1 Name of Medicine
Fosnetupitant (as chloride hydrochloride).
Palonosetron (as hydrochloride).
2 Qualitative and Quantitative Composition
Each vial of Akynzeo IV contains 235 mg fosnetupitant (as chloride hydrochloride) and 250 microgram palonosetron (as hydrochloride).
Akynzeo IV concentrated injection.
After dilution, 1 mL of solution contains 4.7 mg fosnetupitant and 5 microgram palonosetron (see Section 4.2 Dose and Method of Administration, Method of administration).
For the full list of excipients, see Section 6.1 List of Excipients.
Fosnetupitant is a phosphorylated pro-drug of netupitant. When fosnetupitant is administered intravenously, it is converted rapidly to netupitant, (see Section 5.2 Pharmacokinetic Properties).3 Pharmaceutical Form
Akynzeo IV concentrated injection: is a clear, colourless to slightly yellow solution.
4.1 Therapeutic Indications
Akynzeo IV is indicated in adult patients for:
Prevention of acute and delayed nausea and vomiting associated with initial and repeat courses of highly emetogenic cancer chemotherapy.
Prevention of acute and delayed nausea and vomiting associated with initial and repeat courses of moderately emetogenic cancer chemotherapy.
4.2 Dose and Method of Administration
Dosage.
One vial of Akynzeo IV (235 mg fosnetupitant/250 microgram palonosetron) should be administered as an intravenous infusion over 30 minutes, initiated approximately 30 minutes prior to the start of each chemotherapy cycle. At the end of the infusion, flush the infusion line with the same carrier solution to ensure complete drug administration.
The recommended oral dexamethasone dose should be reduced by approximately 50 % when co-administered with Akynzeo IV (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions, Dexamethasone; Section 5.1 Pharmacodynamic Properties, Clinical trials, Tables 3 and 5).
Method of administration.
Akynzeo IV should be administered intravenously and should not be given by the intramuscular or subcutaneous routes. Intravenous administration should occur through a running intravenous infusion over thirty minutes. Do not administer Akynzeo IV as a bolus injection or undiluted solution.
Instructions for dilution of Akynzeo IV Concentrated Injection before administration:
1. Before administration, inspect the solution for particulate matter and discoloration. Discard the vial if particulates and/or discoloration are observed.
2. Aseptically prepare an infusion vial or bag filled with 30 mL of 5% glucose solution for injection or 0.9% sodium chloride solution for injection.
3. Aseptically withdraw the entire volume of solution from the Akynzeo IV vial and transfer it into the infusion vial or bag containing 30 mL of 5% glucose solution or 0.9% sodium chloride solution for injection to yield a total volume of 50 mL.
4. Gently invert the vial or bag until complete dissolution.
5. Before administration, inspect the final diluted solution for particulate matter and discolouration. Discard the vial or bag if particulates and/or discoloration are observed.
Akynzeo IV must not be reconstituted, diluted, or mixed with solutions for which physical and chemical compatibility has not been established (see Section 6.2 Incompatibilities).
Contains no antimicrobial preservative. Akynzeo IV is to be used for one patient on one occasion only. Discard any residue.
Akynzeo IV concentrated injection.
The final diluted solution should be used immediately and the total time from preparation to the start of infusion with or without intravenous dexamethasone sodium phosphate, should not exceed 24 hours when stored at 25°C.
Compatibility.
Akynzeo IV is compatible with intravenous dexamethasone sodium phosphate which can be added simultaneously.
Elderly population.
No dosage adjustment is necessary for elderly patients. Caution should be exercised when using this product in patients over 75 years, due to the long half-life of the active substances and the limited experience in this population.
Paediatric population.
The safety and efficacy of Akynzeo IV in children have not been established. No data are available.
Patients with renal impairment.
Dosage adjustment for Akynzeo IV is not considered necessary in patients with mild to moderate renal impairment. Use of Ayknzeo IV in patients with severe renal impairment or end stage renal disease should be avoided (see Section 4.4 Special Warnings and Precautions for Use, Use in renal impairment).
Patients with hepatic impairment.
No dosage adjustment is necessary for patients with mild or moderate hepatic impairment (Child-Pugh score 5-8). Limited data exist in patients with severe hepatic impairment (Child Pugh score ≥ 9). As use in patients with severe hepatic impairment may be associated with increased exposure to netupitant, Akynzeo IV should be used with caution in these patients (see Section 4.4 Special Warnings and Precautions for Use, Use in hepatic impairment).4.3 Contraindications
Hypersensitivity to palonosetron, fosnetupitant or netupitant, or to any of the excipients listed in Section 6.1 List of Excipients.
Akynzeo IV is contraindicated during pregnancy (see Section 4.6 Fertility, Pregnancy and Lactation).
4.4 Special Warnings and Precautions for Use
General.
Hypersensitivity reactions to palonosetron may occur in patients who have exhibited hypersensitivity to other selective 5-HT3 receptor antagonists.
Akynzeo IV should not be used to prevent or treat nausea and vomiting in the days following chemotherapy if not associated with another chemotherapy administration.
Constipation.
As palonosetron may increase large bowel transit time, patients with a history of constipation or signs of subacute intestinal obstruction should be monitored following administration.
QT prolongation.
A specific thorough ECG study conducted in adult male and female healthy volunteers on netupitant (200 and 600 mg) orally administered with palonosetron (0.5 and 1.5 mg) demonstrated that Akynzeo capsules had no clinically important effects on ECG parameters. The ECG data analyses including a pharmacodynamic-pharmacokinetic (PD-PK) analysis showed no effects on cardiac repolarisation.
However, since Akynzeo IV contains a 5-HT3 receptor antagonist, caution should be exercised in the concomitant use of Akynzeo IV with medicinal products that increase the QT interval or in patients who have or are likely to develop prolongation of the QT interval. These conditions include patients with a personal or family history of QT prolongation, electrolyte abnormalities, congestive heart failure, bradyarrhythmia, conduction disturbances and in patients taking antiarrhythmic agents or other medicinal products that lead to QT prolongation or electrolyte abnormalities. Hypokalaemia and hypomagnesemia should be corrected prior to Akynzeo IV administration.
Serotonin syndrome.
There have been reports of serotonin syndrome with the use of 5-HT3 antagonists either alone or in combination with other serotonergic drugs including selective serotonin reuptake inhibitors (SSRIs) and serotonin noradrenaline reuptake inhibitors (SNRIs).
Concomitant use of CYP3A4 substrates.
Netupitant is a moderate inhibitor of CYP3A4. It has a half-life of 88 hours so its inhibitory effect on CYP3A4 can last for over 4 days. Systemic exposure to chemotherapy agents and other medicines metabolised by CYP3A4 may increase when administered with or after Akynzeo IV. For example, exposure to docetaxel was shown to increase by 37% with concomitant use. Likewise, exposure to dexamethasone is clearly increased with concomitant use.
Patients should be closely monitored for adverse reactions that may arise from this additional systemic exposure to relevant chemotherapies and other medicines (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions for further details).
Women of childbearing potential/contraception in females.
Women of childbearing potential should not be pregnant or become pregnant while being treated with Akynzeo IV. A pregnancy test should be performed on all pre-menopausal women prior to treatment. Women of childbearing potential must use effective contraception during therapy and up to one month after treatment with this medicinal product (see Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy (Category D)).
Race.
