What is in this leaflet
This leaflet answers some common questions about bicalutamide. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using this medicine against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may want to read it again.
What this medicine is used for
Bicalutamide is used in combination with other medicines called LHRH agonists to treat advanced prostate cancer and to prevent a side effect of LHRH agonists.
How it works
Bicalutamide is an anti-androgen medicine. Androgens such as testosterone are natural male sex hormones. In some types of prostate cancer, androgens may help the cancer cells to grow.
Bicalutamide interferes with some of the actions of these hormones.
Bicalutamide should only be taken by men.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed this medicine for another reason.
This medicine is available only with a doctor's prescription.
This medicine is not addictive.
This medicine must not be used in children.
Before you take this medicine
When you must not take it
Do not take this medicine if you have an allergy to:
- bicalutamide
- any of the ingredients listed at the end of this leaflet
- lactose
Some of the symptoms of an allergic reaction may include:
- cough, shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue, throat or other parts of the body
- rash, itching or hives on the skin
- fainting or hayfever-like symptoms.
Do not take this medicine if you are taking cisapride, terfenadine or astemizole
Do not take this medicine if you are female. Women are not treated with this medicine, as it could cause major defects in unborn children if taken by pregnant women, or harm to infants if taken when breastfeeding.
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- liver problems
- diabetes
- heart conditions, including heart rhythm problems (arrhythmia)
Tell your doctor if you are planning to start a family, as this medicine may affect your fertility.
If you have not told your doctor about any of the above, tell them before you start taking this medicine.
Taking other medicines
Tell your doctor and pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interact with bicalutamide. These include:
- cisapride
- terfenadine, astemizole
- medicines used to prevent blood clots, especially warfarin
- cimetidine, used for stomach problems
- ketoconazole, used to treat fungal infections
- midazolam or carbamazepine, used for treating seizures
- cyclosporin, used after organ transplants
- statins used to treat high cholesterol (e.g. simvastatin, atorvastatin, pravastatin, rosuvastatin, simvastatin)
- calcium channel blockers used to treat high blood pressure (e.g. felodipine, nifedipine, amlodipine)
- quinidine, used to treat certain heart problems
- some medicines used to treat viral infections (e.g. ritonavir, saquinavir)
- other medicines which interfere with the liver's CYP450 enzyme system
If you are taking any of these you may need a different dose or you may need to take different medicines.
Other medicines not listed above may also interact with bicalutamide.
How to take this medicine
Follow carefully all directions given to you by your doctor or pharmacist.
They may differ to the information contained in this leaflet.
How much to take
The usual dose is one tablet taken each day. This dose may be reduced if you have severe liver problems.
How to take it
Swallow your tablet whole with a full glass of water.
When to take it
Take this medicine at about the same time each day. Taking it at the same time each day will have the best effect and will also help you remember when to take it. It does not matter if you take it with or without food.
How long to take it for
Continue taking your medicine for as long as your doctor tells you. Make sure you have enough to last over weekends and holidays.
If you forget to take it
If it is almost time to take your next dose (i.e. less than 12 hours to the next dose), skip the missed dose and take your next dose at the usual time.
Otherwise take it as soon as you remember and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for missed doses. This may increase the chance of unwanted side effects.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice or go to the Emergency Department at your nearest hospital if you think that you or anyone else may have taken too much of this medicine. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are taking this medicine
Things you must do
Tell any other doctors, dentists and pharmacists who are treating you that you take this medicine.
If you are about to be started on any new medicine, tell your doctor that you are taking this medicine.
You will need to take another prostate cancer medicine (an LHRH agonist) whilst you are taking bicalutamide. The two medicines need to work together to have an effect.
Keep all your doctor’s appointments, as your doctor may do tests to make sure the medicine is working and to prevent side effects.
Things you must not do
Do not give this medicine to anyone else, even if they have the same condition as you.
Do not take your medicine to treat any other complaint unless your doctor or pharmacist tells you to.
Do not stop taking your medicine, or change the dosage, without first checking with your doctor.
Things to be careful of
Be careful when driving or operating machinery until you know how this medicine affects you. Some people may feel sleepy, dizzy or weak when taking this medicine.
