What is in this leaflet
This leaflet answers some common questions about azathioprine. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using this medicine against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may want to read it again.
What this medicine is used for
Azathioprine is used to help prevent the body from rejecting transplanted organs, such as a heart or kidney.
It is also used to treat autoimmune diseases, where your immune system is reacting against your own body. These include:
- severe rheumatoid arthritis
- systemic lupus erythematosus
- chronic active hepatitis
- certain skin, muscle, and blood diseases.
Azathioprine belongs to a group of medicines called immunosuppressants. It works by supressing the body's immune defence system. Azathioprine is usually taken in combination with other medicines such as corticosteroids or other immunosuppressants.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed this medicine for another reason.
This medicine is available only with a doctor's prescription.
This medicine is not addictive.
Before you take this medicine
When you must not take it
Do not take this medicine if you have an allergy to:
- azathioprine
- 6-mercaptopurine
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue, throat or other parts of the body
- rash, itching or hives on the skin
Do not take this medicine if you have rheumatoid arthritis previously treated with alkylating agents (medicines such as chlorambucil, melphalan or cyclophosphamide).
Do not take this medicine if you are pregnant, plan to become pregnant or intending to father a child. Azathioprine may cause birth defects if either male or female is taking it at the time of conception. Both you and your partner should take adequate contraceptive precautions while taking azathioprine to prevent pregnancy.
Do not take this medicine if you are breastfeeding, or plan to breastfeed. Azathioprine is not recommended for use during breastfeeding as it passes into breastmilk and may affect your baby.
Do not take this medicine after the expiry date printed on the pack or the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- liver or kidney disease
- a condition where your body produces too little of a natural chemical called thiopurine methyltransferase (TPMT)
- Lesch-Nyhan Syndrome
- chickenpox or shingles
- hepatitis B
- irritable bowel disease
- a history of cytomegalovirus disease.
Tell your doctor if you are pregnant, may be pregnant, plan to become pregnant or intending to father a child. You or your partner should take adequate contraceptive precautions while you are taking azathioprine to prevent pregnancy.
Tell your doctor if you have recently been vaccinated or immunised or plan to get a vaccination or immunisation. Azathioprine may affect the way the vaccine works or your reaction to the vaccine.
Tell your doctor if you are planning to have surgery or an anaesthetic.
Tell your doctor if you are currently receiving or are planning to receive dental treatment. Tell your dentist that you are taking azathioprine. Dental work, whenever possible, should be completed before you start taking azathioprine or delayed until your blood cell counts are normal.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and azathioprine may interfere with each other. These include:
- penicillamine, used in the treatment of rheumatoid arthritis
- captopril, used in high blood pressure and heart failure
- cimetidine, used in stomach ulcers and indigestion
- indomethacin, used as a painkiller and anti-inflammatory
- co-trimoxazole, ketoconazole, erythromycin and rifampicin, used to treat infections
- allopurinol, oxipurinol, thiopurinol or febuxostat, used mainly to treat gout
- tubocurarine and succinylcholine, used during anaesthesia
- frusemide, may be used to reduce swelling caused by excess fluid
- warfarin, used to prevent blood clots
- mesalazine, olsalazine or sulphasalazine, used in the treatment of ulcerative colitis
- phenytoin, phenobarbital, rifampicin, ketoconazole or erythromycin
- methotrexate, used in the treatment of cancer
- ribavirin, used to treat a type of respiratory infection
- infliximab, used to treat autoimmune diseases.
These medicines may be affected by azathioprine or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
Other medicines not listed above may also interact with azathioprine.
How to take this medicine
Follow all directions given to you by your doctor or pharmacist carefully. They may differ to the information contained in this leaflet.
If you do not understand the instructions on the label, ask your doctor or pharmacist for help.
How much to take
Your doctor will tell you how much of this medicine you should take. This will depend on your condition and whether you are taking any other medicines.
How to take it
Swallow the tablets whole with a glass of water.
Do not break, chew or crush the tablets.
When to take it
Take this medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
Take this medicine at least one hour before or three hours after food or milk. Food can interfere with the absorption of this medicine.
How long to take it for
Continue taking your medicine for as long as your doctor tells you.
Patients with a transplant will need to take azathioprine continuously to reduce the risk of organ rejection.
For other conditions, your doctor will discuss with you how long you need to take this medicine. It could take some weeks or months for it to take full effect.
Make sure you have enough to last over weekends and holidays.
If you forget to take it
If it is almost time to take your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for missed doses. This may increase the chance of you experiencing side effects.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints to help you remember.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice or go to Accident and Emergency at your nearest hospital, if you think that you or anyone else may have taken too much of this medicine.
Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
If you take too much azathioprine, you may get an unexpected infection, ulcers in the throat, bruising and bleeding.
While you are taking this medicine
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking this medicine.
Tell any other doctors, dentists and pharmacists who are treating you that you take this medicine.
If you become pregnant, father a child or start to breastfeed while taking this medicine, tell your doctor immediately.
Tell your doctor if you plan to have any vaccinations or immunisations. This medicine may affect the way some vaccines work, or your reaction to the vaccine.
Tell your doctor if you are about to have any blood tests.
