What is in this leaflet
This leaflet answers some common questions about APO-Glimepiride. It does not contain all of the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking this medicine against the benefits this medicine is expected to have for you.
Ask your doctor or pharmacist if you have any concerns about taking this medicine.
Keep this leaflet with the medicine.
You may need to read it again.
What this medicine is used for
The name of your medicine is APO-Glimepiride. It contains the active ingredient glimepiride.
This type of diabetes is also known as non-insulin-dependent diabetes mellitus (NIDDM) or maturity onset diabetes.
Glimepiride is used when diet and exercise are not enough to control your blood glucose.
Glimepiride can be used alone, or together with insulin or other medicines for treating diabetes.
How it works
Glimepiride belongs to a group of medicines called sulphonylureas.
Glimepiride lowers high blood glucose by increasing the amount of insulin produced by your pancreas.
If your blood glucose is not properly controlled, you may experience hypoglycaemia (low blood glucose) or hyperglycaemia (high blood glucose).
Hypoglycaemia (low blood glucose) can occur suddenly. Signs may include:
- Weakness, trembling or shaking
- Sweating
- Lightheadedness, dizziness, headache or lack of concentration
- Tearfulness or crying
- Irritability
- Hunger
- Numbness around the lips and tongue
If not treated promptly, these may progress to:
- Loss of co-ordination
- Slurred speech
- Confusion
- Loss of consciousness or seizures
Hyperglycaemia (high blood glucose) usually occurs more slowly than low blood glucose. Signs of high blood glucose may include:
- Lethargy or tiredness
- Headache
- Thirst
- Passing large amounts of urine
- Blurred vision
High blood glucose can lead to serious problems with your heart, eyes, circulation or kidneys.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
This medicine is available only with a doctor's prescription.
There is no evidence that this medicine is addictive.
Before you take this medicine
When you must not take it
Do not take this medicine if you have had an allergic reaction to:
- glimepiride or other sulfonylureas
- antibiotics called sulphonamides
- thiazide diuretics (a type of fluid or water tablets)
- lactose (these tablets contain lactose)
- any of the ingredients listed at the end of this leaflet
Symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue, throat or other parts of the body
- muscle pain or tenderness or joint pain or rash
- itching or hives on the skin.
Check with your doctor if you are not sure if you have any of the above.
Do not take this medicine if you have, or have had any of the following conditions:
- type 1 diabetes mellitus (insulin dependent diabetes mellitus, also known as IDDM, or juvenile or growth onset diabetes)
- a history of ketoacidosis
- unstable diabetes
- diabetic ketoacidosis
- diabetic coma or pre-coma
- severe kidney disease or undergoing dialysis
- severe liver disease
- Glucose-6-phosphate dehydrogenase (G6PD) deficiency
Do not take this medicine if you are pregnant or plan to become pregnant.
Glimepiride may affect your developing baby if you take it during pregnancy. Insulin is more suitable for controlling blood glucose during pregnancy. Your doctor will usually replace glimepiride with insulin while you are pregnant.
Do not take this medicine of you are breast-feeding or planning to breast-feed.
Glimepiride can pass into the breast milk and harm your baby.
Do not give this medicine to children.
There is not enough experience with the use of glimepiride in children.
Do not take this medicine past the expiry date (EXP) printed on the pack.
If you take this medicine after the expiry it may not work as well.
Do not take this medicine if the packaging is torn, shows signs of tampering or if it does not look quite right.
If it has expired or is damaged, return it to your pharmacist for disposal.
Talk to your doctor or pharmacist if you are not sure whether you should start taking this medicine.
Before you start to take it
Tell your doctor if:
- You have allergies to:
- any other medicines
- lactose
- any other substances such as food, preservatives, or dyes - You have or have had any medical conditions, especially the following:
- liver problems
- kidney problems
- a deficiency in the enzyme in your body called glucose-6-phosphate dehydrogenase (G6PD)
- a history of diabetic coma
- adrenal, pituitary or thyroid problems
- heart failure - You drink alcohol in any amount
- You do not eat regular meals
- You do a lot of exercise or you do heavy exercise or work
- You are ill or feeling unwell
- You plan to become pregnant of breast-feed
- You are taking any other anti-diabetic treatments
Alcohol, diet, exercise, and your general health all strongly affect the control of your diabetes.
If you have not told you doctor, pharmacist, of diabetes educator about any of the above, tell them before you start taking this medicine.
