What is in this leaflet
Read this leaflet carefully before taking your medicine.
This leaflet answers some common questions about nebivolol hydrochloride. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
The information in this leaflet was last updated on the date listed on the last page. More recent information on this medicine may be available.
Ask your doctor or pharmacist:
- if there is anything you do not understand in this leaflet,
- if you are worried about taking your medicine, or
- to obtain the most up-to-date information.
You can also download the most up to date leaflet from www.apotex.com.au.
All medicines have risks and benefits. Your doctor has weighed the risks of you using this medicine against the benefits they expect it will have for you.
Pharmaceutical companies cannot give you medical advice or an individual diagnosis.
Keep this leaflet with your medicine. You may want to read it again.
What this medicine is used for
The name of your medicine is APO-NEBIVOLOL Tablet. It contains the active ingredient Nebivolol hydrochloride.
It is used to treat:
- high blood pressure (essential hypertension) or,
- chronic heart failure. It is usually used in combination with other medicines.
How it works
This medicine belongs to a group of medicines called beta-blockers.
High Blood Pressure (Hypertension):
Nebivolol can be used to treat high blood pressure. It works by blocking the transmission of messages to the beta receptors in the heart which slows down the activity of the heart. It also relaxes the blood vessels and reduces the resistance that the heart has to pump against. The blood pressure lowering effects occur after 1 to 2 weeks, although it may take up to 4 weeks for the full effect to occur.
Chronic Heart Failure:
Nebivolol can also be used to treat chronic heart failure. It slows down the heart rate and makes it beat more regularly.
It also reduces the pressure the heart has to pump against and therefore reduces the amount of work that the heart has to do.
This improves the symptoms of chronic heart failure and reduces the likelihood of you being hospitalized and/or dying from your heart condition.
Use in children
There is not enough information to recommend the use of this medicine for children under the age of 18 years.
Before you take this medicine
When you must not take it
Do not take this medicine if:
- You have or have had any of the following:
- have liver problems
- have acute heart failure or cardiogenic shock, a serious heart condition causing low blood pressure
- have a history of slow heart rate or uneven heart beating
- have certain other heart conditions
- have asthma, bronchitis or other lung disease with tightening of the airways
- have untreated phaeochromocytoma, a rare tumour of the adrenal gland
- have metabolic acidosis, where there is too much acid in the blood
- have hypotension, low blood pressure
- have a severe blood vessel disorder causing poor circulation in the arms and legs. - You are pregnant.
Nebivolol may affect your developing baby if you take it during pregnancy. - You are breastfeeding.
Nebivolol may pass into human breast milk. - You are hypersensitive to, or have had an allergic reaction to, Nebivolol or any of the ingredients listed at the end of this leaflet.
Symptoms of an allergic reaction may include: cough, shortness of breath, wheezing, difficulty breathing or tightness in chest; swelling of the face, lips, tongue, throat or other parts of the body; rash, itching or hives on the skin; fainting; or hay fever-like symptoms.
If you think you are having an allergic reaction, do not take any more of the medicine and contact your doctor immediately or go to the Accident and Emergency department at the nearest hospital. - The expiry date (EXP) printed on the pack has passed.
- The packaging is torn, shows signs of tampering or it does not look quite right.
Before you start to take it
Before you start taking this medicine, tell your doctor if:
- You have allergies to:
- any other medicines
- any other substances, such as foods, preservatives or dyes.
- You have or have had any medical conditions, especially the following:
- low blood pressure or poor circulation
- a slow heartbeat, lower than 60 beats per minute
- asthma or breathing difficulties
- angina, chest pain or any other heart problems, or your heart failure has just occurred or worsened recently
- diabetes
- a history of psoriasis, a skin disease with thickened patches of red skin, often with silvery scales
- a severe blood vessel disorder causing poor circulation in the arms and legs
- thyroid disorders
- liver problems
- kidney problems
- phaeochromocytoma, a rare tumour of the adrenal gland
- a lactose intolerance
- You are currently pregnant or you plan to become pregnant. Do not take this medicine whilst pregnant (until you and your doctor have discussed the risks and benefits involved).
