What is in this leaflet
This leaflet answers some common questions about ALVESCO.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
The information in this leaflet was last updated on the date listed on the final page. More recent information on the medicine may be available. You should ensure that you speak to your pharmacist or doctor to obtain the most up to date information on this medicine.
Those updates may contain important information about the medicine and its use of which you should be aware.
All medicines have risks and benefits. Your doctor has weighed the risks of you using this medicine against the benefits he/she expects it will have for you.
If you have any concerns about using this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What ALVESCO is used for
ALVESCO is inhaled into the lungs for the treatment of asthma. Asthma is a disease where the lining of the lungs becomes inflamed (red and swollen), making it difficult to breathe.
ALVESCO contains a medicine called ciclesonide, which belongs to a family of medicines called corticosteroids.
ALVESCO metered dose inhalers deliver a measured amount of ciclesonide, which acts directly on your air passages to reduce inflammation and help keep the air passages open. In doing so, this medication helps to improve your condition and to prevent asthma attacks from occurring. It is a preventer and needs to be taken regularly.
ALVESCO will NOT give you immediate relief of wheezing or breathlessness during sudden, severe or continuous asthma attacks. In this situation you should use a quick acting reliever inhaler, which contains a different medicine, a so-called bronchodilator.
Your doctor may prescribe ALVESCO to replace corticosteroid tablets, which will mean that you need to take both medicines for a short period of time. It is important to follow your doctor's instructions in using your medicines
Your doctor may have prescribed ALVESCO for another reason. Ask your doctor or pharmacist if you have any questions about why ALVESCO has been prescribed for you.
ALVESCO is not addictive.
This medicine is available only with a doctor's prescription.
Before you use ALVESCO
When you must not use it
Do not use ALVESCO if you have an allergy to:
- any medicines containing ciclesonide
- any of the ingredients listed at the end of this leaflet
Some symptoms of an allergic reaction include skin rash, itching, shortness of breath or swelling of the face, lips or tongue, which may cause difficulty in swallowing or breathing.
Do not use ALVESCO to treat an acute asthma attack.
Do not give ALVESCO to a child under the age of 6 years.
Do not use ALVESCO after the expiry date (EXP) printed on the pack or if the packaging is damaged or shows signs of tampering. If it has expired or is damaged return it to your pharmacist for disposal.
If you are not sure whether you should start using ALVESCO, contact your doctor.
Before you start to use it
Tell your doctor or pharmacist if you have any allergies to:
- any other medicines
- any other substances, such as foods, preservatives or dyes
Tell your doctor if you are being treated for any fungal, bacterial or viral infections. Some treatments may interact with ALVESCO.
Tell your doctor if you are pregnant, intend to become pregnant. Like most medicines, ALVESCO is not recommended for use during pregnancy. If there is a need to consider ALVESCO during your pregnancy, your doctor will discuss the risks and benefits of using ALVESCO during pregnancy.
Tell your doctor if you are breastfeeding or plan to breastfeed. Your doctor will discuss the possible risks and benefits of using ALVESCO during breastfeeding.
Tell your doctor if you currently have an infection. If you take ALVESCO while you have an infection, the medication may hide some of the signs of an infection. This may make you think, mistakenly, that you are better or that it is not serious.
Tell your doctor if you have or have had tuberculosis (TB).
Tell your doctor if you have or are currently suffering from liver disease.
If you have not told your doctor about any of the above, tell them before you start using ALVESCO.
Using other medicines
Tell your doctor if you are using any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and ALVESCO may interfere with each other. These include:
- medicines used to treat fungal or viral infections, such as ketoconazole, itraconazole, ritonavir, nelfinavir
These medicines may affect how well ALVESCO works.
How to use ALVESCO
If you are not sure how to use an inhaler, ask your doctor or pharmacist.
Children should only use their inhaler on medical advice and with the help of an adult.
The correct way to use your ALVESCO inhaler is explained on the instructions for use leaflet that comes inside the ALVESCO package. If you do not use your ALVESCO inhaler correctly your lungs might get less medicine than needed. So please read these instructions carefully before you start.
To make sure your ALVESCO inhaler delivers the correct dose you should clean it regularly as outlined in the instruction leaflet.
The doctor may have advised that you use a spacer device with your inhaler. This is also recommended for children 6-11 years of age.
If so, the AeroChamber Plus is considered a suitable device for use with ALVESCO.
When to use it
Use ALVESCO at the same time every day.
How much to use
Your doctor will have spoken to you about how much of your medicine you need to take. This will depend on your individual needs.
Your doctor will normally start you on 80 micrograms once daily if you are not currently using a preventer medication. Otherwise your starting dose will depend on your current medication and how well your asthma is controlled.
There are 2 strengths of ALVESCO and your doctor will have chosen the one which best suits your asthma. The number of puffs you are required to take daily will depend on the strength of ALVESCO you are using. Your doctor may adjust your dose so that you are taking the lowest dose of ALVESCO that will control your symptoms.
When your doctor is changing you from a corticosteroid tablet to ALVESCO, you will be asked to take both medicines for a period of time. Your dose of corticosteroid tablets will gradually be reduced to the lowest possible level. It is important you follow your doctor's instructions.