Intravenous palonosetron pharmacokinetics was characterised in twenty-four healthy Japanese subjects over the dose range of 3 - 90 micrograms/kg. Total body clearance was 25% higher in Japanese subjects compared to Whites; however, no dose adjustment is required. The pharmacokinetics of palonosetron in Blacks has not been adequately characterised. The pharmacokinetics of netupitant has been characterised only in Caucasians.
Use in hepatic impairment.
No studies were performed specifically with fosnetupitant in patients with hepatic impairment. However, due to the transient exposure of fosnetupitant and subsequent conversion to netupitant there are no additional warnings considered necessary with the administration of the prodrug compared to the parent compound.
Hepatic impairment does not significantly affect total body clearance of palonosetron compared to the healthy subjects. Dosage adjustment is not necessary in patients with any degree of hepatic impairment.
Maximum concentrations and total exposure of netupitant were increased in subjects with mild (n=8), moderate (n=8), and severe (n=2) hepatic impairment compared to matching healthy subjects, although there was pronounced individual variability in both hepatically-impaired and healthy subjects. Exposure to netupitant (Cmax, AUC0-t and AUC0-∞) compared to matching healthy subjects was 11%, 28% and 19% higher in mild and 70%, 88% and 143% higher in moderate hepatically-impaired subjects, respectively. As such, no dosage adjustment of Akynzeo IV is necessary for patients with mild or moderate hepatic impairment (Child-Pugh score 5-8). Limited data exist in patients with severe hepatic impairment (Child Pugh score ≥ 9). Considerable increased exposure to netupitant can be expected in severe hepatic impairment. Therefore, Akynzeo IV should be used with caution in these patients.
Use in renal impairment.
No specific studies were performed to evaluate fosnetupitant in patients with renal impairment. However, due to the transient exposure of fosnetupitant and subsequent conversion to netupitant there are no additional warnings considered necessary with the administration of the prodrug compared to the parent compound.
In healthy volunteers receiving oral netupitant, less than 5% of all drug-related material was excreted in the urine and less than 1% of the netupitant dose was eliminated unchanged in the urine, any accumulation of netupitant or metabolites after a single dose is negligible. The pharmacokinetics and safety of netupitant has not been studied in patients with renal impairment or chronic kidney disease.
Mild to moderate renal impairment does not significantly affect palonosetron pharmacokinetic parameters. Total systemic exposure of palonosetron increased by approximately 28% in severe renal impairment relative to healthy subjects.
Therefore, no dose adjustment for Akynzeo IV is necessary in patients with mild to moderate renal impairment. Use of Akynzeo IV in patients with severe renal impairment or end-stage renal disease should be avoided.
Use in the elderly.
Population pharmacokinetic analysis and clinical safety and efficacy data for palonosetron did not reveal any differences between cancer patients ≥ 65 years of age and younger patients (18 to 64 years). In healthy elderly subjects (> 65 years old) the mean AUC0-∞ and Cmax was 25% and 36% higher, respectively, for netupitant, and 37% and 10% higher, respectively, for palonosetron compared to those in healthy younger adults (22-45 years old). No dose adjustment is required for these patients. However, caution should be exercised when using Akynzeo IV in patients over 75 years, due to the long half-life of the active substances and the limited experience in this population.
Paediatric use.
The safety and efficacy of Akynzeo IV in children have not been established. No data are available.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
When administered intravenously fosnetupitant is rapidly converted to netupitant. Interactions with other medicinal products following administration of intravenous fosnetupitant are likely to occur with active substances that interact with oral netupitant. The following information, relating to netupitant, was derived from studies conducted with oral netupitant and studies conducted with intravenous fosnetupitant.
In humans, netupitant is eliminated mainly by hepatic metabolism mediated by CYP3A4 with a marginal renal excretion. At a dose of 300 mg in humans, netupitant is a substrate and moderate inhibitor of CYP3A4.
Palonosetron is eliminated from the body through both renal excretion and metabolic pathways, with the latter mediated via multiple CYP enzymes. Further in vitro studies indicated that palonosetron is not an inhibitor of CYP1A2, CYP2A6, CYP2B6, CYP2C9, CPY2D6, CYP2E1 and CYP3A4/5 (CYP2C19 was not investigated) nor does it induce the activity of CYP1A2, CYP2D6, or CYP3A4/5. Therefore, the potential for clinically significant drug interactions with palonosetron appears to be low.
Based on the in vitro studies, netupitant and its metabolites are unlikely to have in vivo drug-drug interactions via inhibition of CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, and CYP2D6 at the clinical dose of 300 mg.
Netupitant and its metabolites, M1, M2 and M3, are not inducers of CYP1A2, CYB2B6, CYP2C9, CYP2C19 and CYP3A4. When Akynzeo IV is used concomitantly with another CYP3A4 inhibitor, netupitant plasma concentrations could be elevated. When Akynzeo IV is used concomitantly with medications that induce CYP3A4 activity, netupitant plasma concentrations could be reduced and this may result in decreased efficacy of Akynzeo IV. Akynzeo IV can increase plasma concentrations of concomitantly administered medications that are metabolised via CYP3A4.
Interaction between netupitant and oral palonosetron.
No clinically relevant pharmacokinetic interactions have been observed between oral netupitant and oral palonosetron.
Interaction with CYP3A4 substrates.
Netupitant, is a moderate inhibitor of CYP3A4. Akynzeo IV should be used with caution in patients receiving concomitant medications that are primarily metabolised through CYP3A4. The plasma concentrations of CYP3A4 substrates can increase when co-administered with Akynzeo IV. Netupitant has a half-life of 88 hours. The inhibitory effect on CYP3A4 can last over 4 days.
Dexamethasone.
Co-administration of a single dose of netupitant (300 mg) with a dexamethasone regimen (20 mg on Day 1, followed by 8 mg twice daily from Day 2 to Day 4) significantly increased the exposure to dexamethasone in a time and dose dependent manner. During the 4-day treatment period, increase of dexamethasone exposure, as expressed by the AUC, ranged from 1.7 to 2.4fold with co-administration of 300 mg netupitant. Similarly, increase of Cmax and Cmin ranged from 1.1 to 1.7-fold and from 3.5 to 5.1-fold, respectively.
Co-administration of a single intravenous dose of fosnetupitant with dexamethasone significantly increased exposure to dexamethasone. The mean AUC of dexamethasone increased by 1.5 fold on Day 1 and 2.4 fold on Day 4 after 260 mg fosnetupitant. Considering the limited fosnetupitant exposure in human plasma and its fast conversion to netupitant, the effects are ascribed to netupitant.
Interaction with chemotherapeutic agents (docetaxel, etoposide, cyclophosphamide).
The systemic exposure of chemotherapy agents metabolised by CYP3A4 can increase when administered with Akynzeo IV. Chemotherapy agents that are known to be metabolised by CYP3A4 include docetaxel, paclitaxel, etoposide, irinotecan, cyclophosphamide, ifosfamide, imatinib, vinorelbine, vinblastine, and vincristine. Exposure to docetaxel and etoposide was increased 37% and 21%, respectively, when co-administered with Akynzeo capsules. Caution and monitoring for chemotherapeutic related adverse reactions are advised in patients receiving chemotherapy agents metabolised primarily by CYP3A4. No consistent effect was seen with cyclophosphamide after netupitant co-administration.
Interaction with oral contraceptives.
