessive oral intake of vitamin A.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Retinoids for topical use in acne, ATC code: D10AD06.
Mechanism of action.
Aklief cream contains 50 microgram (microgram/g) (w/w) trifarotene, which is a chemically stable, terphenyl acid derivative with retinoid-like activity. It is a relatively potent RARγ agonist (retinoid acid receptor γ agonist), characterised by its high specificity to this receptor over RARα and RARβ (65- and 16-fold, respectively, with no Retinoid X Receptor (RXR) activity).
In addition, trifarotene modulates retinoid target genes (differentiation and inflammatory processes) in human immortalised keratinocytes and human reconstructed epidermis.
Pharmacodynamic effects.
Trifarotene has demonstrated, in the Rhino-mouse model, marked comedolytic activity with the reduction in the comedone count and marked increased epidermis thickness. In this model, trifarotene produced the same comedolytic effect as other known retinoids.
Trifarotene has also shown anti-inflammatory and depigmenting activities.
Clinical trials.
Aklief cream applied once daily in the evening was evaluated for 12 weeks in 2 randomised, multi-centre, parallel group, double-blind, vehicle-controlled studies of identical design. They were conducted in a total of 2420 patients aged, 9 years and older, with moderate facial and truncal acne vulgaris.
Acne severity was evaluated using the 5-point Investigator's Global Assessment (IGA) scale for the face and Physician's Global Assessment (PGA) for the trunk, with moderate acne vulgaris defined as a score of Grade 3-Moderate (see Table 3).
 There were three identical co-primary efficacy endpoints in both pivotal studies 1) the success rate based on the IGA and PGA outcome (percentage of subjects "clear" and "almost clear" and with at least a 2-grade change from baseline) and absolute and percentage change from baseline in 2) inflammatory and 3) non-inflammatory lesion counts at Week 12.
There were three identical co-primary efficacy endpoints in both pivotal studies 1) the success rate based on the IGA and PGA outcome (percentage of subjects "clear" and "almost clear" and with at least a 2-grade change from baseline) and absolute and percentage change from baseline in 2) inflammatory and 3) non-inflammatory lesion counts at Week 12.
Overall, 87% of subjects were Caucasian and 55% were female. Thirty four (1.4%) subjects were 9 to 11 years of age, 1128 (47%) subjects were 12 to 17 years and 1258 (52%) subjects were 18 years and older. All patients had moderate acne vulgaris on the face and 99% on the trunk. At baseline subjects had between 7 and 200 (average 36) inflammatory lesions on the face and between 0 and 220 (average 38) on the trunk. Additionally subjects had 21 to 305 (average 52) non-inflammatory lesions on the face and 0 to 260 (average 46) on the trunk.
The IGA and PGA success rates, mean absolute, and percent reduction in acne lesion counts from baseline after 12 weeks of treatment are presented in Tables 4 and 5.

 Paediatric population.
Paediatric population. Age group 12 to 17 years.
In the phase 3 studies a total of 1128 children aged 12 to 17 years with moderate acne vulgaris were included: 573 of them in study 18251 and 555 children in study 18252.
Long-term efficacy. In Study 3, a one-year open label safety study of 435 patients, 12 years and older (210 children completed 52 weeks of the study), with moderate facial and truncal acne vulgaris, Aklief cream demonstrated a clinically meaningful improvement with IGA and PGA success rates increasing:
from 26.3% at Week 12 visit to 65.9% at Week 52 visit for the face; and
from 38.9% at Week 12 visit to 67.1% at Week 52 visit for the trunk, respectively.
IGA and PGA success experienced by the same subject increased from 21.9% at Week 12 to 58.2% at Week 52.
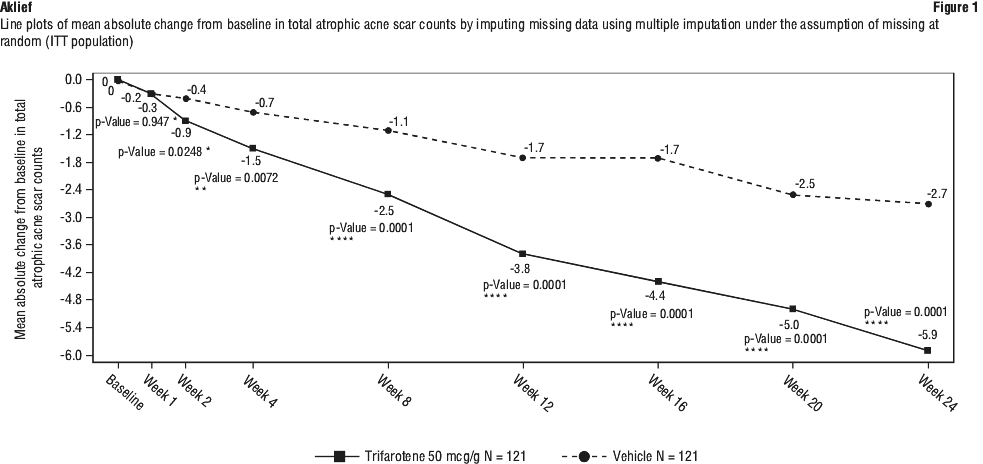
The START study. The effect of Aklief cream in acne scarring during the treatment of acne vulgaris was investigated in the START study. This was a multi-center, randomized, double-blind, vehicle-controlled study using intra-individual comparison (right half-face versus left half-face) investigating the risk of atrophic scar formation during treatment of acne vulgaris in male and female subjects aged 17 to 37 years (n = 121). Subject eligibility was evaluated over a 28-day screening period. Eligible subjects completed baseline assessments and were randomized (left face versus right face) to apply trifarotene cream and trifarotene vehicle cream over a 24-week treatment period, for an intra-individual, split-face comparison.
