What is in this leaflet
This leaflet answers some common questions about ALLMERCAP®. It does not contain all of the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking ALLMERCAP® against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What ALLMERCAP® is used for
ALLMERCAP® contains mercaptopurine monohydrate as the active ingredient. It belongs to a group of medicines called cytotoxics (also called chemotherapy).
ALLMERCAP® is used solely or in combination with other medicines to treat acute leukaemia, a cancer of certain blood cells.
It works by interfering with the growth of cancer cells.
Ask your doctor if you have any questions about why ALLMERCAP® has been prescribed for you. Your doctor may have prescribed it for another purpose. This medicine is only available with a doctor's prescription.
There is no evidence that it is addictive.
Before you take it
When you must not take it
Do not take ALLMERCAP® if you have ever had an allergic reaction to:
- Mercaptopurine monohydrate
- any of the oral liquid suspension ingredients listed at the end of this leaflet.
Symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty in breathing; swelling of the face, lips, tongue or any other parts of the body; rash, itching or hives on the skin.
Do not receive the yellow fever vaccine whilst you are taking ALLMERCAP®.
Do not take ALLMERCAP® if you are planning to become pregnant or father a child unless you and your doctor have discussed the risks and benefits involved. As with all cytotoxic drugs, ALLMERCAP® may harm eggs and sperm. Reliable contraceptive methods must be taken to avoid pregnancy whilst you or your partner is taking this medicine.
Do not take ALLMERCAP® if you are pregnant unless you and your doctor have discussed the risks and benefits involved. It may affect your developing baby if you take it during pregnancy.
Do not take this medicine whilst breast feeding. It is not recommended for use while breast feeding as it is found in breast milk.
Do not take ALLMERCAP® after the expiry date printed on the bottle. If you take it after the expiry (EXP) date has passed, it may not work as well.
Do not take it if the packaging is torn or shows signs of tampering.
ALLMERCAP® contains aspartame (E951), hydroxybenzoates and sorbates.
Aspartame is a source of phenylalanine. It may be harmful if you have phenylketonuria (PKU), a rare genetic disorder in which phenylalanine builds up because the body cannot remove it properly.
Before you start to take it
Tell your doctor if you are allergic to any other medicines or any other foods, dyes or preservatives.
Tell your doctor if you have received the yellow fever vaccine.
Tell your doctor if you have or have had any of the following conditions:
- you have recently received or are receiving radiotherapy or chemotherapy
- you have recently been vaccinated or are planning to be vaccinated
- chickenpox, shingles or hepatitis B (a liver disease caused by a virus)
- kidney or liver disease
- a condition where your body produces too little of a natural chemical called thiopurine methyltransferase (TPMT).
- inflammatory bowel disease – some patients with inflammatory bowel disease who have received mercaptopurine monohydrate have developed a rare and aggressive type of cancer called Hepatosplenic T-cell Lymphoma
- an inherited mutation in the NUDT15 gene (a gene which is involved in the break down of Allmercap in the body) – you may have a higher risk of infections and hair loss and your doctor may give you a lower dose.
- Lesch-Nyhan Syndrome, a rare inherited condition where there is a deficiency of the hypoxanthine-guanine-phosphoribosyltransferase enzyme.
If you have not told your doctor about any of the above, tell them before you start taking ALLMERCAP®.
Taking other medicines
Tell your doctor if you are taking any other medicines, including any that you buy without a prescription from a pharmacy, supermarket or health food shop.
Some medicines may be affected by ALLMERCAP® or may affect how well it works. You may need to take different amounts of your medicine or you may need to take different medicines. These include:
- allopurinol, oxipurinol and/or thiopurinol
- methotrexate
- infliximab
- medicines to treat or prevent blood clots e.g. warfarin
- 6-thioguanine
- aminosalicylate derivatives such as olsalazine, mesalazine or sulphasalazine
- vaccinations with 'live' organism vaccines
- Other medicines used to suppress the body’s immune defence system agents
- ribavirin
- phenytoin
- febuxostat
Your doctor or pharmacist has more information on medicines to be careful with or avoid while taking this medicine.
How to take it
How much to take
Take ALLMERCAP® exactly as directed by your doctor.
Your doctor will decide what dose and for how long you will be taking ALLMERCAP®. This depends on factors such as:
- your age and weight
- any pre-existing conditions such as kidney or liver disease
- your response to the treatment
- other medicines taken in combination with ALLMERCAP®.
For children the usual dose is 2.5 mg/kg bodyweight/day but your dose will be carefully adjusted by your doctor depending on your condition. Your doctor may change the dose and frequency of your medicine as your condition changes. Your doctor may order regular blood cell count, liver function and urine tests while you are taking ALLMERCAP® in order to monitor your condition and to change your dose if necessary.
