What is in this leaflet
Read this leaflet carefully before you start to use ALCAINE Eye Drops.
This leaflet answers some common questions about ALCAINE. It does not contain all of the available information. It does not take the place of talking to your doctor or pharmacist.
The information in this leaflet was last updated on the date listed on the final page. More recent information on the medicine may be available.
You should ensure that you speak to your pharmacist or doctor to obtain the most up to date information on the medicine.
You can also download the most up to date leaflet from www.ebs.tga.gov.au
The updates may contain important information about the medicine and its use of which you should be aware.
All medicines have risks and benefits. Your doctor has weighed the expected benefits of you using ALCAINE against the risks this medicine could have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What ALCAINE is used for
ALCAINE contains the active ingredient proxymetacaine hydrochloride, which belongs to a group of medicines called local anaesthetics.
ALCAINE Eye Drops are only to be used by your doctor during ophthalmic procedures.
ALCAINE is used to temporarily numb or block the feeling (anaesthetise) of pain in your eye(s) during short term medical procedures. These procedures include measuring the pressure in your eye, cataract surgery, removing stitches or a foreign body from eye e.g. a bit of dirt.
The numbing effect of ALCAINE will wear off quickly, usually about 15 minutes after the last drop is put in your eye(s).
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
Before you are given ALCAINE
When you must not be given it
ALCAINE should not be given to you if you have an allergy to:
- Any medicine containing proxymetacaine hydrochloride
- Any other local anaesthetics
- Any of the ingredients in ALCAINE (these are listed under Product Description).
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Do not use this medicine after the expiry date printed on the pack or if the seal around the pack is broken or shows signs of tampering. If it is has expired or is damaged, return it to your pharmacist for disposal.
This medicine has been developed for use in the eye only. It is not to be swallowed or injected.
If you are not sure whether you should be given this medicine talk to your doctor.
Before you are given it
Tell your doctor if you have allergies to any other medicines or any substances such as foods, preservatives or dyes.
Tell your doctor if you have or have had any medical conditions, especially the following:
- Epilepsy
- Heart problems
- Lung or breathing problems
- Thyroid problems
- Myasthenia gravis (a disease of the muscles causing drooping eyelids, double vision, difficulty in speaking and swallowing and sometimes muscle weakness in the arms or legs)
Tell your doctor if you have ever experienced a reaction to any other local anaesthetics.
Tell your doctor if you are pregnant or plan to become pregnant
Tell your doctor if you are breast feeding. Your doctor will discuss the risks and benefits involved.
If you have not told your doctor about any of the above, tell them before you are given ALCAINE.
Long term use may lessen the anaesthetic effect so that more of the medicine is needed to produce anaesthesia.
Long term use may also damage the surface of the eye (cornea), cause an eye infection or permanent loss of sight.
It is very important that you protect your eyes from irritating chemicals, avoid touching or rubbing your eye(s) until the anaesthesia has worn off.
Taking or using other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including medicines that you buy at a pharmacy or health food shop without a doctor’s prescription.
Some medicines and ALCAINE may interfere with each other. These include a group of medicines called "cholinesterase inhibitors".
Your doctor will advise you and decide whether or not to give you ALCAINE.
How ALCAINE is given
How much is used
Your doctor will put the correct number of drops of ALCAINE into your eye(s) before starting the procedure on your eye.
Your doctor will not write a prescription for ALCAINE for your own use. ALCAINE has been specifically developed to help a doctor numb the feeling in your eye(s) while attending to your eye(s).
The usual dose is one to two drops into your eye(s) before starting the procedure on your eye. The number of times you will be given ALCAINE during the procedure will depend on the type and length of procedure to be performed. For example, your doctor will put one to two drops into your eye(s) just before measuring the pressure in your eye. However, if you are to have minor surgery such as removal of stitches your doctor will put one to two drops in your eye(s) every 5 to 10 minutes for up to three doses.
It is very important that you do not touch or rub your eye while your eye is numb because you may scratch the surface of your eye and damage it. Your doctor will put a patch over your eye after finishing the procedure to protect your eye and stop you from touching it.