Taking other medicines
Tell your doctor if you are taking any other medicines, including medicines that you buy without a prescription from a pharmacy, supermarket or health food shop.
Some medicines may interfere with glimepiride. These include:
- other medicines used to treat diabetes (tablets and insulin)
- some medicines used to treat high blood pressure or heart conditions, eg. beta blockers, ACE inhibitors, calcium channel blockers, amiodarone, disopyramide, reserpine or guanethidine
- some hormones used in hormone replacement therapy and oral contraceptives (oestrogen and progestogens)
- monoamine oxidase inhibitors (MAOIs), used for treating depression, Parkinson's Disease or infections
- some medicines used for mental illness (eg. phenothiazines)
- barbiturates, used for epilepsy and sedation during anaesthetics
- phenytoin, used for epilepsy
- medicines for treating high cholesterol / blood fats
- some medicines used to treat arthritis, pain and inflammation (diclofenac, naproxen, ibuprofen, azapropazone, fenyramidol, oxyphenbutazone, phenylbutazone, salicylates)
- antibiotics called sulfonamides, quinolones, sulfinpyrazone, tetracyclines, rifampicin, isoniazid, clarithromycin or chloramphenicol
- miconazole, or fluconazole, used to treat fungal infections
- some medicines used to prevent or treat blood clots (warfarin and similar medicines)
- cimetidine, famotidine, nizatidine and ranitidine, used to treat acid reflux and stomach ulcers
- medicines called corticosteroids (e.g. prednisolone, cortisone)
- anabolic steroids, male sex hormones
- thyroid hormones, used to treat thyroid problems
- oxpentifylline used to prevent or treat blood vessel problems
- clonidine, used for high blood pressure or migraine
- diuretics, also known as fluid tablets (e.g. chlorothiazide)
- diazoxide, used mainly for treating very high blood pressure
- acetazolamide, used to treat glaucoma, epilepsy and oedema (swelling due to fluid)
- some antidepressants
- weight reduction medicines
- tritoqualine, an antihistamine
- trofosfamide, cyclophosphamide, ifosfamide, used for treating certain cancers
- laxatives (long-term use)
- probenecid, used for treating gout
- glucagon, used to help balance blood sugar levels
- medicines used in asthma medicines and cold remedies.
These medicines may be affected by glimepiride or may affect how well it works. This may result in levels of blood sugar which are too high or too low.
In addition, alcohol, certain heart medications such as beta- blockers, clonidine, guanethidine or reserpine, may hide the symptoms of low blood glucose (hypoglycaemia).
You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist can tell you if you are taking any of these medicines. They may also have more information on medicines to be careful with or avoid while taking glimepiride.
Drinking alcohol can also affect your blood sugar levels and how well glimepiride works.
Other interactions not listed above may also occur.
Ask your doctor or pharmacist if you are not sure if you are taking any of these medicines.
How to take this medicine
Follow all directions given to you by your doctor, pharmacist or diabetes educator carefully.
They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How much to take
Your doctor or pharmacist will tell you how many tablets you will need to take. This depends on your condition and whether or not you are taking any other medicines. Your doctor may increase or decrease the dose, depending on your blood glucose levels.
How to take it
Swallow the tablets whole with a glass of water. Do not chew tablets.
When to take it
Take glimepiride immediately before or with a meal.
Glimepiride tablets are usually taken once a day, immediately before breakfast. If you only eat a light breakfast, you should delay taking the tablet until the first main meal of the day (e.g. lunch).
Do not skip meals while taking glimepiride tablets.
Take it at about the same time each day.
It will also help you remember when to take them.
It is important that you eat regular meals.
How long to take it for
Continue taking your medicine for as long as your doctor tells you.
Glimepiride will help control your Type 2 diabetes but will not cure it. Most people will need to take glimepiride for long periods of time.
Make sure you have enough to last over weekends and holidays.
Do not skip meals while taking glimepiride tablets.
Take it at about the same time each day.
It will also help you remember when to take them.
It is important that you eat regular meals.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember (immediately before food), and then go back to taking it as you would normally.
Skipping a dose may result in hyperglycaemia. If you experience any symptoms of hyperglycaemia, contact your doctor immediately.
If you are not sure whether to skip the dose, talk to your doctor or pharmacist.
Do not take a double dose to make up for the dose that you missed.
If you double dose this may cause low blood glucose (hypoglycaemia).