Akynzeo capsules, when given with a single oral dose of 60 microgram ethinylestradiol and 300 microgram levonorgestrel had no significant effect on the AUC of ethinylestradiol and increased the AUC of levonorgestrel by 1.4-fold; clinical effects on the efficacy of hormonal contraception are unlikely. No relevant changes in netupitant and palonosetron pharmacokinetics were observed.
Erythromycin and midazolam.
Exposure to erythromycin and midazolam was increased approximately 1.3 and 2.4 fold, respectively, when each was co-administered with netupitant. The pharmacokinetic profile of netupitant was unaffected by the concomitant administration of either midazolam or erythromycin. The potential effects of increased plasma concentrations of midazolam or other benzodiazepines metabolised via CYP3A4 (alprazolam, triazolam) should be considered when co-administering these agents with Akynzeo IV.
Rifampicin.
Single dose Akynzeo capsule was administered with rifampicin, a strong CYP3A4 inducer, following once daily administration of 600 mg rifampicin for 17 days. Pharmacokinetics of netupitant and palonosetron were compared to that after administration of Akynzeo capsules alone. Co-administration of rifampicin decreased the mean Cmax and AUC0-∞ of netupitant by 62% and 82%, respectively, compared to those after Akynzeo capsule alone. Co-administration of rifampicin decreased the mean Cmax and AUC for palonosetron by 15% and 19%, respectively. Co-administration of a strong CYP3A4 inducer can therefore decrease the efficacy of Akynzeo IV.
Ketoconazole.
A single dose Akynzeo capsule was administered with ketoconazole, a strong CYP3A4 inhibitor, following once daily administration of 400 mg ketoconazole for 12 days.
Pharmacokinetics of netupitant and palonosetron were compared to that after administration of Akynzeo capsule alone. Co-administration with ketoconazole increased mean Cmax and AUC of netupitant by 25% and 140%, respectively, compared to those after administration of Akynzeo capsule alone. The mean AUC and Cmax of palonosetron were 10% and 15% higher, respectively, when co-administered with ketoconazole. No dosage adjustment is necessary for single dose administration of Akynzeo IV.
Interaction with warfarin, tolbutamide.
Akynzeo interactions with CYP2C9 substrates (e.g. warfarin, tolbutamide) are unlikely. In vitro studies using human liver microsomes indicate that palonosetron is not an inhibitor of CYP2C9 and netupitant is not a CYP2C9 inducer or inhibitor at clinically relevant concentrations.
Additional interactions with palonosetron.
There have been reports of serotonin syndrome following concomitant use of 5-HT3 antagonists and other serotonergic drugs (including SSRIs and SNRIs). A study in healthy volunteers involving single-dose IV palonosetron (0.75 mg) and steady state oral metoclopramide (10 mg four times daily) demonstrated no significant pharmacokinetic interaction.
In controlled clinical trials, palonosetron injection has been safely administered with corticosteroids, analgesics, anti-emetics/anti-nauseants, antispasmodics and anticholinergic agents.
Palonosetron did not inhibit the antitumor activity of the five chemotherapeutic agents tested (cisplatin, cyclophosphamide, cytarabine, doxorubicin and mitomycin C) in murine tumour models.
Additional interactions with netupitant.
Based on in vitro studies, netupitant is an inhibitor of P-glycoprotein (P-gp). It cannot be excluded that this effect may be more marked, and then clinically relevant, in cancer patients, notably those having abnormal renal function. Therefore, caution is recommended when netupitant is combined with digoxin or with other P-gp substrates such as dabigatran or colchicine. In addition, metabolite M2 is a substrate for P-gp. Netupitant's potential for being a substrate for P-gp is unknown.
In vitro studies indicate that netupitant and its three major metabolites are unlikely to have in vivo drug-drug interactions with human efflux transporters BSEP, MRP2, and human uptake transporters OATP1B1, OATP1B3, OAT1, OAT3, OCT1, and OCT2 at the clinical dose of 300 mg.
Based on in vitro studies, netupitant is an inhibitor of BCRP transporters. Caution is recommended when netupitant is combined with an oral substrate of glucuronidation isozyme UGT2B7 (e.g. zidovudine, valproic acid, morphine).4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No studies on the effect on fertility have been conducted with the fosnetupitant/palonosetron combination.
No effect on fertility has been observed with fosnetupitant in animal studies in IV doses up to 39 mg/kg/day (> 56 times the human AUC for both fosnetupitant and netupitant at the recommended human dose).
Daily oral administration of up to 30 mg/kg netupitant in female (3 times the human AUC at the recommended human dose) and male rats had no effects on fertility or reproductive performance. Palonosetron at oral doses of up to 60 mg/kg/day (> 30 times the human exposure based on plasma AUC) was found to have no effect on fertility and reproductive performance of male and female rats. Oral dosing of male rats for two months prior to mating was associated with complete infertility at the 120 mg/kg/day dose. Testicular degeneration was confirmed in a 3-month general toxicity study at oral doses of 60 and 120 mg/kg/day. An IV dose of up to 10 mg/kg/day (> 30 times the human exposure based on plasma AUC) had no effect on male fertility and reproductive performance.
(Category D)
Palonosetron had no effect on fetal development at oral doses of up to 18 mg/kg/day in rats and 90 mg/kg/day in rabbits. At 60 and 120 mg/kg/day in rats, fetal weight was reduced.
Palonosetron did not cause fetal abnormalities at these dose levels. However, palonosetron had toxic effects on the dams at 120 mg/kg in rats and 90 mg/kg/day in rabbits.
No effects on embryo-fetal development were observed following daily administration of netupitant in pregnant rats during the period of organogenesis at doses up to 3 times the human AUC at the recommended human dose. However, an increased incidence of positional fetal abnormalities of the limbs and paws, fused sternebrae and agenesis of accessory lung lobe were observed following daily administration of netupitant to rabbits at oral doses of 10 mg/kg/day and higher during the period of organogenesis. In a pilot study in rabbits, cleft palate, microphthalmia and aphakia were observed in fetuses following a maternal dose of 30 mg/kg/day Exposure at the no observed adverse effect level was less than the anticipated human AUC.
In animal reproduction studies, no effects on embryo-fetal development were observed following daily administration of fosnetupitant in pregnant rats during the period of organogenesis at doses up to 13.2 mg/kg/day (2.1 times the human AUC for netupitant at the recommended human dose). At 39.5 mg/kg/day (3.2 times the human AUC for netupitant at recommended human dose), the incidence of unossified pubis was increased. An increase in intrauterine deaths (early/late resorption and dead fetuses) was observed following daily administration of fosnetupitant in pregnant rabbits during the period of organogenesis with doses ≥ 6.25 mg/kg/day at least 20 times the human AUC for fosnetupitant and 0.6 times the human AUC for netupitant at the recommended human dose.
There are no data from the use of palonosetron and netupitant in pregnant women. No studies on the effect on embryofetal development have been conducted with the fosnetupitant/palonosetron combination. Akynzeo IV is contraindicated in pregnancy.