If you are wearing soft contact lenses, you will be asked to remove them prior to instillation of the drops by your doctor. You may put your soft contact lenses back into your eyes after the anaesthesia has worn off and at least 15 minutes after you have been given ALCAINE
If you are given too much (overdose)
The doctor giving you ALCAINE will be experienced in the use of local anaesthetics, so it is unlikely that you will be given an overdose.
If too many drops have been put in accidentally in your eye(s) immediately rinse your eye(s) with warm water.
If you think that you or someone else has swallowed too much ALCAINE, immediately telephone the Poisons Information Centre (in Australia call 13 11 26; in New Zealand call 0800 POISON or 0800 764 766), your doctor or go to Accident and Emergency at the nearest hospital. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
After you are given ALCAINE
Things you must do
You must protect you anaesthetised eye from dust particles, which could cause infection.
Things you must not do
Do not touch or rub your eye(s) while they are numb. You may scratch the surface of the eye and damage it. Your doctor will put a patch over your eye after finishing the procedure to protect your eye and stop you from touching it.
Things to be careful of
Be careful driving or operating machinery until you know how ALCAINE affects you and your vision. As with any eye medicines, temporary blurred vision or other visual disturbances may affect the ability to drive or use machinery in some people. If blurred vision occurs, wait until your vision is clear before driving or operating machinery.
Side Effects
Tell your doctor as soon as possible if you do not feel well after ALCAINE use.
All medicines can have side effects. Sometimes they are serious, however most of the time they are not. You may need medical treatment if you get some of the side effects.
Do not feel alarmed at the list of possible side effects. You may not experience any of them.
Ask your doctor, nurse, or pharmacist to answer any questions you may have.
Tell your doctor, nurse or pharmacist if you notice any of the following and they worry you:
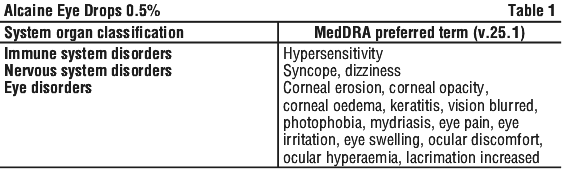
- Irritation, stinging and burning feeling in the eye
- Redness or watery eyes
- Increased winking
- Dilated pupils causing blurred vision
- Drying and splitting of the skin on fingertips
- A severe, immediate allergic reaction involving the front layer (the cornea) of the eye.
Occasionally some people notice untoward effects in the rest of their body as a result of ALCAINE use.
Tell your doctor immediately or go to Accident and Emergency at your nearest hospital at the first sign of an allergic reaction such skin rash or hives. You may need urgent medical attention or hospitalisation.
Tell your doctor if you notice any unwanted effects while you were given ALCAINE, even if they do not appear in the list above.
Other side effects not listed above may also occur in some patients.
After using ALCAINE
Storage
ALCAINE will be stored by your doctor or pharmacist under the recommended conditions. It should be stored between 2°C to 8°C (Refrigerate, do not freeze). Protect from light.
ALCAINE should be used in a single patient only.
Product Description
What it looks like
ALCAINE Eye Drops are a sterile, clear liquid that comes in a 15 mL dropper bottle.
Ingredients
ALCAINE Eye Drops contain proxymetacaine hydrochloride 0.5% (5 mg/mL) as the active ingredient.
ALCAINE Eye Drops also contain:
- glycerol
- benzalkonium chloride (as a preservative)
- purified water
- hydrochloric acid and/or sodium hydroxide (to adjust pH).
Supplier
ALCAINE is supplied in Australia by:
Alcon Laboratories (Australia) Pty Ltd
ABN 88 000 740 83054
15 Talavera Road
Macquarie Park NSW 2113
Telephone: 1800 224 153
Australian registration number
AUST R No. 24299.
Date of Preparation
This leaflet was prepared in May 2023.
Internal document code (alc030523c)
Published by MIMS June 2023

 Additionally, overuse, uncontrolled use or abuse of the product may lead to ocular lesions due to the toxic effects of the anaesthetic to the epithelium.
Additionally, overuse, uncontrolled use or abuse of the product may lead to ocular lesions due to the toxic effects of the anaesthetic to the epithelium.