Ask you pharmacist for some hints if you have trouble remembering to take your medicine.
If you take too much (overdose)
If you think that you or anyone else may have taken too much of this medicine, immediately telephone your doctor or the Poisons Information Centre (Tel: 13 11 26 in Australia) for advice. Alternatively, go to the Accident and Emergency department at your nearest hospital. Also report any other medicine or alcohol which has been taken.
Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
If you take too much glimepiride, you may experience symptoms of hypoglycaemia (low blood glucose).
If not treated quickly, these symptoms may progress to loss of co-ordination, slurred speech, confusion, loss of consciousness and fitting.
At the first signs of hypoglycaemia, raise your blood glucose quickly by eating jelly beans, sugar or honey, drinking non-diet soft drink or taking glucose tablets.
While you are taking this medicine
Things you must do
Take your tablets exactly as your doctor has prescribed.
Tell all doctors, dentists and pharmacists who are treating you that you are taking glimepiride.
If you plan to have surgery that needs a general anaesthetic, tell your doctor or dentist that you are taking glimepiride.
Tell your doctor immediately if you become pregnant.
Tell your doctor if you feel APO-Glimepiride is not helping your condition.
Tell your doctor that you are taking this medicine if you are about to have any blood tests.
Tell your doctor and pharmacist that you are taking glimepiride if you are about to start taking any new medicines.
Tell all doctors, dentists, pharmacists and diabetes educators who are involved with your treatment that you are taking this medicine.
Make sure you, your friends, family and work colleagues can recognise the symptoms of hypoglycaemia (low blood glucose) and hyperglycaemia (high blood glucose) and know what to do. Provide them with the telephone number for your doctor, the Poisons Information Centre (13 11 26 in Australia).
Always carry some sugary food or drink with you.
Tell your doctor immediately if you notice the return of any symptoms of hyperglycaemia that you had before starting glimepiride, or if your blood sugar levels are high.
These may be signs that glimepiride is no longer working, even though you may have been taking it successfully for some time.
If you are elderly or are taking other medicines for diabetes (e.g. insulin or metformin), the risk of hypoglycaemia (low blood sugar) is increased.
The risk of hypoglycaemia is also increased in the following situations:
- too much glimepiride
- too much unexpected exercise
- delayed meal or snack
- too little food
If you experience any of the signs of hyperglycaemia (high blood glucose), contact your doctor immediately.
The risk of hyperglycaemia is increased in the following situations:
- undiagnosed or uncontrolled diabetes
- illness, infection or stress
- too little glimepiride
- taking certain medicines
- too little exercise
- eating more carbohydrates than normal
- sudden immobilisation, e.g. after an accident
Tell your doctor if you:
- become ill
- become dehydrated
- are excessively stressed
- are injured
- have a fever
- have serious infection
- are having surgery.
Your blood glucose may become difficult to control at these times. Your doctor may decide to replace glimepiride with insulin.
Visit your doctor for regular blood tests and checks of your eyes, feet, kidneys, heart, circulation, blood, and blood pressure.
Make sure you check your blood glucose levels regularly.
This is the best way to tell if your diabetes is being controlled properly. You doctor of diabetes educator will show you how and when to do this.
Carefully follow your doctor's and dietician's advice on diet, drinking alcohol and exercise.
Things you must not do
Do not skip meals while taking glimepiride.
Do not give this medicine to anyone else, even if their symptoms seem similar to yours.
Do not stop taking your medicine, or change the dosage, without checking with your doctor.
Do not take your medicine to treat any other complaints unless your doctor or pharmacist tells you to.
Things to be careful of
Protect your skin when you are outdoors or in the sun, especially between 10 am and 3 pm, wear protective clothing and use a 15+ sunscreen. If your skin appears to be burning, tell your doctor immediately.
Glimepiride may cause your skin to be more sensitive to sunlight than it is normally. Exposure to sunlight may cause skin rash, itching, redness or severe sunburn.
Be careful while driving or operating machinery until you know how glimepiride affects you.
If you have to be alert, e.g. when driving, be especially careful not to let your blood glucose levels fall too low.
Low blood glucose levels may slow your reaction time and affect your ability to drive or operate machinery. Drinking alcohol can make this worse. Your vision may also be temporarily affected.
If you are travelling it is a good idea to:
- Wear some form of identification showing you have diabetes
- Carry some form of sugar to treat hypoglycaemia (low blood glucose) if it occurs, for example, sugar sachets or jelly beans
- Carry emergency food rations in case of a delay, for example, dried fruit, biscuits or muesli bars
- Keep glimepiride tablets readily available.