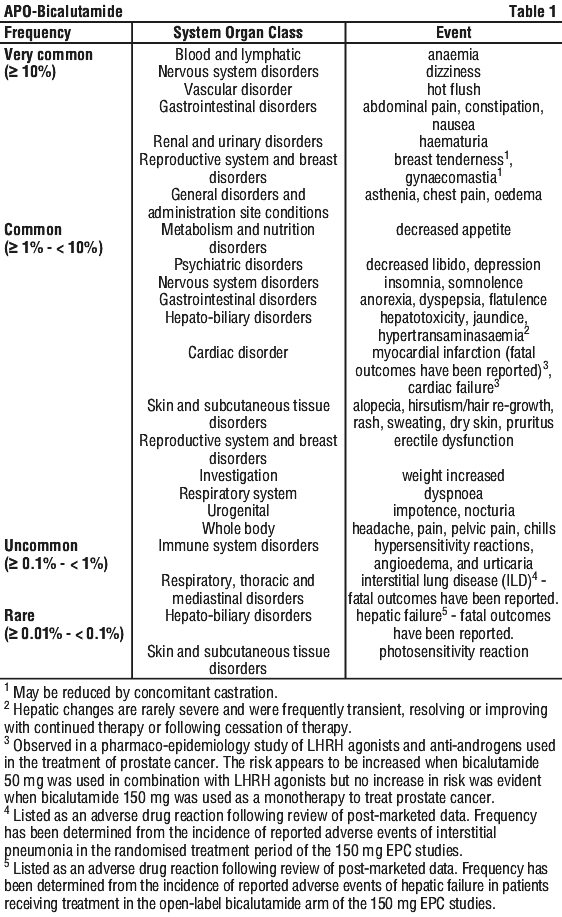
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking bicalutamide.
All medicines can have side effects. Sometimes they are serious but most of the time they are not.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Tell your doctor or pharmacist if you notice any of the following:
- hot flushes or sweating
- breast tenderness or swelling
- mild rash, itching or dry skin
- increased hairiness, or loss of hair
- stomach upsets, such as pain, indigestion, nausea, vomiting, diarrhoea, constipation, wind
- dry mouth
- loss of appetite, weight changes
- depression
- unusual tiredness, weakness, problems sleeping, feeling sleepy
- dizziness or light-headedness
- headache
- chills
- pelvic pain
- decrease in your sexual drive, inability to get or maintain an erection
Tell your doctor as soon as possible if you notice any of the following:
- frequent urination (including at night) blood in the urine
- becoming out of breath and dizzy when exercising, looking pale (anaemia)
- excessive thirst with weight loss, passing large amounts of urine, sweet smelling breath
The above list includes serious side effects that may need medical attention.
If you experience any of the following, stop taking your medicine and contact your doctor immediately or go to the Emergency department at your nearest hospital:
- chest pain, with or without a feeling of tightness radiating to the shoulders back, neck jaw or arms, with sweating, chills, nausea, vomiting and paleness
- yellowing of the skin or eyes, and dark coloured urine (jaundice)
- cough, shortness of breath, wheezing or difficulty breathing; swelling of the face, lips, tongue, or other parts of the body; rash, itching or hives on the skin; fainting; hayfever-like symptoms (signs of an allergic reaction)
- serious breathlessness or sudden worsening of breathlessness, possibly with a cough or fever
The above list includes very serious side effects and you may need urgent medical attention or hospitalisation.
Other side effects not listed above may occur in some patients.
Storage and disposal
Storage
Keep your medicine in its original packaging until it is time to take it. If you take your medicine out of its pack it may not keep well.
Keep your medicine in a cool dry place where the temperature will stay below 25°C.
Do not store your medicine, or any other medicine, in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep this medicine where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine left over.
Product description
What it looks like
White to off white, round, biconvex, film coated tablets debossed "B50" on one side and plain on other side. Available in blister packs of 28 tablets. AUST R 194683
Ingredients
Each tablet contains 50 mg of bicalutamide as the active ingredient.
It also contains the following:
- lactose monohydrate
- sodium starch glycollate type A
- povidone
- magnesium stearate
- hypromellose
- macrogol 400
- titanium dioxide
This medicine does not contain gluten, sucrose, tartrazine or any other azo dyes.
Sponsor
Arrotex Pharmaceuticals Pty Ltd
15 – 17 Chapel Street
Cremorne VIC 3121
www.arrotex.com.au
This leaflet was prepared in January 2024.
Published by MIMS March 2024


 Chemical name: (RS)-4'-cyano-α', α', α',-trifluoro-3-(4-fluorophenylsulphonyl)-2- hydroxy-2-methylpropiono-m-toluidide.
Chemical name: (RS)-4'-cyano-α', α', α',-trifluoro-3-(4-fluorophenylsulphonyl)-2- hydroxy-2-methylpropiono-m-toluidide.