1 Name of Medicine
Garadacimab.
2 Qualitative and Quantitative Composition
Garadacimab 200 mg solution for injection in pre-filled pen (auto-injector).
Each single-dose pre-filled pen contains 200 mg of garadacimab in 1.2 mL of solution.
Garadacimab 200 mg solution for injection in pre-filled syringe with needle safety device.
Each single-dose pre-filled syringe with needle safety device contains 200 mg of garadacimab in 1.2 mL of solution.
Garadacimab is a recombinant, fully human, monoclonal antibody (IgG4/λ-light chain) produced by recombinant DNA technology in Chinese Hamster Ovarian (CHO) cells.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Solution for injection.
The solution of Andembry is slightly opalescent to clear, brownish-yellow to yellow liquid.
4.1 Therapeutic Indications
Andembry is indicated for routine prevention of recurrent hereditary angioedema (HAE) attacks in patients aged 12 years and older with C1-INH HAE (C1-esterase inhibitor deficiency or dysfunction).
4.2 Dose and Method of Administration
This medicinal product should be initiated under the supervision of a healthcare professional experienced in the management of patients with HAE.
Andembry is not intended for the treatment of acute HAE attacks (see Section 4.4 Special Warnings and Precautions for Use).
Dosage.
The recommended dose of Andembry is an initial loading dose of 400 mg administered subcutaneously as two 200 mg injections on the first day of treatment followed by a monthly dose of 200 mg.
Missed doses.
If a dose of Andembry is missed, administer the dose as soon as possible and then administer the next dose according to the original dosing schedule.
Method of administration.
Andembry is intended for subcutaneous (SC) administration only.
Each Andembry unit (pre-filled pen or pre-filled syringe with needle safety device) is intended for single use in one patient only. Discard any residue.
Administer Andembry into the abdomen or thigh. If Andembry is administered by somebody else, the upper arm can also be used. Rotation of the monthly injection site is recommended.
Andembry may be self-administered or administered by a caregiver only after training on SC injection technique by a healthcare professional.
For further information, see the Instructions for Use in the Consumer Medicine Information.
Paediatric population.
The safety and efficacy of garadacimab in children aged less than 12 years have not been established.
No data are available.
Elderly population.
The safety and efficacy of garadacimab is not expected to be affected by age. No dose adjustment is required for patients above 65 years of age (see Section 5.2 Pharmacokinetic Properties).
Hepatic impairment.
No studies have been conducted in patients with hepatic impairment (see Section 4.4 Special Warnings and Precautions for Use; Section 5.2 Pharmacokinetic Properties).
Renal impairment.
No studies have been conducted in patients with renal impairment. Renal impairment is not expected to affect exposure to garadacimab (see Section 5.2 Pharmacokinetic Properties).4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients.
4.4 Special Warnings and Precautions for Use
General.
Andembry is not intended for treatment of acute HAE attacks. In case of breakthrough HAE attack, individualised treatment should be initiated with an approved rescue medicine.
Traceability.
In order to improve the traceability of biological medicinal products, the name and the batch number of the administered product should be clearly recorded.
Hypersensitivity.
Hypersensitivity reactions have been observed (see Section 4.8 Adverse Effects (Undesirable Effects)). The signs and symptoms of hypersensitivity reactions may include hives (local and generalised), tightness of the chest, difficulty breathing, wheezing, hypotension, and/or anaphylaxis during or after injection of Andembry. In case of severe hypersensitivity, institute appropriate treatment immediately and discontinue Andembry administration.
Use in hepatic impairment.
No studies have been conducted in patients with hepatic impairment. The safety of garadacimab in patients with hepatic impairment has not been established.
Use in the elderly.
There were 13 subjects aged 65 years of age or older included in the Phase 3 studies, including 6 in the VANGUARD pivotal study. No difference in the overall safety profile was seen between adults 18-65 and ≥ 65 years of age, suggesting that the safety and tolerability of Andembry in adults ≥ 65 is similar to the 18-65 year old population studied.
Paediatric use.
The safety and efficacy of garadacimab in children (< 12 years old) has not been established. There are limited data for adolescent subjects aged 12 to 17 years. There were 11 adolescent subjects aged 12 to 17 years in the Phase 3 studies, including 6 in the VANGUARD pivotal study. No difference in the overall safety profile was seen between adults and children 12-17 years old, suggesting that the safety and tolerability of Andembry in children 12-17 years old is similar to that in adults.
