What is in this leaflet
This leaflet answers some common questions about Apidra.
It does not contain all the available information. It does not take the place of talking to your doctor, pharmacist or diabetes educator.
All medicines have risks and benefits. Your doctor has weighed the risks of you using Apidra against the benefits they expect it will have for you.
If you have any concerns about using this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Apidra is used for
Apidra is used to reduce high blood sugar (glucose) levels in people with diabetes mellitus.
Apidra is a modified insulin that is very similar to human insulin. It is a substitute for the insulin produced by the pancreas.
Apidra is a short-acting insulin. Your doctor may tell you to use a long-acting insulin in combination with Apidra.
Apidra is not addictive.
Ask your doctor if you have any questions about why Apidra has been prescribed for you.
Before you use Apidra
When you must not use Apidra
Do not use Apidra:
- If you have an allergy to:
- any medicine containing insulin
- any of the ingredients contained in Apidra listed at the end of this leaflet
Some of the symptoms of an allergic reaction may include:
- redness, swelling, rash and itching at the injection site
- rash, itching or hives on the skin
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- If you are experiencing low blood sugar levels (hypoglycaemia - a "hypo").
If you have a lot of hypos discuss appropriate treatment with your doctor.
- After the expiry date printed on the pack or if the packaging is torn or shows signs of tampering.
If you use Apidra after the expiry date has passed, it may not work as well. If it has expired or is damaged, return it to your pharmacist for disposal.
- If the product appears cloudy, discoloured or contains particles, or if the injection pen, cartridge or vial appears damaged.
If you are not sure whether you should start using this medicine, talk to your doctor.
Do not give Apidra to children less than 4 years of age. There is no experience with the use of Apidra in children less than 4 years.
Before you start to use Apidra
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- kidney problems
- liver problems
Tell your doctor if you are pregnant or plan to become pregnant. Pregnancy may make managing your diabetes more difficult.
Tell your doctor if you are breastfeeding or plan to breastfeed.
Tell your doctor if:
- you drink alcohol
- you do not eat regular meals
- you do a lot of exercise
- you are ill or feeling unwell
Alcohol, diet, exercise and your general health all affect the control of your diabetes.
If you have not told your doctor about any of the above, tell them before you start using Apidra.
Taking other medicines
Tell your doctor if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Medicines that may increase the blood sugar lowering effect of Apidra include:
- oral antidiabetic medicines that are used to treat type 2 diabetes
- blood pressure, blood flow, cholesterol and heart medications
- medications for pain and inflammation
- some antidepressants
- sulfonamide antibiotics
Medicines that may reduce the blood sugar lowering effect of Apidra include:
- corticosteroids, glucagon and other hormonal therapies
- oral contraceptives and gynaecological medications
- fluid and glaucoma medications
- tuberculosis and HIV/AIDS treatments
- some psychiatric medications
- adrenaline (epinephrine) and asthma medications such as salbutamol, terbutaline
Certain heart medications, especially beta-blockers, may mask the symptoms of hypoglycaemia.
Your doctor and pharmacist have a full list of medicines with which you must be careful or avoid while using Apidra. Please check with your doctor or pharmacist before starting any new medicines or over the counter products.
How to use Apidra
Your doctor, pharmacist or diabetes educator will have shown you how to use Apidra.
Carefully follow all the directions.
Do not inject Apidra into a vein. Apidra is intended for injection under the skin.
Any change in this medicine should be made cautiously and only under medical supervision.
If you do not understand the instructions, ask your doctor, pharmacist or diabetes educator for help.
How much to use
Your doctor will tell you how much Apidra you need to use each day. Your doctor may increase or decrease the dose, depending on your blood sugar levels.
It is very important that you manage your diabetes carefully. Too much or too little insulin can cause serious effects.
When to use Apidra
Your doctor will tell you when to use Apidra.
Apidra should be used within 15 minutes before or immediately after a meal.
How to use Apidra
ALWAYS CHECK YOUR APIDRA INJECTION PEN, CARTRIDGE OR VIAL.
Do not use Apidra if it is no longer clear and colourless or if it contains particles.
Make sure you are using the correct injection pen, cartridge or vial.
Always check the insulin label on the SoloStar pen, cartridge or vial before each injection to make sure you are using the right insulin.
