What is in this leaflet
This leaflet answers some common questions about ALBEY VENOM.
It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking ALBEY VENOM against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What is ALBEY VENOM used for
Up to 3% of the population in Australia may have an immediate allergic reaction to an insect sting. This can lead to death in some cases. An average of three deaths per year have been recorded in Australia. Venom immunotherapy can reduce the risk of severe reactions in adults from 30% -70% down to less than 2%. ALBEY VENOM is one type of immunotherapy your doctor may choose to administer to reduce the risk of a severe allergic reaction.
ALBEY VENOM contains freeze dried extracts of venom protein known as allergens.
ALBEY VENOM belongs to a group of medicines called Antiallergy preparations.
Antiallergy preparations can be used for both diagnosis and treatment of potential allergic reactions to insect stings.
The exact mechanism by which ALBEY VENOM does this is not completely understood.
Your doctor, however, may have prescribed ALBEY VENOM for another purpose.
Ask your doctor if you have any questions why ALBEY VENOM has been prescribed for you.
ALBEY VENOM is not addictive.
This medicine is available only with a doctor’s prescription and is usually administered under a doctor’s supervision.
Before you are given ALBEY VENOM
Do not take ALBEY VENOM if:
- If you are hypersensitive (allergic) to any of the other ingredients of Albey
- If in addition to the allergic reaction, you have had any unusual renal, muscular, articular, cutaneous, neurological, hematological reactions after a Honey bee, Yellow Jacket or Wasp sting.
- If you have an illness which affects the immune system
- If you suffer from severe and/or unstable asthma or have experienced severe asthma exacerbation
- Children under the age of 2 years
- If you are pregnant
If you are not sure if you should be taking ALBEY VENOM, talk to your doctor.
Before you start to take it:
Severe local or systemic reactions to venom administration can occur immediately (within one hour) or as delayed reactions. Patients should be kept under direct observation for at least one hour following skin testing and/or therapeutic injections and should be instructed to contact the doctor promptly if symptoms of an allergic reaction or shock occur. Patients should be instructed in the use of, and have available, an emergency anaphylaxis kit for self-administration of adrenaline.
Tell your doctor if:
- you experience severe allergic symptoms.
- If you have had recently an asthma attack or your asthma symptoms get noticeably worse than normal. It may be necessary to postpone the injection to a later date.
- You have cardiovascular and/or bronchopulmonary disease.
- If you suffer from mast cell disorder (mastocytosis) and/or increased serum tryptase
- You are taking a beta blocker (i.e., a class of drugs often prescribed for heart conditions and high blood pressure but also present in some eye drops and ointments), as this drug may decrease the effectiveness of adrenaline, a medicine used to treat serious allergic reactions.
- If you are taking MAOIs, tricyclic antidepressants and COMT inhibitors. Allergen immunotherapy in patients treated with mono amine oxidase inhibitors (MAOIs) or tricyclic antidepressants or for Parkinson's disease with COMT inhibitors should be considered carefully as these treatments could potentiate the effect of adrenaline (epinephrine).
- If you are taking Angiotensin converting enzyme inhibitors (ACE inhibitors) as this treatment may increase the risk of serious reactions to insect stings or hymenoptera venom immunotherapy.
- You have cancer or immunodeficiency or if you are taking medicines which affect your immune system.
- Autoimmune disease in remission.
If you have not told your doctor about any of the above, tell them before you start taking ALBEY VENOM.
Taking other medicines
Tell your doctor if you are taking any other medicines, including any that you bought from a pharmacy, supermarket or health food shop.
In particular tell your doctor if you are taking other allergy medicines such as antihistamines, asthma relief medication or steroids or a medication that blocks a substance called immunoglobulin E (IgE), Medicines for heart or blood pressure (β- blockers and ACE inhibitors), Antidepressants (tricyclics and monoamine oxidase inhibitors (MAOIs)), or if you are being treated with other allergens, or if you recently had or if you plan to have a vaccination soon.