No data are available for children aged less than 12 years. Treatment in children aged < 12 years is not recommended.
Effects on laboratory tests.
Coagulation tests.
Garadacimab can prolong activated partial thromboplastin time (aPTT) due to an interaction of garadacimab with the aPTT assay. The reagents used in the aPTT laboratory test initiate intrinsic coagulation through the activation of FXII in the contact system, therefore inhibition of plasma FXIIa by garadacimab can prolong aPTT in this assay. None of the increases in aPTT in patients treated with garadacimab were associated with bleeding adverse events. There were no clinically relevant differences in international normalised ratio (INR) between treatment groups.4.5 Interactions with Other Medicines and Other Forms of Interactions
No medicine interaction studies have been conducted in humans.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
Male and female fertility were unaffected based upon a lack of adverse findings on mating, fecundity, fertility indices, maternal reproductive parameters, embryo survival or sperm assessment in sexually mature rabbits that received garadacimab up to doses of 100 mg/kg IV once every 3 days (resulting in approximately 103- and 83-fold for males and females, respectively, the clinical exposure at the recommended human dose of 200 mg SC once monthly based on AUC).
Effect on fertility has not been evaluated in humans.
(Category B1)
There are limited data from the use of Andembry in pregnant women. A risk to the pregnant woman or developing fetus cannot be excluded. A decision should be made whether to initiate or discontinue treatment with Andembry, taking into account the risk/benefit of therapy. Monoclonal antibodies such as garadacimab are transported across the placenta mainly during the third trimester of pregnancy; therefore, potential effects on a fetus are likely to be greater during the third trimester of pregnancy. No adverse effects were reported in a rabbit embryofetal development study where IV doses up to 100 mg/kg (104-fold maximum clinical exposure based on AUC) were administered during organogenesis. A pre- and post-natal development study conducted in pregnant rabbits at SC doses up to 100 mg/kg/dose from implantation through weaning resulting in exposures of approximately 53 times the maximum clinical exposure (based on AUC) revealed no evidence of harm to the developing fetus or subsequent F1 offspring.
It is unknown whether Andembry is excreted in human milk. Human IgG are known to be excreted in breast milk during the first few days after birth, and decrease to low concentrations soon afterwards. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Andembry and any potential adverse effects on the breastfed infant from Andembry or the underlying condition.4.7 Effects on Ability to Drive and Use Machines
Andembry has negligible influence on the ability to drive or use machines.
4.8 Adverse Effects (Undesirable Effects)
Summary of the safety profile.
The most commonly observed adverse reactions (5.1%) associated with Andembry were injection site reactions (ISRs) including injection site erythema, injection site bruising and injection site pruritus. All these ISRs were mild in nature and resolved within 1-3 days with no change in dosage.
Tabulated list of treatment-emergent adverse events and adverse reactions.
Table 1 and Table 2 summarise treatment-emergent adverse events and adverse reactions observed in the VANGUARD pivotal study conducted with Andembry on 39 HAE patients in a phase 3, multicentre, randomised, double-blind, placebo-controlled parallel group study, where 75 adverse events were reported in 25/39 (64.1%) patients. Of these 75 events, 9 were reported as related to Andembry in 4/39 (10.3%) patients.
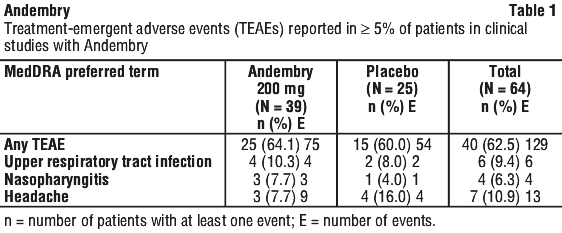 The following list (Table 2) of adverse reactions is based on experience from clinical trials and is displayed by system organ class and frequency: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000) and not known (cannot be estimated from the available data).
The following list (Table 2) of adverse reactions is based on experience from clinical trials and is displayed by system organ class and frequency: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000) and not known (cannot be estimated from the available data).
