What is in this leaflet
This leaflet answers some common questions about ANAMORPH tablets.
It does not contain all the available information about this medicine.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking ANAMORPH against the benefits they expect it will have for you.
Ask your doctor or pharmacist if you have any concerns about taking this medicine.
Keep this leaflet with the medicine. You may need to read it again.
What is ANAMORPH used for
The name of your medicine is ANAMORPH. It is available as a 30 mg tablet.
The active ingredient is called morphine sulfate.
Morphine sulfate belongs to a group of medicines called analgesics. It is an opioid analgesic, and it acts in the central nervous system by blocking pain and your emotional response to pain.
ANAMORPH is used to treat moderate to severe pain.
Your doctor however may have prescribed ANAMORPH for another purpose.
Ask your doctor if you have any questions about why ANAMORPH has been prescribed for you.
This medicine only available with a doctor’s prescription.
ANAMORPH may be habit-forming, so it is important to take it exactly as directed by your doctor.
As with all other opioid containing products, your body may become used to you taking ANAMORPH. Taking it may result in physical dependence. Physical dependence means that you may experience withdrawal symptoms if you stop taking ANAMORPH suddenly, so it is important to take it exactly as directed by your doctor.
Tolerance to [Product] may develop, which means that the effect of the medicine may decrease. If this happens, more may be needed to maintain the same effect.
Before you take it
When you must not take it
Do not take ANAMORPH if you are allergic to:
- morphine sulfate or any other analgesics
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty in breathing; swelling of the face, lips, tongue or other parts of the body; rash, itching or hives on the skin.
This medicine should not be taken in the presence of the following medical conditions / health problems:
- severe breathing or lung problems such as asthma, bronchitis or emphysema
- head injury or brain tumor
- raised pressure in the head or spine
- fits or convulsions such as epilepsy
- kyphoscoliosis (abnormal curvature of the spine)
- heart problems such as chronic pulmonary disease
- irregular heart beat
- severe CNS depression
- alcoholism (or use of excessive amounts of alcohol)
- kidney or liver disease
- recently had biliary tract surgery
- inflammatory bowel disease
- gastrointestinal obstruction or obstructive bowel disorders
Do not take ANAMORPH if you are taking a medicine for depression called a ‘monoamine oxidase inhibitors (MAOIs)’ e.g. Parnate®, Nardil®, Aurorix®, or have taken one within the past two weeks.
Do not take it after the expiry date (EXP.) printed on the pack. If you take it after the expiry date has passed, it may have no effect at all, or worse, there may be an entirely unexpected effect.
Do not take it if the packaging is torn or shows signs of tampering. If it is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
You must tell your doctor if:
- You are allergic to any other medicines or any foods, dyes or preservatives.
- You are pregnant or intend to become pregnant.
Your doctor can discuss the risks and benefits involved of taking ANAMORPH during pregnancy. - You are breast-feeding or intend to breastfeed.
This medicine passes into breast milk, so it is not recommended for nursing mothers.
Tell your doctor if you have had or have any of the following medical conditions / health problems:
- an underactive thyroid gland (hypothyroidism)
- an underactive adrenal gland
- increased prostate size
- low blood pressure
- narrowing of the urinary bladder tract
- inflammation of the pancreas
- epilepsy (a disease that causes fits, convulsions or seizures)
- stomach pains, constipation or diarrhoea
If you have not told your doctor about any of the above, tell them before you start taking ANAMORPH.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including medicines that you buy without a prescription from a pharmacy, supermarket or health food shop.
SOME of these medicines may interfere with ANAMORPH. These include:
- alcohol
- medicines used to treat high blood pressure and heart conditions e.g. propranolol
- some medicines used during surgery and emergency situations such as anaesthetics
- cimetidine, medicine used to treat stomach ulcers
- atropine, medicine used in some eye drops
- some medicines used to treat depression
- medicines used to treat fever, pain and inflammation e.g. paracetamol
- medicines used to help you sleep
- medicines used to help relieve anxiety
- stimulants such as dexamphetamine
- medicines used to control epilepsy e.g. diazepam
- medicines used to treat certain mental and emotional conditions
The above medicines may either reduce the effectiveness of ANAMORPH, or react with it resulting in untoward or sometimes dangerous side effects.
This list is not exhaustive. Your doctor or pharmacist has more information on medicines to be careful with or avoid while taking ANAMORPH.
Tell your doctor or pharmacist that you are taking ANAMORPH before you start to take any other medicine.
Use in Children
This medicine is not recommended for use in children.
Use in Elderly or Debilitated Patients
Elderly or debilitated patients are more likely to have less effective kidney or liver function due to age. This may increase the risk of side effects. You should discuss how much ANAMORPH to use with your doctor or pharmacist.
How to take it
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the pack, ask your doctor or pharmacist for help.
How much to take
The recommended doses of ANAMORPH are:
Adults:
The initial dose is ½ to 1 tablet, every four to six hours as necessary or as directed by your doctor.
Elderly patients may need smaller doses and/or longer dosage intervals.
How to take it
Swallow the tablets whole with a full glass of water.
When to take it
It does not matter if you take ANAMORPH before or after food.