How to take it
It is important to take ALLMERCAP® in the evening to make the medicine more effective.
You can take your medicine with food or on an empty stomach but the choice of method should be consistent from day to day. You should take your medicine at least 1 hour before or 2 hours after having milk or dairy products.
Your pack of ALLMERCAP® contains a bottle of medicine, a cap, a bottle adaptor and two dosing syringes (a purple 1 mL syringe and an orange 5 mL syringe). Always use the syringes provided to take your medicine. The syringes provided must only be used with ALLMERCAP®.
It is important that you use the correct dosing syringe for your medicine. Your doctor or pharmacist will advise which syringe to use depending on the dose that has been prescribed.
The smaller 1 mL syringe (purple), marked from 0.1 mL to 1 mL, is for measuring doses of less than or equal to 1 mL. You should use this one if the total amount you have to take is less than or equal to 1 mL (each graduation of 0.1 mL contains 2 mg of mercaptopurine monohydrate).
The larger 5 mL syringe (orange), marked 1 mL to 5 mL, is for measuring doses of more than 1 mL. You should use this one if the total amount you have to take is more than 1 mL (each graduation of 0.2 mL contains 4 mg of mercaptopurine monohydrate).
If you are a parent or care giver administering the medicine, wash your hands before and after administering a dose. Wipe up spillages immediately. To decrease the risk of exposure disposable gloves should be used when handling ALLMERCAP®.
If ALLMERCAP® comes into contact with skin, eyes or nose, it should be washed immediately and thoroughly with soap and water.
When you use the medicine follow the instructions below:

- Put on disposable hand gloves before handling ALLMERCAP®.
- Shake the bottle vigorously for at least 30 seconds to ensure the medicine is well mixed (figure 1).
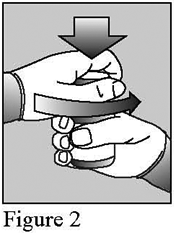
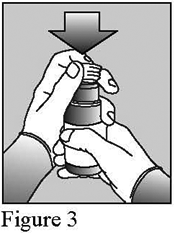
- Remove the bottle cap (figure 2) and push the adaptor firmly into the top of the bottle and leave in place for future doses (figure 3).
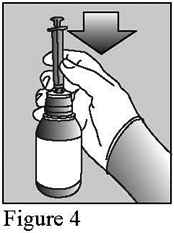
- Push the tip of the dosing syringe into the hole in the adaptor (figure 4). Your doctor or pharmacist will advise you of the correct syringe to use, either the 1 mL (purple syringe) or the 5 mL (orange syringe) in order to give the correct dose.
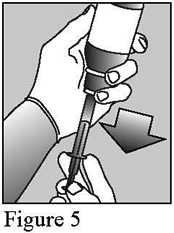
- Turn the bottle upside down (figure 5).
- Pull the plunger of the syringe back so that the medicine is drawn from the bottle into the syringe. Pull the plunger back to the point on the scale that corresponds to the dose prescribed (figure 5). If you are not sure about how much medicine to draw into the syringe, always ask your doctor or nurse for advice.
- Turn the bottle back the right way up and carefully remove the syringe from the adaptor, holding it by the barrel rather than the plunger.
- Gently put the tip of the syringe into your mouth and to the inside of your cheek.
- Slowly and gently push the plunger down to gently squirt the medicine into the inside of your cheek and swallow it. DO NOT forcefully push down the plunger, or squirt the medicine to the back of your mouth or throat, as you may choke.
- Remove the syringe from your mouth.
- Swallow the dose of oral liquid suspension then drink some water, making sure no medicine is left in your mouth.
- Put the cap back on the bottle with the adaptor left in place. Ensure that the cap is tightly closed.
- Wash the syringe with warm ‘soapy’ water and rinse well. Hold the syringe under water and move the plunger up and down several times to make sure the inside of the syringe is clean. Let the syringe dry completely before you use that syringe again for dosing. Store the syringe in a hygienic place with the medicine.
Repeat the above for each dose as instructed by your doctor or pharmacist.
Like all cytotoxic drugs, ALLMERCAP® is irritant to the eyes and skin. To prevent irritation it is important to wash your hands immediately after handling the oral liquid suspension, to avoid contact with the eyes.
How long to take it
Your doctor will tell you how long to take ALLMERCAP® for. Do not stop taking it or change the dose without first checking with your doctor.