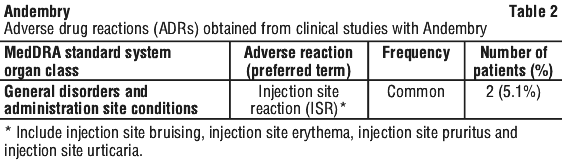
Description of selected adverse reactions.
Injection site reactions.
In the VANGUARD pivotal study, 3 incidents of ISR of mild severity were observed in 2 patients who received Andembry. Temporal relationship (began within 1-3 days after investigational product administration and resolved within 1-3 days with no change in dosage) was identified for all 3 ISRs, all of which were assessed as related to Andembry.
In a Phase 3 open-label extension study, which included 57 patients who rolled over from VANGUARD, 161 patients with HAE administered Andembry 200 mg subcutaneously every month. Injection site reactions (e.g. injection site bruising, injection site erythema, injection site haematoma, injection site pruritus, injection site urticaria) were reported in 16 (10%) patients.
Immunogenicity.
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies. No in vitro data regarding the presence of neutralising antibodies in ADA-positive patients is available.
In the VANGUARD pivotal study, 1 (2.6%) Andembry treated patient had an anti-drug-antibody (ADA)-positive sample (reciprocal titre value of 10) during the 6-month treatment period. Five (3.1%) patients had at least 1 ADA-positive sample (reciprocal titre values of 10-640) during the Phase 3 open-label extension study.
The development of ADA against Andembry did not affect pharmacokinetics (PK), pharmacodynamics (PD), safety or clinical response.
Postmarketing experience.
Hypersensitivity.
Cases of hypersensitivity have been observed during postmarketing use of Andembry (see Section 4.4 Special Warnings and Precautions for Use). Because postmarketing reporting of adverse reactions is voluntary and from a population of uncertain size, it is not always possible to reliably estimate the frequency of these reactions or establish a causal relationship to product exposure.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
No cases of overdose were reported in clinical studies. There is no available information to identify potential signs and symptoms of overdose.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Garadacimab is a specific inhibitor of activated FXII. Garadacimab is a novel fully human IgG4/lambda recombinant monoclonal antibody which binds to the catalytic domain of activated Factor XII (FXIIa and βFXIIa) and potently inhibits its catalytic activity. FXII is the first factor activated in the contact activation pathway and initiates the inflammatory bradykinin-producing kallikrein-kinin system. The inhibition of FXIIa prevents the activation of prekallikrein to kallikrein and the generation of bradykinin, which is associated with inflammation and swelling in HAE attacks, thus blocking the cascade of events leading to an HAE attack.
Pharmacodynamics.
Concentration-dependent inhibition of FXIIa-mediated kallikrein activity was demonstrated after SC administration of Andembry in patients with HAE.
In the VANGUARD pivotal study, Andembry did not prolong the QT/QTc interval.
Clinical trials.
VANGUARD pivotal phase 3 study (NCT04656418).
The efficacy and safety of Andembry for the prevention of HAE attacks in patients 12 years of age and older with Type I or II HAE were investigated in a phase 3, multicentre, randomised, double-blind, placebo-controlled parallel group study.
The study included 58 adult and 6 paediatric patients (12 years of age and older) who experienced at least 2 attacks during the up to 2 month run-in period. Patients were randomised into 2 parallel treatment arms in a 3:2 ratio (garadacimab 200 mg monthly after an initial 400 mg loading dose or volume-matched placebo) for a 6-month treatment period. Patients were required to discontinue other prophylactic HAE medications prior to entering the study. All patients were allowed to use on-demand medications for treatment of HAE attacks during the study.
Overall, 87.5% of patients had Type I HAE. A family history of HAE was reported for 89.1%, a history of laryngeal oedema attacks for 59.4% of patients and 32.8% were on prior prophylaxis. During the study run-in period, attack rates of ≥ 3 attacks/month were observed in 59.4% of patients overall.
Andembry demonstrated clinically meaningful and statistically significant efficacy when compared with placebo across all primary and secondary endpoints in the Intent-to-Treat (ITT) population as seen in (Table 3).