If you become sick with a cold, fever or flu, it is very important to continue taking glimepiride, even if you feel unable to eat your normal meal.
If you have trouble eating solid food, use sugar-sweetened drinks as a carbohydrate substitute or eat small amounts of bland food.
Your diabetes educator or dietician can give you a list of foods to use for sick days.
Possible side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using Glimepiride, or if you have any questions or concerns.
All medicines may have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- signs of hypoglycaemia, which may include weakness, trembling or shaking, sweating, light-headedness, headache, dizziness, lack of concentration, tearfulness or crying, irritability, hunger and numbness around the lips and fingers
- nausea
- vomiting
- abdominal pain of discomfort
- diarrhoea or feeling of fullness in the stomach
- blurred or double vision
Tell your doctor as soon as possible if you notice any of the following.
These may be serious side effects and you may need medical attention:
- hypoglycaemia or hyperglycaemia, symptoms of sunburn such as redness, itching, swelling or blistering which may occur more quickly than normal after being in the sun
- bleeding or bruising more easily than normal, or reddish or purplish blotches under the skin
- signs of frequent or worrying infections, such as fever, severe chills, sore throat or mouth ulcers
- signs of anaemia, such as tiredness, being short of breath and looking pale.
If you experience any of the following, stop taking your medicine and contact your doctor immediately or go to the Accident and Emergency department at your nearest hospital.
These are very serious side effects and you may need urgent medical attention or hospitalisation:
- rash, sores, redness or itching of the skin, itchy hives-like rash of spots (this could mean that you are allergic to glimepiride)
- yellowing of the skin or eyes, also called jaundice
Tell your doctor or pharmacist if you notice anything that is making you fell unwell.
Other side effects not listed above may occur in some people.
Do not be alarmed by this list of possible side effects.
You may not experience any of them.
Storage and disposal
Storage
Keep your tablets in their blister pack until it is time to take them.
If you take the tablets out of the box or the blister pack they may not keep well.
Keep your medicine in a cool dry place where the temperature will stay below 25°C.
Do not store your medicine, or any other medicine, in the bathroom or near a sink. Do not leave it on a window sill or in the car.
Heat and dampness can destroy some medicines.
Keep this medicine where children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine or it has passed its expiry date, your pharmacist can dispose of the remaining medicine safely.
Product description
What APO-Glimepiride looks like
APO-Glimepiride 1 mg Tablets:
Pink, round tablet, G1 on one side and plain on the other.
APO-Glimepiride 2 mg Tablets:
Green, capsule-shaped tablet, G2, scoreline, G2 on one side and scoreline, on the other.
APO-Glimepiride 3 mg Tablets:
Pale yellow, capsule-shaped tablet, G3, scoreline, G3 on one side and scoreline, on the other.
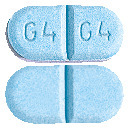
APO-Glimepiride 4 mg Tablets:
Blue, capsule-shaped tablet, G4, scoreline, G4 on one side and scoreline on the other.
Blister packs of 30 tablets.
Ingredients
Each tablet contains 1, 2, 3 or 4 mg of glimepiride as the active ingredient.
It also contains the following inactive ingredients:
- lactose
- microcrystalline cellulose
- povidone
- sodium starch glycollate
- magnesium stearate
- iron oxide red CI77491 (1 mg tablets)
- iron oxide yellow CI77492 (2 mg, 3 mg tablets)
- indigo carmine CI73015 (2 mg, 4 mg tablets)
Australian Registration Numbers
APO-Glimepiride 1 mg tablets
Blister pack
AUST R 151570
APO-Glimepiride 2 mg tablets
Blister pack
AUST R 151571
APO-Glimepiride 3 mg tablets
Blister pack
AUST R 151572
APO-Glimepiride 4 mg tablets
Blister pack
AUST R 151573
APO-Glimepiride does not contain gluten, sucrose, tartrazine or any other azo dyes.
Sponsor
Aspen Pharma Pty Ltd
34-36 Chandos Street,
St. Leonards NSW 2065
Australia
Distributor
Apotex Pty Ltd
16 Giffnock Avenue
Macquarie Park NSW 2113
Australia
APO and APOTEX are registered trade marks of Apotex Inc.
This leaflet was last updated in:
June 2014