It is not known whether palonosetron or netupitant are excreted in human milk. A risk to the newborn/infant cannot be excluded. Breastfeeding should be discontinued during treatment with Akynzeo IV and for one month after the last dose. Pre- and post-natal development study in rats with netupitant does not indicate direct or indirect harmful effects on F1 and F2 generations, after daily administration of netupitant up to 3 times the human AUC at the recommended human dose. Daily administration of 39.5 fosnetupitant in rats (3.2 times the human AUC for netupitant at the recommended human dose) during organogenesis through lactation increased pre-birth loss and pup loss, reduced bodyweight of F1 pups and delayed development (pinna detachment, eye opening, preputial separation and startle response to sound), associated with reduced maternal body weights. No effects were observed in offspring or dams at 13 mg/kg/day (1.3 times the human AUC for netupitant at the recommended human dose).4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed with Akynzeo IV. Akynzeo IV may induce dizziness, somnolence or fatigue, therefore patients should use caution when driving or operating machines.
4.8 Adverse Effects (Undesirable Effects)
The safety of Akynzeo IV was evaluated in 203 patients in an active-controlled multi-cycle (median 4 cycles, range 1-4 cycles) safety clinical study in patients receiving HEC (including cisplatin, cyclophosphamide, carmustine, dacarbazine and mechloretamine) compared to 201 patients receiving Akynzeo capsules. All patients received a single dose of Akynzeo IV 30 minutes prior to the start of each chemotherapy cycle; dexamethasone was co-administered with Akynzeo IV. The safety profile of Akynzeo IV was similar to that seen with Akynzeo capsules.
The safety profile of Akynzeo capsules were evaluated in 1169 cancer patients, including 782 exposed to Akynzeo capsules for at least 4 cycles and 321 exposed for at least 6 cycles, up to a maximum of 12 cycles of chemotherapy. All patients received a single oral dose of Akynzeo capsules one hour prior to the start of each chemotherapy cycle. In all studies, dexamethasone was co-administered with Akynzeo capsules (see Section 5.1 Pharmacodynamic Properties, Clinical trials, Tables 3 and 5).
Adverse reactions, considered as drug-related by the investigator, were reported in approximately 13% of patients treated with Akynzeo capsules. Akynzeo was discontinued due to adverse reactions in 3.7 % of patients. The adverse reaction profile was similar across all cycles. The following is a listing of the adverse reactions reported by ≥ 1% of patients (common adverse events) treated with Akynzeo capsules for one or more cycles. See Table 1.
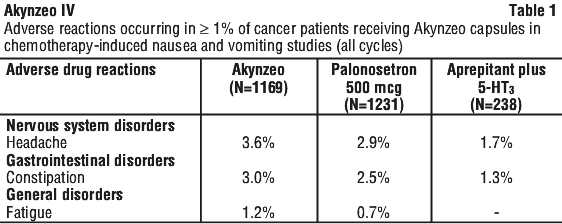 The following uncommon (≥ 0.1% to < 1%) and rare (≥ 0.01% to < 0.1%) adverse reactions were reported as treatment-related adverse events in at least two cancer patients receiving Akynzeo capsules in chemotherapy-induced nausea and vomiting studies (see Table 2):
The following uncommon (≥ 0.1% to < 1%) and rare (≥ 0.01% to < 0.1%) adverse reactions were reported as treatment-related adverse events in at least two cancer patients receiving Akynzeo capsules in chemotherapy-induced nausea and vomiting studies (see Table 2):
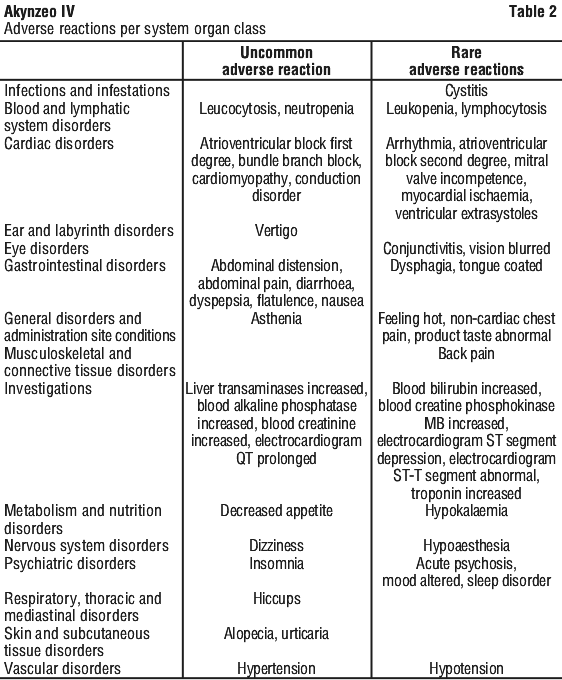
Post marketing experience.
Very rare cases of hypersensitivity reactions including anaphylaxis and shock have been reported from post-marketing use of intravenous palonosetron. There is no post marketing data available at this time for Akynzeo IV.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
No case of overdose has been reported and no specific information is available on the treatment of overdose with Akynzeo IV. Netupitant doses up to 600 mg and palonosetron doses up to 6 mg have been used in clinical studies without any safety concerns. In the unlikely event of overdose, Akynzeo IV should be discontinued and general supportive treatment and monitoring should be provided. Because of the antiemetic activity of netupitant and palonosetron, emesis induced by a medicinal product may not be effective. Dialysis studies have not been performed, however, due to the large volume of distribution of palonosetron and netupitant, dialysis is unlikely to be an effective treatment for Akynzeo IV overdose.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Anti-emetics and anti-nauseants; serotonin (5-HT3) and neurokinin-1 (NK1) receptor antagonists.
ATC code: A04AA55 palonosetron, combinations.
NK1 receptor occupancy.
The receptor occupancy for the Chemotherapy Induced Nausea and Vomiting (CINV) dosing regimen of netupitant has been determined in a human Positron Emission Tomography (PET) study with three single doses tested (100 mg, 300 mg and 450 mg, 2 subjects per dose). Netupitant was shown to cross the blood brain barrier with a high occupancy of brain NK1 receptors. All doses achieved a relatively long duration of blockade of the NK1 receptor. NK1 receptor occupancy in striatum at 6, 24, 48, 72 and 96 hours after administration of 300 mg netupitant was 92.5%, 86.5%, 85.0%, 78.0% and 76.0%, respectively.
Mechanism of action.
Fosnetupitant is the prodrug of netupitant and when administered intravenously is converted rapidly to netupitant (see Section 5.2 Pharmacokinetic Properties). Netupitant is a selective high-affinity antagonist of human substance P/neurokinin 1 (NK1) receptors. Delayed emesis has been largely associated with the activation of tachykinin family neurokinin 1 (NK1) receptors (broadly distributed in the central and peripheral nervous systems) by substance P. As shown in in vitro and in vivo studies, netupitant and palonosetron can contribute to the inhibition of substance P mediated response. In vitro studies have demonstrated that while palonosetron does not bind to the NK1 receptor, it does inhibit cisplatin-induced substance P enhancement of NK1 signalling, in a dose-dependent manner.
Palonosetron is a selective serotonin subtype 3 (5-HT3) receptor antagonist with a strong binding affinity for this receptor. Cancer chemotherapy may be associated with a high incidence of nausea and vomiting, particularly when certain agents, such as cisplatin, are used. 5-HT3 receptors are located on the nerve terminals of the vagus in the periphery and centrally in the chemoreceptor trigger zone of the area postrema. It is thought that chemotherapeutic agents produce nausea and vomiting by releasing serotonin from the enterochromaffin cells of the small intestine and that the released serotonin then activates 5-HT3 receptors located on vagal afferents to initiate the vomiting reflex. The development of acute emesis is known to depend on 5-Hydroxytryptamine serotonin (5-HT) and the 5-HT3 receptor has been demonstrated to selectively participate in the emetic response.