Keep the injection pen, cartridge or vial at room temperature for 1 or 2 hours before use. Cold insulin is more painful to inject.
For Apidra injection pens or cartridges
PREPARING A DOSE FOR INJECTION
Always do a safety test before use. The safety test may highlight a problem with your injection pen. The safety test also removes any air bubbles and helps indicate whether or not a needle is bent or broken.
Becton Dickinson (BD Micro-Fine™+) needles should be used with injection pens.
Reusable pens
We recommend that Apidra cartridges only be used with the AllStar, AllStar Pro, JuniorStar or ClikStar reusable pen.
Carefully follow the instructions provided with the pen, for loading a cartridge, attaching a needle, performing a safety test and administering the insulin injection.
If the reusable injection pen does not work properly, Apidra may be withdrawn from the cartridge into a syringe. Ask your doctor, pharmacist or diabetes educator for help.
Pre-filled disposable pens
Apidra SoloStar disposable pens are pre-filled and ready for use. Once all the insulin is used, you cannot replace the cartridge.
Carefully follow the instructions provided with the Apidra SoloStar pen for attaching a needle, performing a safety test and administering the insulin injection.
Never use an injection pen if it is damaged or if you are not sure that it is working properly. Use a new pen.
INJECTING A DOSE
Apidra should be injected under the skin, being careful not to inject it into a muscle or vein.
Choose a site for injection.
Inject Apidra into the abdomen, thighs or upper arms.
- With one hand, stabilise the skin by spreading it or pinching up a large area, as recommended by your healthcare professional.
- Insert the needle into the skin as recommended by your healthcare professional.
- Inject the full dose of Apidra by pushing the plunger as far as it will go.
- Slowly count to 10 before removing the needle from the skin.
Change the place within the area you inject each day. This will reduce the risk of skin shrinking or thickening or lumps at the site (see "Side effects").
Do not use the exact same spot for each injection
Do not inject where the skin has pits, is thickened, or has lumps
Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin
If you develop skin changes at the injection site. The injection site should be rotated to prevent skin changes such as lumps under the skin. The insulin may not work very well if you inject into a lumpy area.
Contact your doctor if you are currently injecting into a lumpy area before you start injecting in a different area. Your doctor may tell you to check your blood sugar more closely, and to adjust your insulin or your other antidiabetic medications dose.
AFTER INJECTING
Using the outer needle cap, unscrew the needle and dispose of it safely into a sharps container.
Do not share needles, cartridges or injection devices. Do not reuse needles.
Leave the cartridge in the pen until it needs to be replaced.
Do not attempt to replace the cartridge in a pre-filled disposable pen. Empty disposable pens must never be reused and must be properly discarded.
For Apidra vials
PREPARING A DOSE FOR INJECTION
Using one insulin type (Apidra)
- Wash your hands.
- Draw air into a U100 insulin syringe equal to the dose of Apidra to be injected.
- Push the needle through the rubber top of the vial and inject the air into the vial.
- Leave the needle in the vial. Hold the vial and syringe firmly in one hand, with the vial above the syringe.
- Make sure the tip of the needle is in the insulin and withdraw the correct dose into the syringe.
- Before removing the needle from the vial, check the syringe for air bubbles. If bubbles are present, hold the syringe vertically (needle pointed upwards) and tap firmly until the bubbles float to the top. Push the bubbles out with the plunger and then withdraw the correct dose. Remove the needle from the vial.
- If you need to put the syringe down, make sure the needle does not touch anything.
Mixing Apidra with NPH (isophane) insulin
Only mix Apidra with another insulin if your doctor has instructed you to do so.
Do not mix Apidra with insulins other than NPH (Neutral Protamine Hagedorn) insulin.
Draw Apidra into the syringe first, then NPH insulin.
- Wash your hands
- Just before use, roll the vial of cloudy (longer acting) NPH insulin between your hands until the liquid is white and uniformly cloudy. Do not shake the vial.
- Draw air into a U100 insulin syringe equal to the dose of cloudy NPH insulin you are using. Put the needle through the rubber top of the NPH insulin vial and inject air into the vial. Withdraw the needle without drawing up any insulin.
- Draw air into the syringe equal to your dose of Apidra and inject it into the Apidra vial but do not withdraw the needle.