Patients should also avoid high-intensity physical exercise, hot baths, heavy meals and excessive alcohol consumption for several hours after taking medication.
Pregnancy and breast-feeding
It is not known whether ALBEY VENOM is harmful to an unborn baby when taken by a pregnant woman.
Carrying out diagnostic skin tests with ALBEY is not recommended during pregnancy.
Venom allergy treatment with ALBEY must not be initiated during pregnancy.
If you find out that you are pregnant while you are having this treatment, talk to your doctor who will decide with you if you can continue treatment with ALBEY or not.
It is not known whether ALBEY VENOM passes into breast milk. Your doctor will discuss the risks and benefits of using ALBEY VENOM if you are breast-feeding.
Your doctor or pharmacist has more information on medicines to be careful with or avoid while taking ALBEY VENOM.
How is ALBEY VENOM given
Albey venom immunotherapy will be given to you by your doctor.as a subcutaneous injection (under the skin).
Immunotherapy injections should never be given intravenously
How much to take
Your doctor will administer the correct dose as appropriate. You should stay under medical observation for at least 60 min after injection.
The dose administered will depend on the type of therapy decided upon by your doctor.
The dose schedule for children is the same as for adults, and 100 microgram per venom should still be considered the maintenance dose. Because of the smaller size of the child, the larger volumes of solution may produce excessive discomfort. Therefore, in order to achieve the total dose required, the volume of the dose may need to be divided into more than one injection per visit.
How it is it given
The doctor will administer ALBEY VENOM via a subcutaneous injection (under the skin). The treatment will generally last over several weeks, with the doctor increasing the dose until a maintenance dose is reached.
When to take it
Your doctor will advise you on when it is appropriate to be given ALBEY VENOM.
How long to take it
Your doctor will decide the length of treatment. The longer the course of treatment the better the therapeutic effect of the drug. Therefore, the course of treatment may last from three to five years. Once a maintenance dose is reached the frequency of administration should decrease.
While you are being given ALBEY VENOM
Tell all doctors, dentists and pharmacists who are treating you that you are taking ALBEY VENOM.
Tell your doctor if you become pregnant while taking ALBEY VENOM.
Be sure to keep all of your appointments with your doctor so that your progress can be checked.
Things you must not do
Do not take any other medicines, whether they require a prescription or not, without first telling your doctor or consulting a pharmacist.
Things to be careful of
ALBEY VENOM is not expected to affect your ability to drive a car or operate machinery.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking ALBEY VENOM.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of these side effects.
ALBEY VENOM generally causes few side effects and helps most people with hypersensitivity to insect stings, however it may have unwanted side effects in a few people.
- Local reactions at the site of injection in the form of a weal or swelling occur frequently and are not cause for alarm, but if they persist, are indication that dosage may need adjustment.
- Allergic reactions that appear at the injection and/or symptoms in the whole body. Cases of anaphylactic shock that cause a fall in blood pressure and suddenly feeling generally unwell, and that require emergency treatment may occur. Anaphylactic shock is a severe allergic reaction with rapid onset of symptoms which affect the whole body such as intensive itching or a skin rash, breathing difficulty, abdominal pain, or symptoms related to drop in blood pressure such as dizziness or malaise. Contact your doctor immediately if you get any of these side effects.
- If this happens to you, your doctor will have an emergency kit available with a 'ready for use' adrenaline syringe. This is the reason why you are kept under medical supervision for at least one hour minutes after having your injection.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor immediately or go to Accident and Emergency at your nearest hospital if you notice any of the following:
- Local reactions at the injection site that may be accompanied by swelling, painitching, redness, induration
- Rapid heartbeat
- Dizziness
- Hypotension (low blood pressure)
- Systemic manifestations (reactions of the whole body): anaphylactic reaction, hypersensitivity, chest discomfort, chest pain, feeling hot
- General or local swelling, general malaise, feeling of extreme tiredness
- Respiratory problems: asthmatic crisis, difficulty of breathing, cough, blocked nose, swelling in the throat, throat irritation, throat tightness, wheezing
- Arthralgia (pain in knuckles)
- Diarrhea, abdominal pain, stomach pain, nausea
- Face swelling, hives, itching, redness
This is not a complete list of all possible side effects. Others may occur in some people and there may be some side effects not yet known.