- You are currently breastfeeding or you plan to breast-feed. Do not take this medicine whilst breastfeeding (until you and your doctor have discussed the risks and benefits involved).
- You have recently been vaccinated or plan to get a vaccination.
- You are planning to have surgery or an anaesthetic.
- You are currently receiving or are planning to receive dental treatment.
- You are taking or are planning to take any other medicines. This includes vitamins and supplements that are available from your pharmacy, supermarket or health food shop.
Taking other medicines
Some medicines may interact with Nebivolol. These include:
- those used to treat irregular or abnormal heartbeat (e.g. quinidine, flecainide, disopyramide, lidocaine, mexiletine, amiodarone)
- those used to treat high blood pressure (e.g. clonidine)
- calcium channel blockers for high blood pressure or heart problems (e.g. verapamil, diltiazem)
- insulin and tablets used to treat diabetes
- those used to treat depression and anxiety (e.g. phenelzine, tranylcypromine, paroxetine, sertraline, sedatives, phenothiazine)
- those used to treat excessive stomach acid (e.g. cimetidine).
- you should take Nebivolol during a meal, and the antacid between meals.
- digoxin, a medicine used to treat heart failure
- drugs used to treat asthma, blocked nose or certain eye disorders such as glaucoma or dilation of the pupil
- dextromethorphan, an ingredient in cough medicines
- anaesthetics
- other beta-blocker medicines, including beta-blocker eye drops
If you are taking any of these you may need a different dose or you may need to take different medicines.
Other medicines not listed above may also interact with nebivolol.
How to take this medicine
Follow carefully all directions given to you by your doctor. Their instructions may be different to the information in this leaflet.
How much to take
Your doctor will tell you how much of this medicine you should take. This will depend on your condition and whether you are taking any other medicines.
Do not stop taking your medicine or change your dosage without first checking with your doctor.
How to take it
Swallow the tablets whole with a full glass of water.
Do not crush or chew the tablets
When to take it
Take this medicine at the same time each day. Taking it at the same time each day will have the best effect and will also help you remember when to take it.
It does not matter if you take it before, with or after food.
How long to take it for
Continue taking your medicine for as long as your doctor tells you.
Make sure you have enough to last over weekends and holidays.
If you forget to take it
If it is almost time to take your next dose, skip the missed dose and take your next dose at the usual time. Otherwise, take it as soon as you remember and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for missed doses. This may increase the chance of you experiencing side effects.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints to help you remember.
If you take too much (overdose)
If you think that you or anyone else may have taken too much of this medicine, immediately telephone your doctor or the Poisons Information Centre (Tel: 13 11 26 in Australia) for advice. Alternatively, go to the Accident and Emergency department at your nearest hospital.
Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are taking this medicine
Things you must do
Tell your doctor that you are taking this medicine if:
- you are about to be started on any new medicine
- you plan to have any vaccinations or immunisations
- you are pregnant or are planning to become pregnant
- you are breastfeeding or are planning to breast-feed
- you are about to have any blood tests
- you are going to have surgery or an anaesthetic or are going into hospital.
Your doctor may occasionally do tests to make sure the medicine is working and to prevent side effects. Go to your doctor regularly for a check-up.
Tell any other doctors, dentists and pharmacists who are treating you that you take this medicine.
Things you must not do
Do not:
- Give this medicine to anyone else, even if their symptoms seem similar to yours.
- Take your medicine to treat any other condition unless your doctor tells you to.
- Stop taking your medicine, or change the dosage, without first checking with your doctor.
Things to be careful of
Be careful when driving or operating machinery until you know how this medicine affects you.
Possible side effects
Tell your doctor as soon as possible if you do not feel well while you are taking nebivolol or if you have any questions or concerns.