Clinical trials.
Oral administration of Akynzeo in combination with dexamethasone has been shown to prevent acute and delayed nausea and vomiting associated with highly and moderately emetogenic cancer chemotherapy in two separate pivotal studies.
Highly emetogenic chemotherapy (HEC) study.
In a multi-centre, randomised, parallel, double-blind, controlled clinical study of 694 patients the efficacy and safety of single doses of oral netupitant in combination with oral palonosetron, were compared with a single oral dose of palonosetron in cancer patients receiving a chemotherapy regimen that included cisplatin (median dose = 75 mg/m2). Lung and respiratory tract, head and neck, and ovarian cancers were the most frequent cancer types; approximately half of the enrolled patients had metastatic disease.
The primary efficacy analysis included 135 patients who received a single oral dose of netupitant 300 mg plus palonosetron 500 microgram, and 136 patients who received oral palonosetron 500 microgram alone.
The treatment regimens for the oral netupitant 300 mg plus palonosetron 500 microgram and the palonosetron 500 microgram arms are defined in the following table (Table 3).
 Of the 135 patients who received netupitant 300 mg plus palonosetron 500 microgram, 43% were women, and all patients were White. The age ranged from 19 to 77 years, with a mean age of 53 years. During the study 86% of the 135 treated patients in the netupitant 300 mg plus palonosetron 500 microgram arm received a concomitant chemotherapeutic agent in addition to protocol-mandated cisplatin. The most common chemotherapeutic agents and the proportion of patients exposed were cyclophosphamide (34%), fluorouracil (24%), etoposide (21%) and doxorubicin (16%).
Of the 135 patients who received netupitant 300 mg plus palonosetron 500 microgram, 43% were women, and all patients were White. The age ranged from 19 to 77 years, with a mean age of 53 years. During the study 86% of the 135 treated patients in the netupitant 300 mg plus palonosetron 500 microgram arm received a concomitant chemotherapeutic agent in addition to protocol-mandated cisplatin. The most common chemotherapeutic agents and the proportion of patients exposed were cyclophosphamide (34%), fluorouracil (24%), etoposide (21%) and doxorubicin (16%).
The primary efficacy endpoint was complete response (CR) rate (defined as no emetic episodes, no rescue medication) within 120 hours (overall phase) after the start of the highly emetogenic chemotherapy administration.
Major secondary efficacy endpoints included:
CR for the 0-24-hour interval (acute phase) and for the 25-120 hour interval (delayed phase);
Complete protection (defined as no emesis, no rescue therapy, no significant nausea) for the overall, acute and delayed phases;
No emesis for the overall, acute and delayed phases;
No significant nausea (maximum VAS < 25 mm) and no nausea (maximum VAS < 5 mm) for the overall, acute and delayed phases.
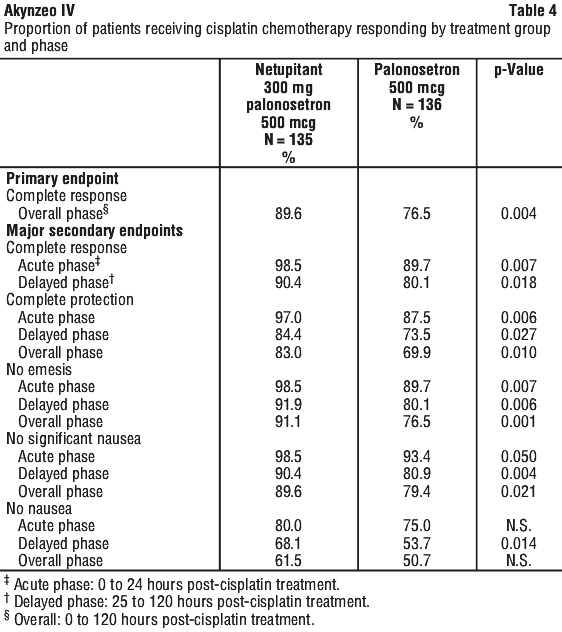
A summary of the key results from this study is shown in Table 4.
The proportion of patients with CR in the overall phase was 13.2% (95% CI: 4.4 to 21.9%) higher (p-value = 0.004) in the netupitant 300 mg plus palonosetron 500 microgram group (89.6%) than in the palonosetron group (76.5%). In the acute phase, the proportion of patients with CR was 8.8% higher (p-value=0.007) in the netupitant 300 mg plus palonosetron 500 microgram group (98.5%) than in the palonosetron group (89.7%). In the delayed phase, the proportion of patients with CR was 10.2% higher (p-value = 0.018) in the netupitant 300 mg plus palonosetron 500 microgram group (90.4%) than in the palonosetron group (80.1%).
Moderately emetogenic chemotherapy (MEC)1 study.
In a multicentre, randomised, parallel, double-blind, active-controlled, superiority study, the efficacy and safety of a single oral dose of Akynzeo was compared with a single oral dose of palonosetron 500 microgram in cancer patients scheduled to receive the first cycle of an anthracycline and cyclophosphamide regimen for the treatment of a solid malignant tumour.
Almost all patients (97%) had breast cancer, which was metastatic in 16% of cases.
At the time of the study, anthracycline-cyclophosphamide containing chemotherapy regimens were considered to be moderately emetogenic. Recent guidelines have updated these regimens to highly emetogenic.
All patients received a single oral dose of dexamethasone.
1 At the time of the study, anthracycline-cyclophosphamide containing chemotherapy regimens were considered to be moderately emetogenic. Recent guidance has updated these regimens to highly emetogenic.
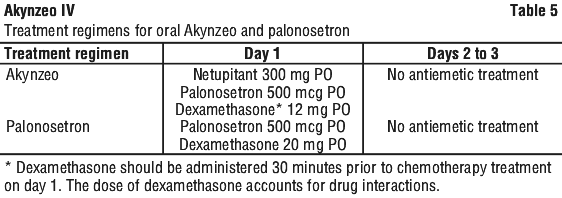
The treatment regimens for the oral Akynzeo and the palonosetron 500 microgram PO arms are displayed in the following table (Table 5).
 After completion of cycle 1, patients had the option to participate in a multiple-cycle extension, receiving the same treatment as assigned in cycle 1. There was no pre-specified limit of the number of repeat consecutive cycles for any patient. A total of 1450 patients (Akynzeo n=725; Palonosetron n=725) actually received study medication: of these, 1438 patients (98.8%) completed cycle 1 and 1286 patients (88.4%) continued treatment in the multiple-cycle extension. A total of 907 patients (62.3%) completed the multiple-cycle extension up to a maximum of eight treatment cycles.
After completion of cycle 1, patients had the option to participate in a multiple-cycle extension, receiving the same treatment as assigned in cycle 1. There was no pre-specified limit of the number of repeat consecutive cycles for any patient. A total of 1450 patients (Akynzeo n=725; Palonosetron n=725) actually received study medication: of these, 1438 patients (98.8%) completed cycle 1 and 1286 patients (88.4%) continued treatment in the multiple-cycle extension. A total of 907 patients (62.3%) completed the multiple-cycle extension up to a maximum of eight treatment cycles.
Of the 725 patients who received Akynzeo study medication, 711 (98%) were women; 79% were White, 14% Asian, 6% Hispanic, and < 1% were Black or Other. Age ranged from 22 to 79 years, with a median age of 54 years. A total of 724 patients (99.9%) were treated with cyclophosphamide. All patients were additionally treated with either doxorubicin (68.0%) or epirubicin (32.0%).