In all 121 subjects, baseline characteristics were similar in both halves of the face, with acne scar severity being mostly mild and moderate according to SGA (> 80%) and all (100%) acne lesions rated by IGA as moderate and severe.
The median age for all subjects (N = 121) was 22 years, with a minimum of 17 years and a maximum of 34 years. The majority of subjects (102 [84.3%]) were ≥ 18 years old. Subjects were predominantly female (88 [72.7%] subjects), White (97 [80.2%] subjects), and non-Hispanic or Latino (95 [78.5%] subjects).
The primary efficacy endpoint was the absolute change from baseline in total atrophic acne scar count per half-face at Week 24.
The study met its primary endpoint and secondary endpoint in total atrophic acne scar counts. The primary endpoint analysis showed that drug therapy reduced the total number of acne scars (see Table 6).
 The absolute change in atrophic acne scar counts from Baseline to Week 24 showed a greater reduction of scar counts in the trifarotene cream treated area compared to the vehicle cream treated area (-5.9 vs. -2.7 scar counts), with a statistically significant bilateral difference of -3.2 (p-Value < 0.0001). Furthermore, descriptive statistics showed that trifarotene performed numerically better in 2-4 mm compared to > 4 mm atrophic scars. See Figure 1.
The absolute change in atrophic acne scar counts from Baseline to Week 24 showed a greater reduction of scar counts in the trifarotene cream treated area compared to the vehicle cream treated area (-5.9 vs. -2.7 scar counts), with a statistically significant bilateral difference of -3.2 (p-Value < 0.0001). Furthermore, descriptive statistics showed that trifarotene performed numerically better in 2-4 mm compared to > 4 mm atrophic scars. See Figure 1.
5.2 Pharmacokinetic Properties
Absorption.
The absorption of trifarotene from Aklief cream was evaluated in adult and paediatric (10-17 years old) subjects with acne vulgaris. Subjects were treated once daily for 30 days with 2 gram/day of Aklief applied on the face, shoulders, chest, and upper back.
Overall, systemic exposure levels were low and similar between adults and paediatric populations.
After 4 weeks treatment, seven of nineteen (37%) adult subjects had quantifiable trifarotene plasma levels. Cmax ranged from below the limit of quantification (LOQ < 5 picogram/mL) to 10 picogram/mL and AUC0-24h ranged from 75 to 104 picogram.hr/mL.
Three of the seventeen (18%) of paediatric subjects presented quantifiable systemic exposure. Cmax ranged from below the limit of quantification (LOQ < 5 picogram/mL) to 9 picogram/mL and AUC0-24h ranged from 89 to 106 picogram.hr/mL.
Steady state conditions were achieved in both adult and paediatric subjects following 2 weeks of topical administration. No drug accumulation is expected with long-term use.
Distribution.
Trifarotene penetrates into the skin with an exponential distribution from the stratum corneum to the epidermis and dermis.
An in vitro study demonstrated that trifarotene is greater than 99.9% bound to plasma proteins. No significant binding of trifarotene to erythrocytes was observed.
Metabolism.
In vitro studies using human hepatic microsomes and recombinant CYP450 enzymes have shown that trifarotene is primarily metabolised by CYP2C9 and to a lesser extent by CYP3A4, CYP2C8 and CYP2B6.
Excretion.
In nonclinical studies, trifarotene is primarily excreted by the faeces.
5.3 Preclinical Safety Data
Genotoxicity.
Based on the weight of evidence, Aklief cream was considered negative in an in vitro bacterial reverse mutation (Ames) assay, an in vitro mouse lymphoma assay with L5178Y/TK+/- cells and an in vivo micronucleus assay in rats. While an equivocal result was seen an in vitro micronucleus assay in primary human lymphocytes, the weight of evidence indicates a low genotoxic potential with trifarotene.