This medicine should be taken at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
How long to take it
Continue taking your medicine for as long as your doctor tells you. If you stop having this medicine suddenly, your pain may worsen and you may experience some or all of the following withdrawal symptoms:
- nervousness, restlessness, agitation, trouble sleeping or anxiety
- body aches, weakness or stomach cramps
- loss of appetite, nausea, vomiting or diarrhoea
- increased heart rate, breathing rate or pupil size
- watery eyes, runny nose, chills or yawning
- increased sweating.
ANAMORPH given to the mother during labour can cause breathing problems and signs of withdrawal in the newborn.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take your dose as soon as you remember, and then go back to taking it as you would normally.
Do not take a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you are unsure about whether to take your next dose, speak to your doctor or pharmacist.
If you have trouble remembering when to take your medicine, ask your pharmacist for some hints.
If you take too much (Overdose):
If you or someone else receive too much (overdose), and experience one or more of the symptoms below, immediately remove the patch(es) and call triple zero (000) for an ambulance.
Keep the person awake by talking to them or gently shaking them every now and then. You should follow the above steps even if someone other than you have accidentally used ANAMORPH that was prescribed for you.
If someone takes an overdose they may experience one or more of the following symptoms:
- Slow, unusual or difficult breathing
- Drowsiness, dizziness or unconsciousness
- Slow or weak heartbeat
- Nausea or vomiting
- Convulsions or fits
If you think you or someone else may have used too much ANAMORPH, you should immediately:
- phone the Poisons Information Centre (by calling 13 11 26), or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
When seeking medical attention, take this leaflet and remaining medicine with you to show the doctor. Also tell them about any other medicines or alcohol which have been taken.
While you are taking it
Things you must do
Immediately stop taking ANAMORPH if a skin rash or any other allergic reaction occurs.
Tell your doctor or pharmacist that you are taking this medicine if you are about to start any new medicine.
Tell any other doctors, dentists and pharmacists who are treating you that you are taking it.
Tell your doctor immediately if you become pregnant.
Tell your doctor if you are breast-feeding while you are taking this medicine.
Tell the surgeon or anaesthetist that you are taking this medicine if you are going to have surgery.
Keep all of your doctor’s appointments so that your progress can be checked.
Always discuss with your doctor any problems or difficulties during or after taking ANAMORPH.
Things you must not do
Do not take ANAMORPH to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop taking your medicine or lower the dosage without first checking with your doctor.
Things to be careful of
Be careful driving or operating machinery until you know how ANAMORPH affects you. This medicine may cause dizziness, light-headedness or drowsiness in some people. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
Be careful when drinking alcohol while you are taking ANAMORPH. If you drink alcohol, it could make some of the unwanted side effects worse.
Your doctor may suggest that you avoid alcohol completely or reduce the amount of alcohol you drink while you are using ANAMORPH.
If you feel light-headed, dizzy or faint when getting out of bed or standing up, get up slowly. Standing up slowly, especially when you get up from bed or chairs, will help your body get used to the change in position and blood pressure. If this problem continues or gets worse, talk to your doctor.
You can become addicted to ANAMORPH even if you take it exactly as prescribed.
ANAMORPH may become habit forming causing mental and physical dependence. If abused it may become less able to reduce pain.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking ANAMORPH. This medicine helps most people with the medical conditions listed in the beginning of this leaflet, but it may have unwanted side effects in some people.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Elderly patients may have an increased chance of getting side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- drowsiness, and dizziness
- restlessness
- confusion
- nervousness
- headache
- dry mouth
- weakness
- stomach pain or cramps
- mood changes
- flushing of the face
- nausea (feeling sick)
- vomiting
- loss of appetite
- constipation
- sweating
- inability to sleep
- unusual movements including tremor
- breathing difficulty
These side effects are common. They are usually mild and short-lived.
Tell your doctor as soon as possible if you notice any of the following:
- skin rashes which include severe itching
- spinning sensation
- fainting
- pounding or irregular heart beats
- problems in passing urine
- seeing, feeling or hearing things that are not there (hallucinations)
These are serious side effects that may require medical attention. Serious side effects are rare.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- seizures, fits or convulsions
- coma
These are very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare.
Some people may get other side effects while taking ANAMORPH.
Check with your doctor as soon as possible if you have any problems while taking ANAMORPH, even if you do not think the problems are connected with the medicine or are not listed in this leaflet.
After taking it
Storage
Keep your tablets in the pack until it is time to take them.Keep your tablets in a cool dry place where the temperature stays below 30°C. Protect from light.
Do not store it or any other medicine in the bathroom or near a sink.
Do not leave it in the car or on windowsills. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Do not take ANAMORPH if the tablets do not look quite right.
Disposal
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
Product description
What it looks like
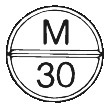
ANAMORPH tablets (oral) are 30 mg; round, white, flat bevelled edge tablets, scored on one side and marked with “M” above the breakline and “30” below the breakline; blister pack; 6’s, 8’s, 10’s, 20’s.
Not all pack sizes may be available.
Ingredients
ANAMORPH tablets contain 30 mg of morphine sulfate pentahydrate as the active ingredient.
ANAMORPH tablets also contain the following ingredients:
- lactose
- croscarmellose sodium
- magnesium stearate
ANAMORPH tablets are gluten and sucrose free, but they contain sugars as lactose.
Sponsor
Arrotex Pharmaceuticals Pty Ltd
15-17 Chapel Street
Cremorne VIC 3121
The Australian Registration Number for ANAMORPH 30 mg tablets is AUST R 34032.
This leaflet was revised in June 2023.
Published by MIMS August 2023