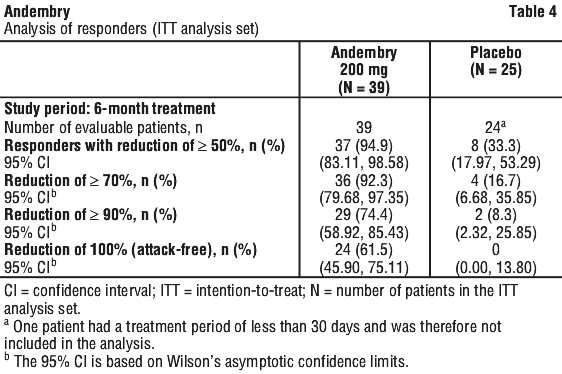
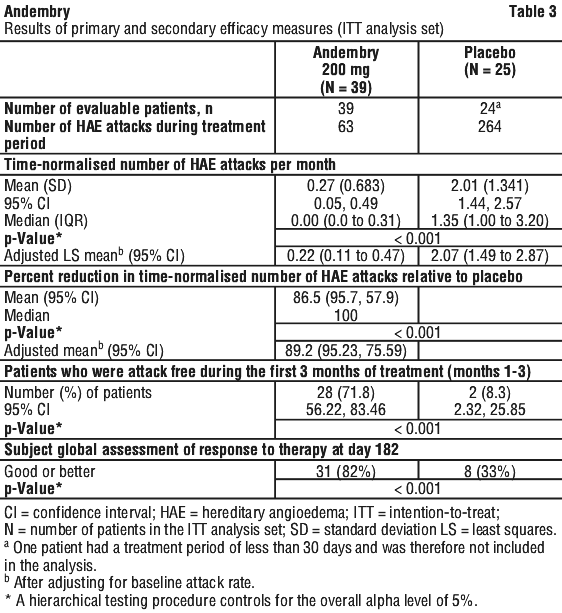 The minimum response was defined as a ≥ 50% reduction in the time normalised number of HAE attacks during the treatment period vs run-in. The proportion of patients with ≥ 50% reduction (responders), ≥ 70% reduction and ≥ 90% reduction in HAE attack rates during the treatment period compared to run-in is presented in Table 4. 71.8% of patients treated with Andembry were attack-free throughout the first 3 months of treatment vs. 8.3% of patients treated with placebo. Over the entire 6 months of treatment, the percentage of attack-free patients was 61.5% in the Andembry arm compared with zero patients attack-free in the placebo arm.
The minimum response was defined as a ≥ 50% reduction in the time normalised number of HAE attacks during the treatment period vs run-in. The proportion of patients with ≥ 50% reduction (responders), ≥ 70% reduction and ≥ 90% reduction in HAE attack rates during the treatment period compared to run-in is presented in Table 4. 71.8% of patients treated with Andembry were attack-free throughout the first 3 months of treatment vs. 8.3% of patients treated with placebo. Over the entire 6 months of treatment, the percentage of attack-free patients was 61.5% in the Andembry arm compared with zero patients attack-free in the placebo arm.
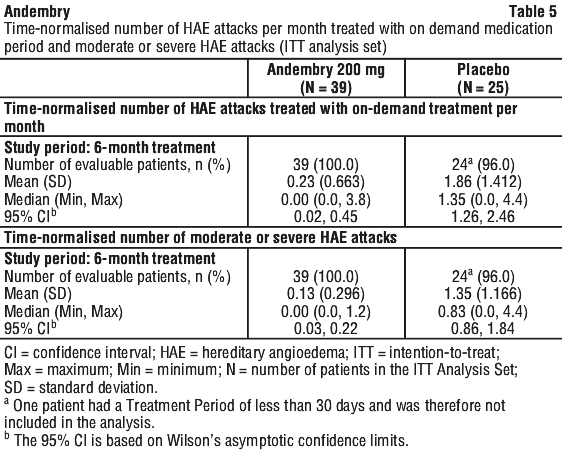
 The time-normalised number of HAE attacks per month treated with on-demand medication and the number of moderate or severe HAE attacks were reduced with Andembry treatment compared with placebo (Table 5).
The time-normalised number of HAE attacks per month treated with on-demand medication and the number of moderate or severe HAE attacks were reduced with Andembry treatment compared with placebo (Table 5).
 The proportion of subjects who achieved an improvement in quality of life as measured by the Angioedema Quality of Life (AE-QoL) questionnaire (minimal clinically important difference ≥ 6 for the AE-QoL total score) was 87.9% for the Andembry arm and 55.0% for the placebo arm.