- Leave the needle in the vial. Hold the vial and syringe firmly in one hand, with the vial above the syringe.
- Make sure the tip of the needle is in the Apidra and withdraw the correct dose into the syringe.
- Pull the needle out of the Apidra vial. Expel any air from the syringe and check that the dose is correct.
- Insert the needle into the vial of cloudy NPH insulin.
- Leave the needle in the vial. Hold the vial and syringe firmly in one hand, with the vial above the syringe.
- Make sure the tip of the needle is in the insulin and withdraw your dose of cloudy NPH insulin.
- Remove the needle from the vial.
- If you need to put the syringe down for a brief moment, make sure the needle does not touch anything.
INJECTING A DOSE
Apidra should be injected under the skin, being careful not to inject it into a muscle or vein.
Choose a site for injection.
Inject Apidra into the abdomen, thighs or upper arms.
Apidra mixed with NPH insulin should be injected immediately after mixing.
- With one hand, stabilise the skin by spreading it or pinching up a large area, as recommended by your healthcare professional.
- Insert the needle into the skin as recommended by your healthcare professional.
- Inject the full dose of Apidra, or Apidra mixed with NPH insulin, by pushing the plunger as far as it will go.
- Slowly count to 10 before removing the needle from the skin.
Change the place within the area you inject each day. This will reduce the risk of skin shrinking or thickening or lumps at the site (see "Side effects").
Do not use the exact same spot for each injection
Do not inject where the skin has pits, is thickened, or has lumps
Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin
If you develop skin changes at the injection site. The injection site should be rotated to prevent skin changes such as lumps under the skin. The insulin may not work very well if you inject into a lumpy area.
Contact your doctor if you are currently injecting into a lumpy area before you start injecting in a different area. Your doctor may tell you to check your blood sugar more closely, and to adjust your insulin or your other antidiabetic medications dose.
AFTER INJECTING
Dispose of your insulin syringes safely into a sharps container.
Do not share vials, needles or syringes. Do not reuse needles.
How long to use Apidra
Continue using Apidra for as long as your doctor recommends.
Make sure you keep enough Apidra to last over weekends and holidays.
If you take too much (overdose) - Hypoglycaemia, a "Hypo"
If you accidentally use too much Apidra your blood sugar level may become too low (hypoglycaemia).
Immediately telephone your doctor or the Poisons Information Centre (13 11 26 in Australia; 0800 POISON or 0800 764 766 in New Zealand) if you think that you or anyone else may have used too much Apidra.
Do this even if there are no signs of discomfort or poisoning.
The risk of hypoglycaemia is increased if you:
- accidentally use too much Apidra
- have too much or unexpected exercise
- delay eating meals or snacks
- eat too little food
- are ill
The first symptoms of mild to moderate hypoglycaemia can come on suddenly. They may include:
- cold sweat, cool pale skin
- fatigue, drowsiness, unusual tiredness and weakness
- nervousness, anxious feeling, tremor, rapid heart beat
- confusion, difficulty concentrating
- excessive hunger
- vision changes
- headache, nausea
Always carry some sugary food or drink with you.
If you experience any of these symptoms of hypoglycaemia, you need to raise your blood sugar urgently. You can do this by taking one of the following:
- 5-7 jelly beans
- 3 teaspoons of sugar or honey
- 1/2 can of a sugar-containing soft drink (not a diet soft drink)
- 2-3 concentrated glucose tablets
Follow up with extra carbohydrates, e.g. plain biscuits, fruit or milk, when over the initial symptoms. Taking this extra carbohydrate will prevent a second drop in your blood sugar level.
If not treated quickly, the initial symptoms of hypoglycaemia may progress to loss of co-ordination, slurred speech, confusion, loss of consciousness and seizures.
If severe hypoglycaemia is not treated, it can cause brain damage and death.
Tell your relatives, friends, close workmates or carers that you have diabetes. It is important that they recognise the signs and symptoms of a "hypo".
Make sure they know to turn you on your side and get medical help immediately if you lose consciousness.
Make sure they know not to give you anything to eat or drink if you are unconscious. This is because you could choke.
Provide them with the telephone number for your doctor, the Poisons Information Centre (13 11 26 in Australia; 0800 POISON or 0800 764 766 in New Zealand) and Emergency Services.