During the first cycle, 32% of the 725 patients treated with Akynzeo received a concomitant chemotherapeutic agent in addition to protocol-mandated regimens, with the most common chemotherapeutic being fluorouracil (28.3%) and docetaxel (2.6%).
The primary efficacy endpoint was the CR rate in the delayed phase, 25-120 hours after the start of the chemotherapy administration. Major secondary efficacy endpoints included:
CR for the acute and the overall phase;
Complete protection (defined as no emesis, no rescue therapy, no significant nausea) for the delayed, the acute and the overall phase;
No emesis for the delayed, the acute and the overall phase;
No significant nausea (maximum VAS < 25 mm) and no nausea (maximum VAS < 5 mm) for the delayed, the acute and the overall phase.
A summary of the key results from Study 2 is shown in Table 6.
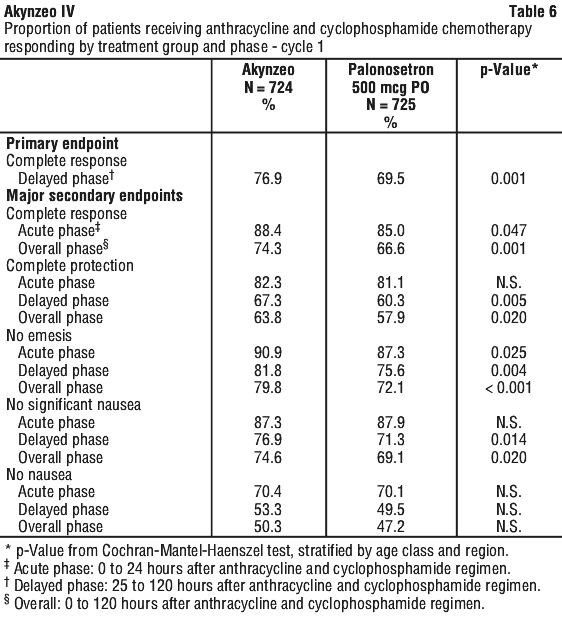
Complete response.
The primary efficacy analysis was conducted in 1449 randomised patients who received the chemotherapy regimen and the study treatment. The proportion of patients with CR in the delayed phase was 7.4% (95% CI: 2.9 to 11.9%) higher (p-value = 0.001) in the Akynzeo group (76.9%) than in the palonosetron group (69.5%). In the acute phase, the proportion of patients with CR was 3.4% higher (p-value=0.047) in the Akynzeo group (88.4%) than in the palonosetron group (85.0%). In the overall phase, the proportion of patients with CR was 7.7% higher (p-value = 0.001) in the Akynzeo group (74.3%) than in the palonosetron group (66.6%).
Multiple cycles.
Patients continued into the Multiple-Cycle extension for up to 7 additional cycles of chemotherapy. The proportion of patients with complete response and no significant nausea in the delayed phase by treatment group at each cycle (cycles 2 to 6) is displayed in Figure 1. A limited number of patients received treatment beyond cycle 6. During all cycles the CR rate and the proportion of patients with no significant nausea rates in the delayed phase were higher for Akynzeo than for palonosetron PO. Antiemetic activity of Akynzeo was maintained throughout repeat cycles for those patients continuing in each of the multiple cycles.
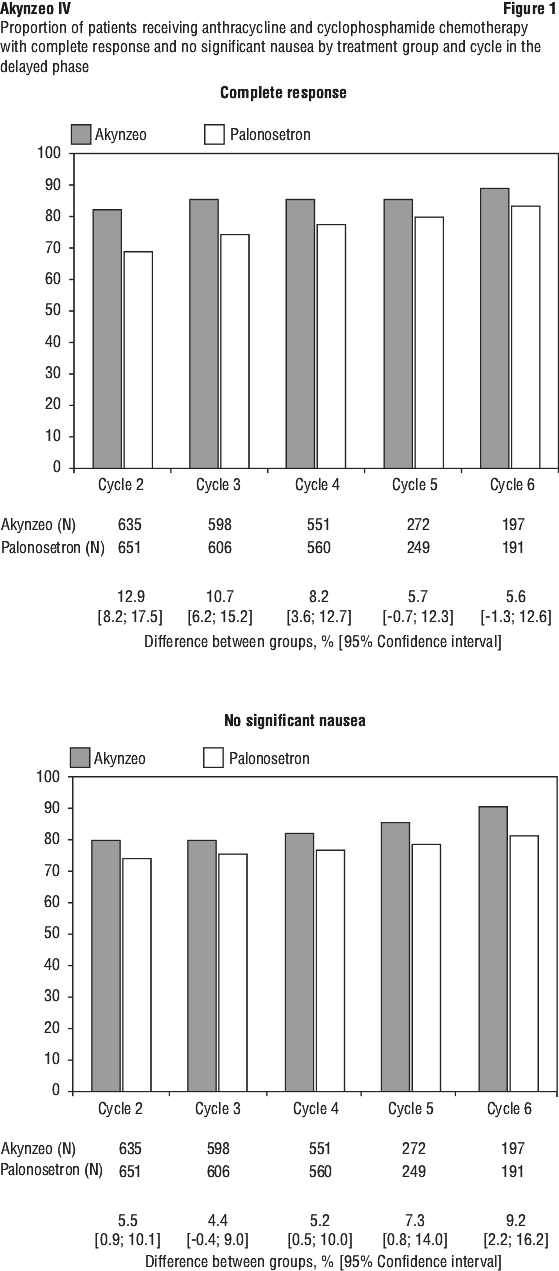
Patient reported outcomes.
The impact of nausea and vomiting on patients' daily lives was assessed using the Functional Living Index-Emesis (FLIE), a validated specific patient-reported outcome measure of the impact of nausea and vomiting on daily life. The proportion of patients with Overall no impact in daily life was 6.3% higher (p-value =0.005) in the Akynzeo group (78.5%) than in the palonosetron group (72.1%).
The proportion of patients with no impact in daily life of the Vomiting Domain was 5.6% higher (p-value =0.001) in the Akynzeo group (90.1%) than in the palonosetron group (84.4%). The proportion of patients with no impact in daily life of the Nausea Domain was 5.8% higher (p-value =0.015) in the Akynzeo group (71.5%) than in the palonosetron group (65.8%).
Clinical studies for Akynzeo IV.
In a multicenter, multinational, randomised, active controlled, double blind, double dummy, parallel group, clinical non-inferiority study, the efficacy of a single dose of intravenous palonosetron 250 micrograms administered as a 30-minute IV infusion was compared to palonosetron 250 micrograms administered as a 30-second IV bolus for the prevention of CINV in cancer patients scheduled to receive a HEC regimen. HEC regimens included cisplatin administered as a single IV dose of 70 mg/m2, cyclophosphamide 1500 mg/m2, carmustine (BCNU) > 250 mg/m2, dacarbazine (DTIC) and mechloretamine (nitrogen mustard). A total of 440 patients (intravenous palonosetron infusion n = 225; intravenous palonosetron bolus n = 215) received study medication and HEC.