Carcinogenicity.
Trifarotene was not carcinogenic when topically applied to mice daily for up to 24 months in the vehicle of Aklief cream at doses of up to 0.02 mg/kg (at a concentration of 0.001% w/w). Systemic exposure at the highest dose in mice was 90-fold greater than the anticipated maximum human exposure with Aklief cream. Trifarotene was also not carcinogenic when administered orally in a solution to rats daily for up to 24 months at doses of up to 0.75 mg/kg/day in males and 0.2 mg/kg/day in females. Systemic exposure at the highest dose in rats was 642-(males) and 1642-fold (females) times greater than the anticipated human exposure at the MRHD of Aklief cream.6 Pharmaceutical Particulars
6.1 List of Excipients
Allantoin, Simulgel 600 PHA (acrylamide/sodium acryloyldimethyltaurate copolymer, isohexadecane, polysorbate 80, sorbitan oleate), cyclomethicone, ethanol, phenoxyethanol, propylene glycol, medium chain triglycerides, purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
2 years.
After first opening: use within 6 months.
Information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25 degrees Celsius.
6.5 Nature and Contents of Container
Tube, physician's sample.
5 g.
White Low-density polyethylene (LDPE)/Aluminium (Al)/High density polyethylene (HDPE) laminated tubes with a white high-density polyethylene (HDPE) head and a white polypropylene (PP) closure.
Multidose container with airless pump system.
15 g; 30 g; 75 g.
Polypropylene (PP)/High density polyethylene (HDPE) white airless bottle closed with a white polypropylene (PP) pump and a white polypropylene (PP) overcap.
Pack sizes: 1 tube of 5 g; 1 bottle of 15, 30 or 75 g.
Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
Any unused medicinal product or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Australian Approved Name (AAN): Trifarotene.
Chemical structure.
 Molecular formula: C29H33NO4.
Molecular formula: C29H33NO4.
Molecular weight: 459.6.
CAS number.
895542-09-3.7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Summary Table of Changes


 Local cutaneous reactions such as erythema, scaling, dryness, and stinging/burning) were collected separately from other adverse events as a measure of local tolerance. These cutaneous reactions are very common and of mild, moderate and severe intensity for up to 39%, 29.7% and 6.2% of patients, respectively on the face. On the trunk, up to 32.9%, 18.9%, 5.2% of patients had mild, moderate and severe reactions respectively. The maximum severity typically occurred at Week 1 for the face, and at Week 2 to 4 for the trunk, and decreased with continued use of the medication (see Section 4.4 Special Warnings and Precautions for Use).
Local cutaneous reactions such as erythema, scaling, dryness, and stinging/burning) were collected separately from other adverse events as a measure of local tolerance. These cutaneous reactions are very common and of mild, moderate and severe intensity for up to 39%, 29.7% and 6.2% of patients, respectively on the face. On the trunk, up to 32.9%, 18.9%, 5.2% of patients had mild, moderate and severe reactions respectively. The maximum severity typically occurred at Week 1 for the face, and at Week 2 to 4 for the trunk, and decreased with continued use of the medication (see Section 4.4 Special Warnings and Precautions for Use).
 There were three identical co-primary efficacy endpoints in both pivotal studies 1) the success rate based on the IGA and PGA outcome (percentage of subjects "clear" and "almost clear" and with at least a 2-grade change from baseline) and absolute and percentage change from baseline in 2) inflammatory and 3) non-inflammatory lesion counts at Week 12.
There were three identical co-primary efficacy endpoints in both pivotal studies 1) the success rate based on the IGA and PGA outcome (percentage of subjects "clear" and "almost clear" and with at least a 2-grade change from baseline) and absolute and percentage change from baseline in 2) inflammatory and 3) non-inflammatory lesion counts at Week 12.

 The absolute change in atrophic acne scar counts from Baseline to Week 24 showed a greater reduction of scar counts in the trifarotene cream treated area compared to the vehicle cream treated area (-5.9 vs. -2.7 scar counts), with a statistically significant bilateral difference of -3.2 (p-Value < 0.0001). Furthermore, descriptive statistics showed that trifarotene performed numerically better in 2-4 mm compared to > 4 mm atrophic scars. See Figure 1.
The absolute change in atrophic acne scar counts from Baseline to Week 24 showed a greater reduction of scar counts in the trifarotene cream treated area compared to the vehicle cream treated area (-5.9 vs. -2.7 scar counts), with a statistically significant bilateral difference of -3.2 (p-Value < 0.0001). Furthermore, descriptive statistics showed that trifarotene performed numerically better in 2-4 mm compared to > 4 mm atrophic scars. See Figure 1.
 Molecular formula: C29H33NO4.
Molecular formula: C29H33NO4.