Tell your doctor if you are going to have surgery or an anaesthetic or are going into hospital.
Keep all your doctor's appointments so that your progress can be checked. Your doctor will perform blood tests every week for the first eight weeks, then at least once a month after that, while you are taking azathioprine.
Before you start azathioprine, your doctor should also test for thiopurine methyltransferase (TPMT) enzyme deficiency.
Try to avoid contact with people who have infectious diseases, such as the flu, chickenpox or shingles. Tell your doctor immediately if you do come into contact with someone who has chickenpox or shingles.
Avoid contact sports or other situations where bruising or injury may occur. Be careful to avoid cutting yourself with sharp objects (e.g. razors).
Protect yourself from the sun while you are taking azathioprine. If you go out in the sun, wear a hat, protective clothing and use sunscreen.
Tell your doctor immediately if you notice new moles, changes in existing moles, lumps on your body or you feel unwell. Azathioprine suppresses your immune system. Lowering your body's immune defence system increases your risk of skin cancer, cervical cancer, lymphoma and other cancers.
If you are female, tell your doctor if you notice unusual vaginal discharge or bleeding, and make sure to have regular Pap smears.
Things you must not do
Do not give this medicine to anyone else, even if they have the same condition as you.
Do not take your medicine to treat any other complaints unless your doctor tells you to.
Do not stop taking your medicine or change the dosage without first checking with your doctor. If you stop taking it suddenly, your condition may worsen.
Things to be careful of
Be careful when driving or operating machinery until you know how this medicine affects you. Azathioprine may cause dizziness and tiredness in some people. If you have any of these symptoms, do not drive, or operate machinery or do anything else that is dangerous.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking azathioprine.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor as soon as possible if you notice any of the following:
- any infection or fever
- unexpected bruising or bleeding, black stools or blood in the urine or stools
- you come into contact with anyone who is suffering from chickenpox or shingles
- new marks on skin or any change to marks that may have been there previously
- headache, stiff neck and extreme sensitivity to bright light
- nausea and vomiting
- tiredness, dizziness or generally unwell
- irregular heart beat
- sores in the mouth and on the lips
- feeling of ants creeping in or under the skin
- change in sense of smell or taste.
If any of the happen, stop taking your medicine and contact your doctor immediately or go to the Accident and Emergency department at your nearest hospital:
- muscle weakness, pain or stiffness
- severe joint pain
- kidney problems
- feeling faint especially when standing up
- severe abdominal pain
- diarrhoea
- jaundice, a yellow discoloration of the skin/eyes
- serious skin reactions such as blistering or peeling
- muscle weakness, with or without a skin rash
- a rash that appears as tender red or purple lumps or patches on the skin that may ulcerate, tiredness, lack of energy and feeling unwell, fever, acing joints and muscles, mouth ulcers and sore red eyes – symptoms of Sweet's syndrome
- symptoms of an allergic reaction including cough, shortness of breath, wheezing or difficulty breathing; swelling of the face, lips, tongue, throat or other parts of the body; rash, itching or hives on the skin
Side-effects reported particularly in organ transplant patients are:
- viral, fungal and bacterial infections
- hair loss (particularly following a kidney transplant)
- diarrhoea, usually with blood and mucus
- stomach pain with fever and vomiting.
The above list includes very serious side effects and you may need urgent medical attention or hospitalisation.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may occur in some patients.
Some of these side effects can only be found when your doctor does tests from time to time to check your progress (e.g. low blood cell count).
Storage and disposal
Storage
Keep your medicine in the pack until it is time to take it. If you take your medicine out of the pack it may not keep well.
Keep your medicine in a cool dry place where the temperature stays below 25°C.
Do not store your medicine or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep this medicine where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
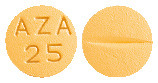
25 mg tablets
Orange coloured, round, biconvex film coated tablet with "AZA 25" embossed on one side and break line on other side. AUST R 205759.
50 mg tablets
Pale yellow coloured, round, biconvex film coated tablet with "AZA 50" embossed on one side and break line on other side. AUST R 205762
Available in packs of 100 tablets.
* Not all strengths, pack types and/or pack sizes may be available.
Ingredients
Each tablet contains 25 or 50 mg of azathioprine as the active ingredient.
It also contains the following:
- lactose monohydrate
- microcrystalline cellulose
- maize starch
- magnesium stearate
- purified water
- Opadry complete film coating system 03B52231 yellow (25 mg tablet only)
- Opadry clear YS-1R-7006 (50 mg tablet only).
This medicine contains sugars as lactose.
This medicine does not contain gluten, sucrose, tartrazine and other azo dyes.
Sponsor
Apotex Pty Ltd
16 Giffnock Avenue
Macquarie Park NSW 2113
Australia
Tel: (02) 8877 8333
Web: www1.apotex.com/au
APO and APOTEX are registered trademarks of Apotex Inc.
This leaflet was prepared in December 2021.
Published by MIMS January 2022


 Chemical name: 6-[(1-methyl-4-nitro-1H-imidazol-5-yl)sulfanyl]-7-H-purine.
Chemical name: 6-[(1-methyl-4-nitro-1H-imidazol-5-yl)sulfanyl]-7-H-purine.