If you forget to use it
If you forget to use your ALVESCO inhaler, skip the dose you missed and use your next dose when you are meant to.
Do not use a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to use your medicine, ask your pharmacist for some hints.
How long to use it
ALVESCO helps control your asthma. Therefore, you must use ALVESCO every day. Continue using the medicine for as long as your doctor tells you.
Do not stop using it unless your doctor tells you to - even if you feel better.
If you use too much (overdose)
Do not use more than the recommended dose unless your doctor tells you to.
The risk of effects from taking too many puffs of ALVESCO is low.
If you are worried that you or anyone else may have used too much ALVESCO, telephone your doctor, or the Poisons Information Centre (telephone Australia 13 11 26).
While you are using ALVESCO
Things you must do
If you have an Asthma Action Plan that you have agreed with your doctor, follow it closely at all times.
Tell any other doctors, dentists and pharmacists who are treating you that you are using ALVESCO.
Tell your doctor or pharmacist that you are using ALVESCO if you are about to be started on any new medicines.
If you plan to have surgery (that needs a general anaesthetic), tell your doctor or dentist that you are using ALVESCO.
If you become pregnant while you are using ALVESCO, tell your doctor or pharmacist.
Keep all of your doctor's appointments so that your progress can be checked.
Things you must not do
Do not take any other medicines for your breathing problems without checking with your doctor.
Do not give ALVESCO to anyone else, even if they have the same condition as you.
Do not use ALVESCO to treat any other complaints unless your doctor or pharmacist tells you to.
Do not stop using ALVESCO, or lower the dosage, without checking with your doctor or pharmacist.
Things to be careful of
If ALVESCO does not have its usual effect
If your asthma gets worse tell your doctor. Your doctor may tell you to take extra puffs of ALVESCO or add another medicine (such as a reliever puffer) to your treatment.
If your breathing suddenly becomes more difficult just after you have used ALVESCO, you will need to use a reliever puffer and tell your doctor immediately.
Tell your doctor as soon as possible if:
- your reliever puffer does not help your breathing as much as usual
- the effect of your reliever puffer does not last as long as usual
- you need more puffs of your reliever puffer than usual to get relief.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using ALVESCO.
All medicines can have unwanted side effects. Sometimes they are serious, but most of the time they are not. You may need medical attention if you get some of the side-effects.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- nausea
- bad taste in your mouth
- mouth dryness
- irritation in your mouth or throat
- skin rash
- high blood pressure
- increased difficulty in breathing immediately following the use of ALVESCO
If you experience any of the following, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- swelling of the lips, mouth, tongue or throat
- difficulty breathing
- severe rash or welts on your skin
You may need urgent medical attention or hospitalisation.
Very rarely, inhaled corticosteroids may make you feel anxious, depressed, irritable, or have disturbed sleep.
It is possible that some people, particularly those taking higher doses of ALVESCO for a long time may rarely suffer from the following side effects:
- rounded face
- loss of bone density
Tell your doctor if you notice any issues with your eyes such as blurred vision or other problems with your eyesight. Your doctor may need to send you to an ophthalmologist (eye doctor) to check that you don't have eye problems such as cataracts (clouding of the eye lens), glaucoma (increased pressure in your eyeballs) or other rare eye conditions reported with corticosteroid use.
Children and adolescents who are receiving treatment for a long period of time may have their height checked regularly by their doctor. If your growth rate is slowed your doctor may adjust your treatment.
Your adrenal glands may be affected by taking high doses of corticosteroids over a long period of time or if you change from or use high doses of oral corticosteroids. Your doctor may need to check how the adrenal glands are working.
Your doctor may also tell you to take additional oral corticosteroids during periods when your body is stressed such as during trauma, surgery and infection.
Other side effects not listed above may occur in some patients. Tell your doctor or pharmacist if you notice anything else that is making you feel unwell.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
After using ALVESCO
Cleaning
Clean and dry your inhaler each time you use it. It is important to keep your inhaler clean and dry. It may not work as well if it gets dirty. Follow the instructions at the end of this leaflet for cleaning your inhaler.
Storage
Keep your inhaler in a cool, dry place where the temperature stays below 25°C.
Do not store ALVESCO or any other medicine in the bathroom, near a sink.
Do not leave it in the car on hot days or on a window sill. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop using ALVESCO, or it has passed its expiry date, ask your pharmacist what to do with any that are left over.
Do not puncture or incinerate even when empty as the canister may explode.
Product description
What it looks like
The ALVESCO inhaler comprises an aluminium canister inside a plastic inhaler unit. The inhaler unit has a mouthpiece and a plastic cover.
ALVESCO comes in two different strengths - 80 micrograms and 160 micrograms.
ALVESCO 80 inhaler is registered* in 120 puffs.
ALVESCO 160 inhaler is registered* in 60 and 120 puffs.
*not all registered strengths and/or pack sizes might be available in Australia
Ingredients
ALVESCO contains ciclesonide as the active ingredient.