The primary efficacy endpoint was complete response (CR defined as no emetic episode and no use of rescue medication) in the 24 hours (acute phase) after the start of the scheduled chemotherapy. In the IV infusion group, 82.7% of patients achieved CR in the acute phase compared to 86.5% of patients in the IV bolus group, with a difference of -3.8% (99% CI: - 12.2% to 4.7%). Non-inferiority of the IV infusion group versus the IV bolus group in the prevention of CINV in cancer patients receiving HEC was demonstrated as the lower limit of the two-sided 99% CI for the difference in 22 proportions of patients with CR was greater (i.e. closer to zero) than the pre-defined non inferiority margin set at 15%.
Paediatric population.
The safety and efficacy of Akynzeo IV in children have not been established. No data are available.
5.2 Pharmacokinetic Properties
Absorption.
After single dose administration of Akynzeo IV administered as a 30-minute infusion to cancer patients, fosnetupitant achieved Cmax at the end of the infusion and disappeared rapidly from the systemic circulation with an apparent terminal half-life lower than one hour. Within 30 minutes of infusion completion, fosnetupitant concentration decreased to less than 1% of Cmax. See Table 7.
 Fosnetupitant Cmax and AUC were lower in patients than in healthy subjects, although the systemic exposures to netupitant were comparable.
Fosnetupitant Cmax and AUC were lower in patients than in healthy subjects, although the systemic exposures to netupitant were comparable.
In healthy subjects, there was a dose-proportional increase in the systemic exposure of fosnetupitant with the dose increase of fosnetupitant from 17.6 to 353 mg.
Distribution.
Netupitant.
After a single oral 300 mg dose administration in cancer patients, netupitant disposition was characterised by a two-compartment model with an estimated median systemic clearance of 20.5 L/h and a large distribution volume in the central compartment (486 L). Human plasma protein binding of netupitant and its two major metabolites M1 and M3 is > 99% at drug concentrations ranging from 10 to 1500 nanogram/mL. The third major metabolite, M2, is > 97% bound to plasma proteins.
Palonosetron.
Palonosetron has a volume of distribution of approximately 8.3 ± 2.5 L/kg. Approximately 62% of palonosetron is bound to plasma proteins.
Fosnetupitant.
After intravenous administration of Akynzeo IV, fosnetupitant plasma concentrations declined according to a bi-exponential profile that reflects rapid distribution. The mean volume of distribution (Vz) in patients was 296 ± 535 L. The human plasma protein binding of fosnetupitant was 92.14% at 1 microM and 94.86% at 10 microM. The free fraction was in the range 5 - 8%.
Metabolism.
Netupitant.
In humans, netupitant is eliminated mainly by hepatic metabolism mediated by CYP3A4. Three major metabolites have been detected in human plasma at netupitant oral doses of 30 mg and higher (the desmethyl derivative, M1; the N-oxide derivative, M2; the OH-methyl derivative, M3). After administration of a single oral dose of 300 mg netupitant, mean plasma netupitant/plasma radioactivity ratios ranged from 0.13 to 0.49 over 96 h post-dose. The ratios were time dependent with values decreasing gradually beyond 24 h post-dose, indicating that the drug is being rapidly metabolised. Mean Cmax was approximately 11%, 47% and 16% of the parent for M1, M2 and M3, respectively; M2 had the lowest AUC relative to the parent (14%) whereas M1 and M3 AUC were approximately 29% and 33% of the parent, respectively. M1, M2 and M3 metabolites were all shown to be pharmacologically active in an animal pharmacodynamic model, where M3 was most potent and M2 least active.
At a dose of 300 mg in humans, netupitant is a substrate and moderate inhibitor of CYP3A4. Netupitant and its metabolites, M1, M2 and M3, are not inducers of CYP1A2, CYP2C9, CYP2C19 and CYP3A4. When Akynzeo is used concomitantly with another CYP3A4 inhibitor, netupitant plasma concentrations could be elevated. When Akynzeo is used concomitantly with medications that induce CYP3A4 activity, netupitant plasma concentrations could be reduced and this may result in decreased efficacy of Akynzeo. Akynzeo can increase plasma concentrations of concomitantly administered oral medications that are metabolised via CYP3A4 (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Palonosetron.
Palonosetron is eliminated by multiple routes with approximately 50% metabolised to form two primary metabolites: N-oxide-palonosetron and 6-S-hydroxy-palonosetron. These metabolites each have less than 1% of the 5-HT3 receptor antagonist activity of palonosetron. In vitro metabolism studies have suggested that CYP2D6 and to a lesser extent, CYP3A4 and CYP1A2 are involved in the metabolism of palonosetron. However, clinical pharmacokinetic parameters are not significantly different between poor and extensive metabolisers of CYP2D6 substrates.
Fosnetupitant.
Fosnetupitant is rapidly converted in vivo to netupitant by metabolic hydrolysis. In patients receiving Akynzeo IV, netupitant exposure was 17-fold fosnetupitant exposure, as determined by their AUC ratio. Netupitant metabolites M1, M2 and M3 were rapidly generated from the released netupitant. In patients, metabolite M1, M2 and M3 exposures were 32%, 21% and 28% of netupitant exposure, as determined by their AUC ratio. The median tmax for M1, M2, and M3 were 12, 2 and 12 hours, respectively.
Excretion.
Netupitant.
Netupitant is eliminated from the body in a multi-exponential fashion, with an apparent mean elimination half-life of 88 hours in cancer patients (after a single oral dose of Akynzeo capsules).
Renal clearance is not a significant elimination route for the netupitant-related entities. The mean fraction of an oral dose of netupitant excreted unchanged in urine is less than 1%; a total of 3.95% and 70.7% of the radioactive dose was recovered in the urine and faeces, respectively. Approximately half the radioactivity administered orally as [14C]-netupitant was recovered from urine and faeces within 120 h of dosing. Elimination via both routes was estimated to be complete by Day 29-30 post-dose.
Palonosetron.
Following administration of a single oral 0.75 mg dose of [14C]-palonosetron to six healthy subjects, 85% to 93% of the total radioactivity was excreted in urine, and 5% to 8% was eliminated in faeces. The amount of unchanged palonosetron excreted in the urine represented approximately 40% of the administered dose. In healthy subjects given palonosetron capsules 500 microgram, the terminal elimination half-life (t½) of palonosetron was 37 ± 12 hours (mean ± SD), and in cancer patients, t½ was 48 ± 19 hours. After a single dose of approximately 0.75 mg intravenous palonosetron, the total body clearance of palonosetron in healthy subjects was 160 ± 35 mL/h/kg (mean ± SD) and renal clearance was 66.5 ± 18.2 mL/h/kg.
Fosnetupitant.
After Akynzeo IV administration, fosnetupitant plasma concentrations declined according to a bi-exponential profile. Thirty minutes after the end of the infusion, the mean plasma concentration of fosnetupitant was less than 1% of Cmax.
5.3 Preclinical Safety Data
Genotoxicity.
Fosnetupitant.
Fosnetupitant was not genotoxic in the Ames test and in in vivo rat micronucleus test. In human lymphocytes, fosnetupitant did not induce structural abberations.
Netupitant.
Netupitant did not show mutagenic or clastogenic activity in a standard battery of in vitro and in vivo genotoxicity tests (Ames, gene mutation in mouse lymphoma cells and rat micronucleus test).
Palonosetron.
Palonosetron was not genotoxic in the Ames test, the Chinese hamster ovarian cell (CHO/HGPRT) forward mutation test, the ex vivo hepatocyte unscheduled DNA synthesis test or the mouse micronucleus test. It was, however, positive for clastogenic effects in the CHO cell chromosomal aberration test.