An injection of the hormone glucagon may speed up recovery from unconsciousness. This can be given by a relative, friend, workmate or carer who knows how to give it.
If glucagon is used, have some sugary food or drink as soon as you are conscious again.
If you do not feel better after this, contact your doctor, diabetes educator, or the closest hospital.
If you do not respond to glucagon treatment, you will have to be treated in a hospital.
See your doctor if you keep having "hypos", or if you have ever become unconscious after using Apidra. Your dose of Apidra or other medicines may need to be changed.
If severe hypoglycaemia is not treated, it can cause brain damage and death.
If you miss a dose - Hyperglycaemia
If you forget to take your insulin dose check your blood sugar level as soon as possible. Your blood sugar level may become high (hyperglycaemia).
Do not take an extra dose at a later time just because you have missed a dose.
If you miss taking your dose at the regular scheduled time, taking a dose at another time can increase your risk of having a hypo. You should therefore plan in advance with your doctor or healthcare professional so that you know what to do in case you miss a dose.
If you have missed a dose and are not sure what to do, contact your doctor or healthcare professional for specific advice.
Do NOT use a double dose of your insulin. If you double a dose, this may cause low blood sugar levels.
The risk of hyperglycaemia is increased if you:
- miss doses of Apidra or other insulins, or use less Apidra than you need
- have uncontrolled diabetes
- exercise less than usual
- eat more carbohydrates than usual
- are ill or stressed
- take certain other medicines
High blood sugar levels over a period of time can lead to too much acid in the blood (diabetic ketoacidosis).
Contact your doctor immediately if your blood sugar level is very high or you experience any of the following symptoms.
Symptoms of mild to moderate hyperglycaemia include:
- drowsy feeling
- flushed face
- thirst, loss of appetite
- fruity odour on the breath
- blurred vision
- passing larger amounts of urine than usual
- getting up at night more often than usual to pass urine
- high levels of glucose and acetone in the urine
Symptoms of severe hyperglycaemia include:
- heavy breathing
- fast pulse
- nausea, vomiting
- dehydration
- loss of consciousness
Severe hyperglycaemia can lead to unconsciousness and, in extreme cases, death if untreated.
Discuss any worries you may have about this with your doctor, pharmacist or diabetes educator.
While you are using Apidra
Things you must do
Measure your blood sugar level regularly. This is the best way to tell if your diabetes is being controlled properly. Your doctor or diabetes educator will show you how and when to do this.
It is important to keep using Apidra even if you feel well.
Apidra helps to control your condition but does not cure it.
Tell your doctor if you often have hypoglycaemia or if you have ever become unconscious after using Apidra. Your doctor may need to adjust your dose of Apidra or of other medicines you are taking.
Always carry some sugary food or drink with you. If you experience any of the symptoms of hypoglycaemia, immediately eat some sugary food or have a drink, e.g. jelly beans, sugar, honey, sugar-containing soft drink, glucose tablets. Diet and low calorie soft drinks do NOT contain sugar and are unsuitable to take for hypoglycaemia.
Make sure that you tell every doctor, dentist, pharmacist or other healthcare professional who is treating you that you have diabetes and are using Apidra.
Tell your doctor, pharmacist or diabetes educator if you are travelling. Ask your doctor for a letter explaining why you are taking injecting pens and needles with you.
Each country you visit will need to see this letter, so you should take several copies.
You may need to inject Apidra and eat your meals at different times because of time differences in and between countries.
If you are travelling, it is a good idea to:
- wear some form of identification showing you have diabetes
- carry some form of sugar to treat hypoglycaemia if it occurs, e.g. sugar sachets or jelly beans
- carry emergency food rations in case of a delay, e.g. dried fruit, biscuits or muesli bars
- keep Apidra readily available; take enough Apidra for your expected needs whilst travelling - you may not be able to get Apidra in the country you are visiting
Your doctor, pharmacist or diabetes educator can provide you with some helpful information.
Tell your doctor if you are having trouble or difficulty with your eyesight.
Visit your doctor for regular checks of your eyes, feet, kidneys, heart, circulation, blood and blood pressure.
Carefully follow your doctor's and/or dietician's advice on diet, drinking alcohol and exercise.