Tell your doctor if you notice anything else that is making you feel unwell, even if it is not on this list.
Ask your doctor or pharmacist if you don’t understand anything in this list.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Storage
Your medicine will generally be stored by your doctor. It should be kept refrigerated between 2 to 8 deg. C. It should not be frozen.
At the time of reconstitution, your doctor will record the date of reconstitution and expiration date of reconstituted product in the space provided (day, month, year) on the product label and store the reconstituted vial in the refrigerator at between 2 to 8 deg. C.
Expiration date of the reconstituted venom depends on the type of reconstituting fluid used. Products reconstituted in Albumin-Saline have an expiration date of 6 months from date of reconstitution. Date of expiration after reconstitution must not exceed Final Expiration Date indicated on the container label
Syringes and vials should be disposed of appropriately.
Product Description
Availability
These freeze-dried products should be dissolved in Albumin-Saline (which contains sodium chloride 0.9%, phenol 0.4% and normal human serum albumin 0.03%) to a concentration of 100 microgram/mL. Dilutions of this concentration should be made only with Albumin-Saline.
Mannitol and Sodium chloride are included as inactive components in Bee Venom.
Mannitol is included as an inactive component in Paper Wasp and Yellow Jacket Venom.
ALBEY VENOM is available as:
Albey Honey Bee Venom Injection
Honey bee (Apis mellifera) venom; freeze dried powder for reconstitution. To be diluted with Albumin-Saline diluent.
Pack - 550 micrograms per vial.
Albey Wasp Venom Injection (Injection)
Wasp venom (Polistes sp.); freeze dried powder for reconstitution. To be diluted with Albumin-Saline diluent.
Pack - 550 micrograms per vial.
Albey Yellow Jacket Venom Injection (Injection)
Vespula spp. freeze dried powder for reconstitution. To be diluted with Albumin-Saline diluent.
Pack - 550 micrograms per vial.
Albumin-Saline Diluent
To be used only for the dilution of the above Albey Venoms.
The albumin-saline diluent is available in cartons consisting of the following pack sizes:
- 20 vials of 9.0 mL
- 5 vials of 1.8 mL.
Not all pack sizes may be marketed.
AUST R: 161298 (Albey Bee Venom)
AUST R: 18770 (Albey Yellow Jacket Venom)
AUST R: 18695 (Albey Paper Wasp Venom)
AUST R: 32486 (Albumin-Saline Diluent)
NAME AND ADDRESS OF THE SPONSOR IN AUSTRALIA:
Stallergenes Australia Pty Ltd
Suite 2408,
4 Daydream St.,
Warriewood, NSW 2102
Ph: 1800 824 166
NAME AND ADDRESS OF THE SPONSOR IN NEW ZEALAND:
Stallergenes Greer New Zealand Limited
Level 1, 24 Manukau Road,
Epsom, Auckland 1023
New Zealand
Ph: 0800 824 166
Date prepared: 24 June 2024
Published by MIMS September 2024



 Examples of the preceding dilution table are shown in Table 3.
Examples of the preceding dilution table are shown in Table 3.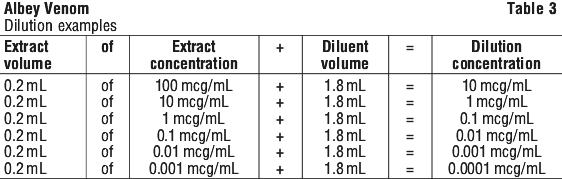 Reconstitute and dilute the freeze dried venom as directed previously.
Reconstitute and dilute the freeze dried venom as directed previously.


