What is in this leaflet
This leaflet answers some common questions about AmBisome.
The medicine your doctor has prescribed for you is called AmBisome for Injection. This is the brand name of a drug called liposomal amphotericin B (amphotericin).
All medicines have risks and benefits. Your doctor has weighed the risks of you having AmBisome against the benefits it is expected to have for you.
If you have any concerns about having this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What is AmBisome used for
AmBisome contains amphotericin B (amphotericin) which belongs to a group of medicines called antifungals.
It is used to help the body overcome serious fungal infections by either killing the fungus or stopping it spreading.
Amphotericin B (amphotericin) is also used to treat a disease which is rare in Australia called leishmaniasis.
Ask your doctor if you have any questions about why AmBisome has been prescribed for you. Your doctor may have prescribed AmBisome for another reason.
AmBisome is not addictive.
This medicine is available only with a doctor’s prescription.
Before you are given AmBisome
When you must not have it:
Do not have AmBisome if you are allergic to:
- amphotericin B (amphotericin) or
- any of the other ingredients in AmBisome listed at the end of this leaflet.
Before you are given it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes. Tell your doctor if you have, or have had, any of the following medical conditions:
- kidney disease
- liver disease
- diabetes
Tell your doctor if you are pregnant, or likely to become pregnant during your course of medication. Your doctor can discuss with you the benefits and risks of having AmBisome.
Tell your doctor if you are breastfeeding, or likely to breastfeed during your course of medication. AmBisome is not recommended during breastfeeding, as it may pass into breast milk.
If you have not told your doctor or pharmacist about any of the above, tell them before you are given AmBisome.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and AmBisome may interfere with each other. These include:
- anticancer agents (chemotherapy)
- digoxin (e.g. Lanoxin)
- flucytosine (e.g. Ancotil)
- cortisone-like medicine (e.g. hydrocortisone, betamethasone, dexamethasone, prednisolone, methylprednisolone)
- corticotrophin (e.g. Acthargel)
- dipyridamole (e.g. Persantin)
These medicines may be affected by AmBisome or may affect how well it works. You may need different amounts of the medicine, or you may need to take different medicines. Your doctor or pharmacist will advise you.
How AmBisome is given
AmBisome is to be administered only by or under the supervision of your doctor.
AmBisome must be reconstituted using Water for Injection and then filtered using a 5-micron filter into the correct amount of 5% Glucose Injection. It is given as a long running infusion (e.g. a slow injection via a “drip” into a vein).
If you have too much (overdose)
As AmBisome is most likely to be given to you in hospital under the supervision of your doctor, it is very unlikely that you will receive an overdose.
Severe side effects due to AmBisome are more likely to occur at high doses, particularly if you receive an overdose. Symptoms of AmBisome overdose include the side effects listed, but are usually of a more severe nature.
However, if you experience any severe side effects after being given AmBisome, tell your doctor immediately.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are given AmBisome.
All medicines can have side effects. If they occur most are minor and temporary. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects. Do not be alarmed by these lists of possible side effects. You may not experience any of them.
Tell your doctor if you notice any of the following and they worry you:
Common side effects:
- nausea and vomiting
- rash
- mild headaches
Less common side effects. The following side effects have been reported:
- back pain
- joint pain and bone pain
- fever, chills
- chest tightness
- kidney problems
- liver problems
Check with your doctor or pharmacist if you experience or do not understand any of the side effects described above.
Tell your doctor if you notice anything else that is making you feel unwell. Other side effects not listed above may also occur in some people.
After receiving AmBisome
Storage
AmBisome should be stored in the pharmacy or on the hospital ward.
Keep out of reach of children.
Do not keep or use the medicine after the expiry date marked on the packet.
Unopened vials of AmBisome should be stored below 25°C.
Product Description
Ingredients
Each injection contains amphotericin equivalent to 50 mg of amphotericin B (amphotericin) plus hydrogenated soy phosphatidylcholine, cholesterol, distearoylphosphatidylglycerol (as the sodium salt), alpha tocopherol, sucrose and sodium succinate hexahydrate.
Manufacturer
AmBisome is supplied in Australia by:
Gilead Sciences Pty Ltd
Level 6, 417 St Kilda Road
Melbourne, Victoria 3004
AUST R 53783
AmBisome is supplied in New Zealand by:
Gilead Sciences (NZ)
c/- PricewaterhouseCoopers
Level 8 Pricewaterhousecoopers Tower
188 Quay Street
Auckland 1010
AmBisome® is a registered trademark licensed to Gilead Sciences Pty. Limited.
Date of Preparation: 30 March 2017
Published by MIMS March 2026


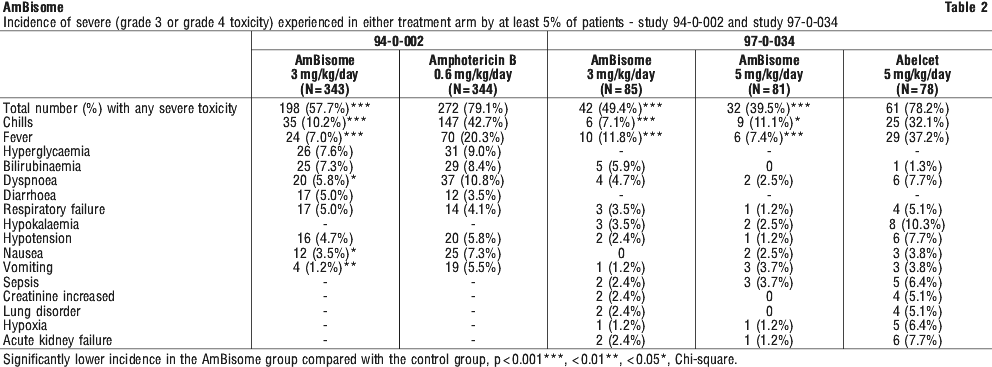


 In study 94-0-002, there were significantly more emergent proven infections in conventional amphotericin B treated patients compared to AmBisome treated patients (7.8% vs. 3.2%, respectively, p = 0.009).
In study 94-0-002, there were significantly more emergent proven infections in conventional amphotericin B treated patients compared to AmBisome treated patients (7.8% vs. 3.2%, respectively, p = 0.009). The values in Table 6 were determined by pooling serial measurements of serum data at each dose level, generating a curve fit and calculating the pharmacokinetic parameters from that curve. Patient numbers were small and the number of measurements pooled was not constant across all patients at each dose level.
The values in Table 6 were determined by pooling serial measurements of serum data at each dose level, generating a curve fit and calculating the pharmacokinetic parameters from that curve. Patient numbers were small and the number of measurements pooled was not constant across all patients at each dose level.