Things you must not do
Do not stop using Apidra unless your doctor tells you to.
Do not skip meals while using Apidra.
Do not use Apidra if you think it has been frozen or exposed to excessive heat (temperatures above 25°C).
Do not re-use empty cartridges.
Do not give Apidra to anyone else, even if they have the same condition as you.
Things to be careful of
Be careful driving or operating machinery until you know how Apidra affects you. Be careful not to let your blood sugar levels fall too low.
Tell your doctor if you drink alcohol. Alcohol may mask the symptoms of hypoglycaemia.
Tell your doctor if you are ill. Illness, especially with nausea and vomiting, may cause your insulin needs to change. Even if you are not eating, you still require insulin. You and your doctor should design an insulin plan for those times when you are sick.
If you become sick with a cold or flu, it is very important to continue using Apidra, even if you feel unable to eat your normal meal. If you have trouble eating solid foods, use sugar-sweetened drinks as a carbohydrate substitute or eat small amounts of bland food. Your diabetes educator or dietician can give you a list of foods to use for sick days.
Tell your doctor if you are exercising more than usual. Exercise may lower your need for Apidra. Exercise may also speed up the effect of a dose of Apidra, especially if the exercise involves the area of the injection site (e.g. the leg should not be used for injection prior to jogging or running).
Tell your doctor if your diet changes. Changes in diet may cause your insulin needs to change.
Side effects
Tell your doctor, pharmacist or diabetes educator as soon as possible if you do not feel well while you are using Apidra.
Apidra helps most people with diabetes, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor, pharmacist or diabetes educator to answer any questions you may have.
The most common side effect when using insulin is low blood sugar levels (hypoglycaemia - a "hypo").
Tell your doctor if you notice any of the following and they worry you:
- hypoglycaemia (mild to moderate)
- redness, swelling or itching at the injection site; usually these symptoms disappear within a few weeks during continued use
- a depression or thickening of the skin around the injection site (lipodystrophy); this can often occur if you inject too often at the same injection site
The above list includes the more common side effects of your medicine. They are usually mild and short-lived.
Skin changes at the injection site:
If you inject insulin too often at the same place, the fatty tissue may either shrink (lipoatrophy) or thicken (lipohypertrophy). Lumps under the skin may also be caused by build-up of a protein called amyloid (localized cutaneous amyloidosis). The insulin may not work very well if you inject into a lumpy area. Change the injection site with each injection to help prevent these skin changes.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- More severe symptoms of hypoglycaemia, including:
- disorientation
- seizures, fits or convulsions
- loss of consciousness
- Signs of a serious allergic reaction, including:
- skin rashes over a large part of the body
- shortness of breath, wheezing
- swelling of the face, lips or tongue
- fast pulse
- sweating
The above list includes some very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare.
Tell your doctor if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
After using Apidra
Storage
All medicines should be kept where children cannot reach them.
CARTRIDGES
Keep unopened cartridges of Apidra in a refrigerator where the temperature is between 2-8°C. Do not allow it to freeze. Discard if frozen.
When the cartridge has been inserted in the injection pen, the cartridge-pen combination should not be put in the refrigerator and should be kept below 25°C. Do not leave it near heat or in direct light. Discard the cartridge within 28 days of first use. Cartridges that are first carried as a spare for a while must also be discarded 28 days after being removed from the refrigerator.
PRE-FILLED DISPOSABLE PENS
Before use, keep unopened Apidra pre-filled pens in a refrigerator where the temperature is between 2-8°C. Do not allow to freeze. Discard if frozen.
Before first use, store the pre-filled pen at room temperature for 1 to 2 hours. Once in use, the pre-filled pen should not be put in the refrigerator and it should be kept below 25°C. Do not leave it near heat or in direct light. Discard the pre-filled pen within 28 days of first use. Pre-filled pens that are first carried as a spare for a while must also be discarded 28 days after being removed from the refrigerator.
VIALS
Keep Apidra in a refrigerator where the temperature is between 2-8°C. Do not allow it to freeze. Discard if frozen.
Once opened, the vial should be refrigerated between 2-8°C, but may be kept unrefrigerated for up to 28 days as long as it is kept below 25°C. Do not leave it near heat or in direct light. Discard the vial within 28 days of first use. Vials that are first carried as a spare for a while must also be discarded 28 days after being removed from the refrigerator.
