What is in this leaflet
This leaflet answers some common questions about Alodorm.
It does not contain all of the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have benefits and risks. Your doctor has weighed the risks of you taking Alodorm against the benefits expected for you.
If you have any concerns about taking this medicine, talk to your doctor or pharmacist.
Keep this leaflet with your medicine. You may need to read it again.
What Alodorm is used for
Alodorm is used to treat insomnia (sleeping problems).
Alodorm contains the active ingredient nitrazepam, which belongs to a group of medicines called benzodiazepines. These medicines are thought to work by their action on brain chemicals.
Ask your doctor if you have any questions about why Alodorm has been prescribed for you. Your doctor may have prescribed Alodorm for another reason.
In general, benzodiazepines such as Alodorm should be taken for short periods only (for example 2 to 4 weeks). Continuous long-term use is not recommended unless advised by your doctor. The use of benzodiazepines may lead to dependence on the medicine.
Alodorm is available only with a doctor's prescription.
Before you take Alodorm
When you must not take it
Do not take Alodorm if you are allergic to:
- nitrazepam
- any other benzodiazepine medicines such as diazepam, temazepam, oxazepam, alprazolam and clonazepam
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include skin rash, itching or hives; swelling of the face, lips or tongue which may cause difficulty in swallowing or breathing; wheezing or shortness of breath.
Do not take Alodorm if you have:
- severe and chronic lung disease (e.g. chronic obstructive airway disease) and difficulty breathing
- severe liver disease.
Do not take Alodorm after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
Do not give Alodorm to a child unless advised by the child's doctor. The safety and effectiveness of this medicine in children have not been established.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you are allergic to any other medicines, foods, dyes or preservatives.
Tell your doctor if you are pregnant or plan to become pregnant. Like other benzodiazepine medicines, Alodorm may cause unwanted effects in the newborn baby if used during pregnancy. Your doctor will discuss the risks and benefits of taking Alodorm during pregnancy.
Tell your doctor if you are breastfeeding or wish to breastfeed. Alodorm passes into breast milk and may cause drowsiness and/or feeding problems in the baby. Your doctor will discuss the risks and benefits of taking Alodorm when breastfeeding.
Tell your doctor if you have any medical conditions, especially the following:
- liver, kidney or lung disease
- high or low blood pressure
- glaucoma (increased pressure in the eye)
- myasthenia gravis, a condition where there is severe muscle weakness
- depression, psychosis or schizophrenia
- epilepsy, fits or convulsions.
Your doctor may want to take special care if you have any of these conditions.
Tell your doctor if you drink alcohol regularly. Alcohol may increase the effects of Alodorm.
Tell your doctor if you have a history of fall or are unsteady when walking.
Tell your doctor if you plan to have surgery.
If you have not told your doctor about any of the above, tell them before you start taking Alodorm.
Taking other medicines
Tell your doctor if you are taking any other medicines, including any that you buy without a prescription from a pharmacy, supermarket or health food shop.
Some medicines may be affected by Alodorm, or may affect how well it works. These include:
- other sleeping tablets, sedatives or tranquillisers
- medicines for depression
- medicines used to treat epilepsy
- antihistamines, medicines for allergies, colds or travel sickness
- muscle relaxants
- pain relievers such as opioid medicines
- cimetidine, a medicine used to treat reflux and stomach ulcers
- disulfiram, a medicine used to deter alcohol consumption.
These medicines may increase the effects of Alodorm or be affected by it. You may need to take different amounts of your medicine or you may need to take different medicines.
Your doctor can tell you what to do if you are taking any of these medicines.
If you are not sure whether you are taking any of these medicines, check with your doctor or pharmacist. Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking Alodorm.
How to take Alodorm
Follow all directions given to you by your doctor and pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the label, ask your doctor or pharmacist.
How much to take
The dose may vary from person to person.
Your doctor will tell you how many tablets you need to take each day.
The usual adult dose is 5 mg to 10 mg (one to two tablets) at bedtime.
For the elderly, the recommended dose is 2.5 mg to 5 mg (half to one tablet) at bedtime.
Alodorm is not recommended for children.
How to take it
Swallow the tablets with a glass of water.
If you forget to take it
If you forget to take Alodorm before you go to bed and you wake up late in the night or early in the morning, do not take Alodorm as you may have trouble waking in the morning.
If you have any questions about this, check with your doctor or pharmacist.
How long to take it
Take Alodorm only for as long as your doctor recommends.
Usually, Alodorm should be taken for short periods only (for example 2 to 4 weeks). Continuous long-term use is not recommended unless advised by your doctor. The use of benzodiazepines may lead to dependence on the medicine.
If you take too much (overdose)
Immediately telephone your doctor, or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital, if you think you or anyone else may have taken too much Alodorm.
Do this even if there are no signs of discomfort or poisoning. Also report if any other medicine or alcohol has been taken. You may need urgent medical attention.
If you take too much Alodorm, the mild symptoms are drowsiness, mental confusion and lethargy. In more serious cases, symptoms are inability to move, loss of muscle tone, hypotension, breathing difficulties, coma and very rarely death.
While you are taking Alodorm
Things you must do
Take Alodorm exactly as your doctor has prescribed.
Tell all the doctors, dentists and pharmacists who are treating you that you are taking Alodorm.
If you plan to have surgery that requires a general anaesthetic, tell your doctor or dentist that you are taking Alodorm.