Carcinogenicity.
Fosnetupitant.
Long term studies in animals to evaluate carcinogenic potential have not been performed with fosnetupitant.
Netupitant.
No carcinogenicity study was performed with netupitant.
Palonosetron.
In a 104-week carcinogenicity study in CD-1 mice, animals were treated with oral doses of palonosetron at 10, 30 and 60 mg/kg/day. Treatment with palonosetron was not tumorigenic. The highest tested dose produced a systemic exposure to palonosetron (plasma AUC) of > 100 times the human exposure at the recommended oral of 500 micrograms in the combination.
In a 104-week palonosetron carcinogenicity study in Sprague-Dawley rats, male and female rats were treated with oral doses of 15, 30 and 60 mg/kg/day and 15, 45 and 90 mg/kg/day, respectively. The lowest and highest doses, respectively, produced a systemic exposure to palonosetron (plasma AUC) of > 4.4 times and > 95.5 times the human exposure at the recommended dose.
Treatment with palonosetron produced increased incidences of adrenal benign pheochromocytoma and combined benign and malignant pheochromocytoma in both male and female rats, of pancreatic Islet cell adenoma and combined adenoma and carcinoma of pancreatic acinar cell adenoma and combined adenoma and adenocarcinoma and of pituitary adenoma in male rats. Increased incidences of skin keratoacanthomas and tail squamous cell papillomas were also observed, mainly in males. In female rats, palonosetron produced hepatocellular adenoma and combined hepatocellular adenoma and carcinoma, and increased the incidences of thyroid C-cell adenoma and combined adenoma and carcinoma, and of mammary gland adrenocarcinoma.6 Pharmaceutical Particulars
6.1 List of Excipients
Akynzeo IV concentrated injection.
Mannitol, disodium edetate, sodium hydroxide (for pH adjustment), hydrochloric acid (for pH adjustment), water for injection.
6.2 Incompatibilities
Akynzeo IV is incompatible with any solutions containing divalent cations (e.g. Ca2+, Mg2+), including Hartmann's and lactated Ringer's solutions.
Limited data are available on the compatibility of Akynzeo IV with other intravenous substances, additives or other medications with the exception of intravenous dexamethasone sodium phosphate (See Section 4.2 Dose and Method of Administration), and they should not be added to Akynzeo IV or infused simultaneously. If the same intravenous line is used for sequential infusion of several different medicinal products, flush the line before and after infusion of Akynzeo IV with 0.9% sodium chloride solution for injection.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Akynzeo IV concentrated injection.
Store below 25°C. Do not freeze. Store in the original container. Protect from light.
The final diluted solution should be used immediately and the total time from preparation to the start of infusion should not exceed 24 hours when stored at 25°C.
6.5 Nature and Contents of Container
Akynzeo IV concentrated injection.
One 20 mL Type I glass vial with a rubber stopper with an aluminium cap and violet polypropylene flip-off disc.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Upon reconstitution prior to administration, the pH of Akynzeo IV should be between 7.5 and 10.0.
Chemical structure.
Fosnetupitant chloride hydrochloride is chemically described as 2-(3,5-bis-trifluoromethylphenyl)-N-methyl-N[6-(4-methyl-4-O-methylene-phosphatepiperazinium-1-yl)-4-o-tolyl-pyridin-3-yl]-isobutyramide chloride hydrochloride. The empirical formula is C31H37Cl2F6N4O5P, with a molecular weight 761.5. Fosnetupitant chloride hydrochloride exists as a single isomer and has the following structural formula:
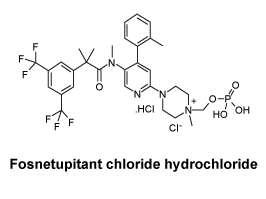
CAS number.
Fosnetupitant chloride hydrochloride: 1643757-72-5.
Fosnetupitant (free base): 1431216-59-9.
Chemical structure.
Palonosetron hydrochloride is chemically described: (3aS)-2-[(S)-1-Azabicyclo [2.2.2]oct-3yl]2,3,3a,4,5,6-hexahydro-1-oxo-1H-benz[de]isoquinoline hydrochloride. The empirical formula is C19H24N2O.HCl, with a molecular weight of 332.9. Palonosetron hydrochloride exists as a single isomer and has the following structural formula:

CAS number.
Palonosetron hydrochloride: 135729-62-3.
Palonosetron (free base): 135729-61-2.7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine (S4).
Summary Table of Changes


 The following uncommon (≥ 0.1% to < 1%) and rare (≥ 0.01% to < 0.1%) adverse reactions were reported as treatment-related adverse events in at least two cancer patients receiving Akynzeo capsules in chemotherapy-induced nausea and vomiting studies (see Table 2):
The following uncommon (≥ 0.1% to < 1%) and rare (≥ 0.01% to < 0.1%) adverse reactions were reported as treatment-related adverse events in at least two cancer patients receiving Akynzeo capsules in chemotherapy-induced nausea and vomiting studies (see Table 2):
 Of the 135 patients who received netupitant 300 mg plus palonosetron 500 microgram, 43% were women, and all patients were White. The age ranged from 19 to 77 years, with a mean age of 53 years. During the study 86% of the 135 treated patients in the netupitant 300 mg plus palonosetron 500 microgram arm received a concomitant chemotherapeutic agent in addition to protocol-mandated cisplatin. The most common chemotherapeutic agents and the proportion of patients exposed were cyclophosphamide (34%), fluorouracil (24%), etoposide (21%) and doxorubicin (16%).
Of the 135 patients who received netupitant 300 mg plus palonosetron 500 microgram, 43% were women, and all patients were White. The age ranged from 19 to 77 years, with a mean age of 53 years. During the study 86% of the 135 treated patients in the netupitant 300 mg plus palonosetron 500 microgram arm received a concomitant chemotherapeutic agent in addition to protocol-mandated cisplatin. The most common chemotherapeutic agents and the proportion of patients exposed were cyclophosphamide (34%), fluorouracil (24%), etoposide (21%) and doxorubicin (16%).
 After completion of cycle 1, patients had the option to participate in a multiple-cycle extension, receiving the same treatment as assigned in cycle 1. There was no pre-specified limit of the number of repeat consecutive cycles for any patient. A total of 1450 patients (Akynzeo n=725; Palonosetron n=725) actually received study medication: of these, 1438 patients (98.8%) completed cycle 1 and 1286 patients (88.4%) continued treatment in the multiple-cycle extension. A total of 907 patients (62.3%) completed the multiple-cycle extension up to a maximum of eight treatment cycles.
After completion of cycle 1, patients had the option to participate in a multiple-cycle extension, receiving the same treatment as assigned in cycle 1. There was no pre-specified limit of the number of repeat consecutive cycles for any patient. A total of 1450 patients (Akynzeo n=725; Palonosetron n=725) actually received study medication: of these, 1438 patients (98.8%) completed cycle 1 and 1286 patients (88.4%) continued treatment in the multiple-cycle extension. A total of 907 patients (62.3%) completed the multiple-cycle extension up to a maximum of eight treatment cycles.

 Fosnetupitant Cmax and AUC were lower in patients than in healthy subjects, although the systemic exposures to netupitant were comparable.
Fosnetupitant Cmax and AUC were lower in patients than in healthy subjects, although the systemic exposures to netupitant were comparable.