Disposal
Dispose of your needles and disposable injection devices safely into a sharps container.
If your doctor tells you to stop using Apidra or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
Apidra is a clear, colourless solution available in 3mL cartridges and 10mL vials.
Apidra SoloStar is a pre-filled disposable pen containing a 3mL cartridge of Apidra.
Ingredients
Active ingredient:
- insulin glulisine (100IU/mL)
Inactive ingredients:
- metacresol
- trometamol
- sodium chloride
- polysorbate 20
- hydrochloric acid
- sodium hydroxide
- water for injections
Supplier
Apidra is supplied in Australia by:
sanofi-aventis australia pty ltd
12-24 Talavera Road
Macquarie Park, NSW 2113
Freecall No: 1800 818 806
Apidra is supplied in New Zealand by:
sanofi-aventis new zealand limited
56 Cawley Street
Ellerslie, Auckland
Freecall No: 0800 283 684
®= Registered Trademark
Apidra SoloStar 3mL injector pen AUST R 132816
Apidra 3mL cartridge AUST R 99146
Apidra 10mL vial AUST R 99145
Full Product Information is available from sanofi-aventis australia pty ltd at http://products.sanofi.com.au/ aus_pi_apidra.pdf or by calling 1800 818 806 (in Australia) or 0800 283 684 (New Zealand).
This leaflet was prepared in June 2021.
Further information
You can get more information about diabetes and insulin from:
- Diabetes Australia: Freecall helpline 1300 136 588
www.diabetesaustralia.com.au - Diabetes NZ: 0800 369 636
www.diabetes.org.nz
apidra-ccdsv12-cmiv13-10jun21
Published by MIMS July 2025

 The following related adverse reactions from clinical investigations were listed below by system organ class and in order of decreasing incidence (very common: > 1/10; common > 1/100, < 1/10; uncommon: > 1/1,000, < 1/100; rare: > 1/10,000, < 1/1,000; very rare: < 1/10,000).
The following related adverse reactions from clinical investigations were listed below by system organ class and in order of decreasing incidence (very common: > 1/10; common > 1/100, < 1/10; uncommon: > 1/1,000, < 1/100; rare: > 1/10,000, < 1/1,000; very rare: < 1/10,000).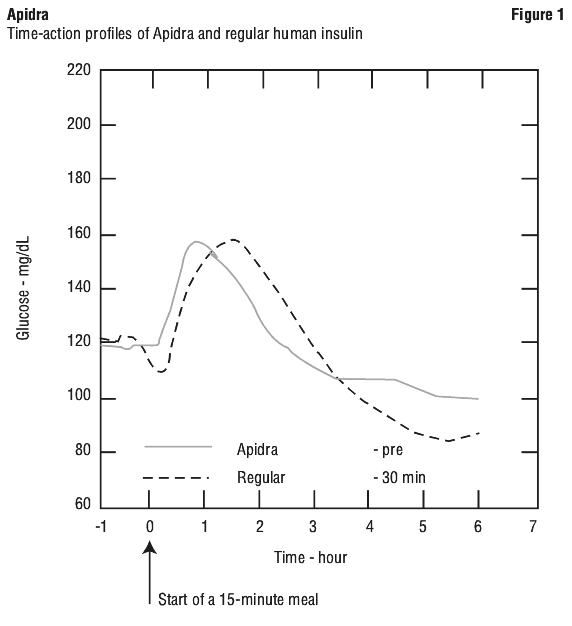






 When Apidra was injected subcutaneously into different areas of the body, the time concentration profiles were similar with a slightly faster absorption when administered in the abdomen compared to the deltoid or thigh. The absolute bioavailability of insulin glulisine after subcutaneous administration is about 70%, regardless of injection area (abdomen 73%, deltoid 71%, thigh 68%).
When Apidra was injected subcutaneously into different areas of the body, the time concentration profiles were similar with a slightly faster absorption when administered in the abdomen compared to the deltoid or thigh. The absolute bioavailability of insulin glulisine after subcutaneous administration is about 70%, regardless of injection area (abdomen 73%, deltoid 71%, thigh 68%).