It also contains:
- norflurane (a non-CFC propellant which does not deplete ozone from the atmosphere)
- ethanol
ALVESCO does not contain gluten, sucrose, lactose, tartrazine or any other azo dyes.
Supplier
Chiesi Australia Pty Ltd
Suite 3, 22 Gillman Street,
Hawthorn East, VIC 3123
Email: [email protected]
Website: www.chiesi.com
This leaflet was prepared on July 2021
Australian Registration Number(s)
80 micrograms: AUST R 93724
160 micrograms: AUST R 93725
Instructions for Use
ALVESCO metered dose inhaler
Before using ALVESCO, please read these instructions carefully and follow them closely.
This leaflet does not contain all the available information. If you have any questions about ALVESCO, please refer to the Consumer Medicine Information on this leaflet. If this is a new inhaler or if you have not used your inhaler for one week or more, it must be tested before use by removing the mouthpiece cover and pressing down on the canister inside the inhaler. Release 3 puffs into the air, away from you.
You do not need to shake your inhaler before taking your medicine.
The medicine is already in a very fine solution, mixed to ensure you receive the correct dose with each puff.
Follow these instructions carefully and use the pictures to help you.
- Remove the mouthpiece cover and check inside and outside to make sure that the mouthpiece is clean and dry.

- Hold the inhaler upright with your forefinger on the top of the canister and your thumb on the base, below the mouthpiece.
- Breathe out as far as is comfortable.
- Place the mouthpiece in your mouth and close your lips firmly around it.
- Just after starting to breathe in through your mouth, press down on the top of the inhaler to release a puff while still breathing in slowly and deeply.
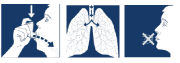
- Hold your breath, take the inhaler from your mouth and remove your finger from the top of the inhaler. Continue holding your breath for approximately ten seconds or as long as is comfortable.
- Breathe out slowly through your mouth.

- If you have been instructed to take another puff, wait about half a minute and repeat steps 3 to 7.
- After use, always replace the mouthpiece cover to keep out dust.

Replace firmly and snap into position.
It is important that you do not rush steps 3 to 7
A correct technique will ensure the right amount of ALVESCO is getting into your lungs every time you use your inhaler.
Care and Cleaning instructions
The mouthpiece of your inhaler should be cleaned weekly with a dry tissue or cloth.
DO NOT WASH OR PUT ANY PART OF THE ALVESCO INHALER IN WATER.
You should practice in front of the mirror for the first couple of times until you are confident that you are operating your inhaler properly. Make sure that none of your medicine is escaping from the top of inhaler or sides of your mouth.
If you find it difficult to use the inhaler with one hand, try using both hands. Put your two forefingers on top of the inhaler and both thumbs on the base below the mouthpiece. Tell your doctor, nurse or pharmacist if you are having difficulties.
When the canister is completely empty you will not feel or hear any of the propellant being discharged.
Use of inhaler with a spacer device
If your inhaler technique is poor, your doctor may recommend the use of a spacer device in combination with your inhaler. Different brands of spacers may affect the amount of medicine delivered to the lungs, so it is important that you be monitored for any loss of asthma control.
Before use, the spacer should be removed from the inhaler and washed in warm water and detergent and allowed to dry without rinsing or drying with a cloth. Wash the spacer at least monthly after use.
When using a spacer, you should inhale immediately after you press down on the top of the inhaler to release a puff.
Why would your Inhaler have stopped working?
Your inhaler may not work if:
- the mouthpiece is clogged and dirty
- the inhaler is empty (check by shaking the canister)
- the canister is wrongly positioned
If you have any questions about your ALVESCO medication, please refer to the Consumer Medicine Information.
Published by MIMS September 2021



 Patients previously maintained on another ICS may require a higher dose depending on their current maintenance dose. Alvesco can be administered as 1 or 2 puffs once daily either in the morning or evening. In the case of a higher dose, twice daily administration is recommended (see Adult patients - higher doses in certain circumstances). The dose should be adjusted to the lowest dose at which effective control of asthma is maintained.
Patients previously maintained on another ICS may require a higher dose depending on their current maintenance dose. Alvesco can be administered as 1 or 2 puffs once daily either in the morning or evening. In the case of a higher dose, twice daily administration is recommended (see Adult patients - higher doses in certain circumstances). The dose should be adjusted to the lowest dose at which effective control of asthma is maintained. The following adverse reactions have also been reported in clinical trials with Alvesco. (See Table 3.)
The following adverse reactions have also been reported in clinical trials with Alvesco. (See Table 3.) Paradoxical bronchospasm may occur immediately after dosing and is an unspecific acute reaction to all inhaled medications, which may be related to the drug, the excipient or evaporation cooling in the case of metered dose inhalers. In the majority of cases, this reaction is mild and does not require withdrawal of Alvesco. In severe cases, withdrawal of Alvesco should be considered.
Paradoxical bronchospasm may occur immediately after dosing and is an unspecific acute reaction to all inhaled medications, which may be related to the drug, the excipient or evaporation cooling in the case of metered dose inhalers. In the majority of cases, this reaction is mild and does not require withdrawal of Alvesco. In severe cases, withdrawal of Alvesco should be considered.