Before starting any new medicine, tell your doctor or pharmacist that you are taking Alodorm.
If you become pregnant while taking Alodorm, tell your doctor immediately.
Visit your doctor regularly. Your doctor needs to check your progress to see whether you need to keep taking Alodorm. You may also need to have tests to check your blood and liver function.
Tell your doctor if you feel Alodorm is not helping your condition.
Things you must not do
Do not drive or operate machinery until you know how Alodorm affects you. Alodorm may cause drowsiness or dizziness in some people. Even though you take Alodorm at night, you may still be drowsy or dizzy the next day. Make sure you know how you react to Alodorm before you drive a car, operate machinery or do anything else that could be dangerous. This is very important if you are taking other drugs that also make you drowsy.
Do not take Alodorm for a longer time than your doctor has prescribed. Alodorm should be taken for short periods only (for example 2 to 4 weeks) unless advised otherwise by your doctor.
Do not stop taking Alodorm, or change the dose, without checking with your doctor. Stopping Alodorm suddenly may cause some unwanted effects. Your doctor may want you to gradually reduce the amount of Alodorm you are taking before stopping completely.
Do not suddenly stop taking Alodorm if you suffer from epilepsy. Stopping this medicine suddenly may make your epilepsy worse.
Do not use Alodorm to treat any other conditions unless your doctor tells you to.
Do not give Alodorm to anyone else, even if they have the same condition as you.
Things to be careful of
Some sleep medicines may cause short-term memory loss. When this occurs, a person may not remember what has happened for several hours after taking the medicine. This is usually not a problem since most people fall asleep after taking the medicine. To reduce this risk, ensure that you are able to get a full night's sleep (7 to 8 hours) before you need to be active again.
Be careful drinking alcohol while taking Alodorm. Combining alcohol with Alodorm can make you more drowsy, dizzy or lightheaded, or increase the risk of sleep-walking and some other related sleep behaviours, which may include sleep-driving, making phone calls or preparing and eating food whilst asleep.
This risk is also increased if you take more than the recommended dose.
Your doctor may suggest that you avoid alcohol or reduce the amount of alcohol you drink while you are taking Alodorm.
You should not take Alodorm if you experience complex sleep behaviours such as sleep walking, sleep driving or any other bizarre sleep-related behaviours.
Be careful if you are elderly, unwell or taking other medicines. You may have an increased chance of getting side effects such as drowsiness, confusion, dizziness and unsteadiness, which may increase the risk of a fall.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Alodorm. Alodorm helps most people with insomnia and is usually well tolerated, but it may have unwanted side effects in some people.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Tell your doctor if you notice any of the following and they worry you:
- drowsiness
- dizziness
- fatigue
- confusion
- unsteadiness when walking
- impairment of memory
- headache
- hangover feeling in the morning
- slurred speech
- clumsiness, lack of coordination
- numbed emotions
- reduced alertness
- muscle weakness
- double vision
- inattention
- unpleasant dreams
- reoccurrence of insomnia
The above list includes the milder side effects of your medicine.
Tell your doctor immediately or go to Accident and Emergency at the nearest hospital if you notice any of the following:
- swelling of the tongue or throat
- difficulty in breathing.
The side effects listed above are very serious and require urgent medical attention or hospitalisation.
Like other medicines, Alodorm can cause some side effects. If they occur, they are most likely to be minor and temporary. However, some may be serious, such as complex sleep behaviours, and need medical attention.
Tell your doctor if you notice any unusual changes in your sleep behaviour.
Tell your doctor if you notice anything that is making you feel unwell when you are taking, or soon after you have finished taking Alodorm.
Other side effects not listed above may also occur in some people.
After using Alodorm
Storage
Keep Alodorm where children cannot reach it.
Keep your tablets in the bottle until it is time to take them. If you take the tablets out of the bottle they may not keep well.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Keep your tablets in a cool dry place where the temperature stays below 25°C. Protect from light.
Do not store Alodorm or any other medicine in the bathroom or near a sink.
Do not leave Alodorm in the car or on window sills. Heat and dampness can destroy some medicines.
Disposal
If your doctor tells you to stop taking Alodorm, or your tablets have passed their expiry date, ask your pharmacist what to do with any that are left over.
Product description
What it looks like
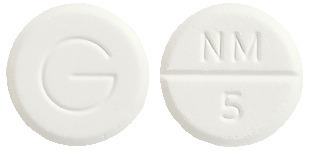
Alodorm is a round white tablet marked NM/5 on one side and G on the other.
Each bottle contains 25 tablets.
Ingredients
The active ingredient in Alodorm is nitrazepam. Each Alodorm tablet contains 5 mg of nitrazepam.
The tablets also contain the following inactive ingredients:
- lactose monohydrate
- maize starch
- pregelatinised maize starch
- purified talc
- colloidal anhydrous silica
- magnesium stearate.
Alodorm also contains sugars (as lactose), traces of galactose and sulfites. The tablets are gluten free.
Manufacturer
Alodorm is made in Australia by:
Alphapharm Pty Ltd
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point NSW 2000
www.mylan.com.au
Australian registration number:
AUST R 54009
This leaflet was prepared in
December 2020.
Alodorm_cmi\Dec20/00
Published by MIMS February 2021

 Molecular formula: C15H11N3O3.
Molecular formula: C15H11N3O3.