After first opening of the bottle, discard any unused contents after 8 weeks (56 days). Write the date of first opening on the bottle to help you remember.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (in Australia telephone 131126, in New Zealand telephone 0800 764 766, in Hong Kong telephone your doctor or the nearest hospital) for advice, if you think you or anyone else may have taken too much ALLMERCAP®, even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are taking it
Things you must do
Tell your doctor if, for any reason, you have not taken your medicine exactly as directed. Otherwise, your doctor may think that it was not working as it should and change your treatment unnecessarily.
Do not take a double dose to make up for the one you have missed. Visit your doctor regularly so they can check your progress and make sure your medicine is working.
Tell any other specialist, doctor, dentist or pharmacist that you are on ALLMERCAP®, especially if you are about to be started on any new medicines, immunisations, vaccinations or radiotherapy.
Tell your doctor if you become pregnant, are trying to become pregnant or trying to father a child.
Use a sunscreen with a high SPF and protective clothing and limit exposure to sunlight and UV light.
Things you must not do
Do not give this medicine to anyone else, even if their symptoms seem similar to yours.
Do not use ALLMERCAP® to treat any other complaints unless your doctor says to.
Things to be careful of
Be careful driving or operating machinery until you know how ALLMERCAP® affects you.
Do not have any vaccinations without your doctor's approval. ALLMERCAP® can lower the number of white blood cells and platelets in your blood. This means that you have an increased chance of getting an infection or bleeding. The risk of viral, fungal and bacterial infections is increased and the infections may be more serious.
Side effects
Tell your doctor as soon as possible if you do not feel well while you are taking ALLMERCAP®
All medicines can cause some side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
The following side effects have been reported with ALLMERCAP®
- production of bone marrow cells may be reduced. You may notice an increase in infections. Your doctor will do regular blood tests, but you should tell them at once if you notice any signs of fever or infection or any unexpected bruising, bleeding or signs of blood in your urine
- increased risk of liver disease or damage
- in men, sperm production may be reduced
- as with all cytotoxic medicines, there is an increased risk of some cancers
- as with all cytotoxic medicines, there is an increased risk of damage to the genes in some cells.
Tell your doctor if you notice any of the following:
- nausea and vomiting
- diarrhoea
- infections such as fever, severe chills, sore throat or mouth ulcers
- bruising or bleeding more easily than normal
- loss of appetite and/or weight loss
- jaundice, a yellowing of the whites of the eyes or the skin
- a condition in which there is a decreased number of red blood cells, called anaemia. Symptoms include tiredness, headaches, shortness of breath, dizziness and looking pale.
- painful, swollen joints
- skin rash
- hair loss.
If you get any of the following side effects, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- wheezing
- swelling of the lips/mouth
- difficulty in breathing
- hayfever
- lumpy rash (hives)
- fainting
- sudden abdominal pain with nausea and vomiting.
Tell your doctor if you notice anything else that is making you feel unwell, even if you think the problems are not connected with this medicine and are not referred to in this leaflet. Other side effects not listed above may also occur in some people.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
After taking it
Storage
Keep it in a cool, dry place, protected from light, where the temperature stays below 25°C.
Do not leave it in a car, on window sills, near a sink or in the bathroom. Heat and dampness can affect some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
After first opening of the bottle, discard any unused contents after 8 weeks (56 days). Write the date of first opening on the bottle to help you remember.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
Product description
What it looks like
ALLMERCAP® is a pink to brown oral liquid suspension. It comes in bottles of 100 mL capped with a child-resistant closure. Each pack contains one bottle, a bottle adaptor and two dosing syringes (a purple syringe graduated to 1 mL and an orange syringe graduated to 5 mL). Your doctor or pharmacist will advise which syringe to use depending on the dose that has been prescribed.
Ingredients
Active ingredient:
One mL of suspension contains 20 mg of mercaptopurine monohydrate.
Inactive ingredients:
- xanthan gum
- aspartame (E951)
- concentrated raspberry juice
- sodium methylhydroxybenzoate (E219)
- sodium ethylhydroxybenzoate (E215)
- potassium sorbate (E202)
- sodium hydroxide
- purified water.
Sponsor
In Australia:
Link Medical Products Pty Ltd
5 Apollo Street,
Warriewood NSW 2012
Australia
Ph: 1800 181 060
linkhealthcare.com.au
In New Zealand:
Link Pharmaceuticals Ltd
Suite 32, Level 26, PwC Tower
188 Quay Street
Auckland 1010
New Zealand
In Hong Kong:
Unit 16, Floor 12
Corporation Square
8 Lam Lok Street
Kowloon Bay
Hong Kong
Australian Registration Number: AUST R 213881
This leaflet was prepared in January 2021.
Published by MIMS March 2021