The proportion of subjects who achieved an improvement in quality of life as measured by the Angioedema Quality of Life (AE-QoL) questionnaire (minimal clinically important difference ≥ 6 for the AE-QoL total score) was 87.9% for the Andembry arm and 55.0% for the placebo arm.
The safety and efficacy profile in paediatric patients 12 years of age and older (n = 6) was consistent with that of the overall population.
VANGUARD open label extension study (NCT04739059).
The ongoing VANGUARD open label extension study is a phase 3, multicentre, open-label, uncontrolled study designed to investigate long-term safety and efficacy of Andembry. The study enrolled 161 patients, including 57 patients from the VANGUARD pivotal study, 35 patients from a phase 2 multicentre, randomised, placebo-controlled, parallel-arm study, and 69 newly enrolled patients naïve to treatment with Andembry.
At the time of the interim analysis 2 (data cutoff 13-Feb-2023) the mean (SD) time-normalised number of HAE attacks per month for the 36 garadacimab-treated patients from the VANGUARD pivotal study who rolled over into the open-label extension study was 0.11 (0.316) attacks/month.
Overall, the safety and efficacy results in the VANGUARD open label extension study were consistent with those of the VANGUARD pivotal study and support the continued safety and efficacy over time of Andembry for the prevention of HAE attacks.
5.2 Pharmacokinetic Properties
Based on population PK analysis, garadacimab pharmacokinetics and steady-state exposure in subjects with HAE in VANGUARD pivotal study given 400 mg (2 x 200 mg) SC loading dose followed by 200 mg once monthly SC are provided in Table 6. Following SC administration of garadacimab, peak plasma concentration is reached within ~6 days, and terminal elimination half-life is ~19 days. Steady state garadacimab exposure was achieved after the initial SC administration of loading dose of 400 mg (2 x 200 mg).

Specific populations.
Population PK analyses showed that age, gender, and race did not meaningfully influence the PK of garadacimab after correcting for body weight. Body weight was identified as an important covariate describing the variability of clearance and volume of distribution, resulting in higher exposure in lighter patients. However, this difference is not clinically relevant and no dose adjustments are recommended for any of these demographics.
Paediatric population.
Based on population PK analyses that included paediatric patients (12 to 17 years, N = 11), an approximately 30% higher predicted garadacimab exposure was found compared to adults. Age does not have a clinically meaningful impact on the PK of garadacimab and no dose adjustments for adolescent patients are recommended.
PK of garadacimab in paediatric patients below the age of 12 has not been investigated.
Elderly population.
Based on population PK analyses that included elderly patients (≥ 65 years, N = 13), age does not have a clinically meaningful impact on the PK of garadacimab and no dose adjustments are recommended.
Renal impairment.
No dedicated studies have been conducted to evaluate the PK of garadacimab in patients with renal impairment. Based on population PK analysis (estimated GFR: ≥ 90 mL/min/1.73 m2 [normal, N = 145], 60 to < 90 mL/min/1.73 m2 [mild, N = 26], and 30 to < 60 mL/min/1.73 m2 [moderate, N = 2]) renal impairment had no effect on the PK of garadacimab.
Hepatic impairment.
No dedicated hepatic impairment studies have been conducted with garadacimab and there are no data in patients with hepatic impairment from clinical studies. Clinically relevant differences in PK of garadacimab are not expected in patients with hepatic impairment.
Concomitant medications.
Based on population PK analyses, the use of analgesic, antibacterial, antihistamine, anti-inflammatory and anti-rheumatic medications had no effect on the PK of garadacimab.
Based on population PK analyses, for breakthrough HAE attacks, use of rescue medications such as plasma-derived or recombinant C1-INH, and icatibant had no effect on the PK of garadacimab.
5.3 Preclinical Safety Data
Genotoxicity.
Animal studies have not been conducted to evaluate the genotoxic potential of garadacimab. Garadacimab is not expected to interact directly with DNA or other chromosomal material due to its biological origin. As such, garadacimab has low potential for genotoxicity.
Carcinogenicity.
Animal studies have not been conducted to evaluate the carcinogenic potential of garadacimab. A carcinogenicity risk assessment based on the weight of evidence approach suggests a low risk for carcinogenicity.6 Pharmaceutical Particulars
6.1 List of Excipients
Histidine; arginine hydrochloride; proline; polysorbate 80; water for injections.
6.2 Incompatibilities
Not applicable.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store in a refrigerator (2°C to 8°C).
Do not freeze.
Keep the solution (pre-filled pen or pre-filled syringe with needle safety device) in the outer carton in order to protect from light.
The solution (pre-filled pen or pre-filled syringe with needle safety device) may be stored at room temperature up to 25°C for up to 2 months, but not beyond the expiry date. Do not return Andembry to refrigerated storage after storage at room temperature up to 25°C.
6.5 Nature and Contents of Container
Andembry 200 mg solution for injection in pre-filled pen.
1.2 mL of solution in a pre-filled glass syringe (type I glass) with a bromobutyl stopper, 27G x 1/2 5B special thin-walled (STW) staked needle. The pre-filled syringe is assembled with a pen. Andembry is available as unit packs containing 1 pre-filled pen and in multipacks containing 3 pre-filled pens.
Andembry 200 mg solution for injection in pre-filled syringe with needle safety device.
1.2 mL of solution in a pre-filled glass syringe (type I glass) with a bromobutyl stopper, 27G x 1/2 5B special thin-walled (STW) staked needle, and is assembled with an extended finger flange and needle safety device. Andembry is available as unit packs containing 1 assembled pre-filled syringe and in multipacks containing 3 pre-filled syringes.
Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Andembry is supplied as a sterile, preservative-free solution for SC administration. The solution of Andembry is slightly opalescent to clear, brownish-yellow to yellow liquid, with a pH of approximately 6.1. The solution has an osmolality of approximately 470 mOsm/kg.
Chemical structure.
Garadacimab has a molecular weight of approximately 148 kDa.
CAS number.
2162134-62-3.7 Medicine Schedule (Poisons Standard)
Schedule 4, Prescription Only Medicine.
Summary Table of Changes


 The following list (Table 2) of adverse reactions is based on experience from clinical trials and is displayed by system organ class and frequency: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000) and not known (cannot be estimated from the available data).
The following list (Table 2) of adverse reactions is based on experience from clinical trials and is displayed by system organ class and frequency: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000) and not known (cannot be estimated from the available data).
 The minimum response was defined as a ≥ 50% reduction in the time normalised number of HAE attacks during the treatment period vs run-in. The proportion of patients with ≥ 50% reduction (responders), ≥ 70% reduction and ≥ 90% reduction in HAE attack rates during the treatment period compared to run-in is presented in Table 4. 71.8% of patients treated with Andembry were attack-free throughout the first 3 months of treatment vs. 8.3% of patients treated with placebo. Over the entire 6 months of treatment, the percentage of attack-free patients was 61.5% in the Andembry arm compared with zero patients attack-free in the placebo arm.
The minimum response was defined as a ≥ 50% reduction in the time normalised number of HAE attacks during the treatment period vs run-in. The proportion of patients with ≥ 50% reduction (responders), ≥ 70% reduction and ≥ 90% reduction in HAE attack rates during the treatment period compared to run-in is presented in Table 4. 71.8% of patients treated with Andembry were attack-free throughout the first 3 months of treatment vs. 8.3% of patients treated with placebo. Over the entire 6 months of treatment, the percentage of attack-free patients was 61.5% in the Andembry arm compared with zero patients attack-free in the placebo arm. The time-normalised number of HAE attacks per month treated with on-demand medication and the number of moderate or severe HAE attacks were reduced with Andembry treatment compared with placebo (Table 5).
The time-normalised number of HAE attacks per month treated with on-demand medication and the number of moderate or severe HAE attacks were reduced with Andembry treatment compared with placebo (Table 5). The proportion of subjects who achieved an improvement in quality of life as measured by the Angioedema Quality of Life (AE-QoL) questionnaire (minimal clinically important difference ≥ 6 for the AE-QoL total score) was 87.9% for the Andembry arm and 55.0% for the placebo arm.
The proportion of subjects who achieved an improvement in quality of life as measured by the Angioedema Quality of Life (AE-QoL) questionnaire (minimal clinically important difference ≥ 6 for the AE-QoL total score) was 87.9% for the Andembry arm and 55.0% for the placebo arm.