Do not be alarmed by the following lists of side effects. You may not experience any of them. All medicines can have side effects. Sometimes they are serious but most of the time they are not.
Tell your doctor if you notice any of the following:
- headache, tiredness nausea or vomiting stomach upset, wind, diarrhoea or constipation
- disturbed sleep or nightmares
- coldness or numbness in the hands or fee
Tell your doctor as soon as possible if you notice any of the following.
These may be serious side effects and you may need medical attention:
- slow heart beat
- dizziness or fainting
- tingling or "pins and needles"
- abnormal thinking, depression or hallucinations
- sexual problems
- skin rashes and itching
- dry eyes or problems with eye sight
- an increase in a cramp-like pain in one or both legs that develops on walking.
If you experience any of the following, stop taking your medicine and contact your doctor immediately or go to the Accident and Emergency department at your nearest hospital.
These are very serious side effects and you may need urgent medical attention or hospitalisation:
- swelling of the face, lips, tongue or throat that may cause difficulty swallowing or breathing
- chest tightness, shortness of breath or difficulty breathing
- swelling of the feet and ankles
- chest pain or changes in heart rate
Other side effects not listed above may occur in some patients.
Allergic reactions
If you think you are having an allergic reaction to nebivolol, do not take any more of this medicine and tell your doctor immediately or go to the Accident and Emergency department at your nearest hospital.
Symptoms of an allergic reaction may include some or all of the following:
- cough, shortness of breath, wheezing, difficulty breathing or tightness in chest
- swelling of the face, lips, tongue, throat or other parts of the body
- rash, itching or hives on the skin
- fainting
- hay fever-like symptoms.
Storage and disposal
Storage
Keep your medicine in its original packaging until it is time to take it.
If you take your medicine out of its original packaging it may not keep well.
Keep your medicine in a cool dry place where the temperature will stay below 30°C.
Do not store your medicine, or any other medicine, in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep this medicine where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor (or pharmacist) tells you to stop taking this medicine or it has passed its expiry date, your pharmacist can dispose of the remaining medicine safely.
Product description
What APO-NEBIVOLOL looks like
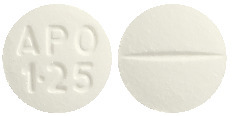
1.25 mg tablet: White to off-white, round, biconvex tablets debossed "APO" over "1.25" on one side and single score on the other side.
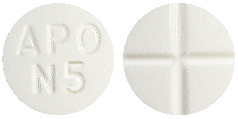
5 mg tablet: White to off-white, round, biconvex tablets debossed "APO" over "N5" on one side and cross scored on the other side
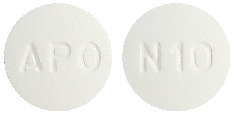
10 mg tablet: White to off-white, round, biconvex tablets debossed "APO" on one side and "N10" on the other side
* Not all strengths, pack types and/or pack sizes may be available.
Ingredients
Each tablet contains 1.25mg, 5mg or 10 mg of nebivolol hydrochloride as the active ingredient.
It also contains the following inactive ingredients:
- lactose monohydrate,
- crospovidone,
- copovidone,
- pregelatinised starch
- magnesium stearate
This medicine contains sugars as lactose.
This medicine is gluten-free, tartrazine-free and free of other azo dyes.
Australian Registration Numbers
APO-Nebivolol 1.25 mg tablet (Blister pack): AUST R 281799.
APO-Nebivolol 5 mg tablet (Blister pack): AUST R 281795.
APO-Nebivolol 10 mg tablet (Blister pack): AUST R 281798.
Sponsor
Apotex Pty Ltd
16 Giffnock Avenue
Macquarie Park NSW 2113
APO and APOTEX are registered trademarks of Apotex Inc.
This leaflet was last updated in: June 2023.
Published by MIMS July 2023

 Other adverse events reported during US clinical trials with an incidence of at least 1% include: hypercholesterolaemia, hyperuricaemia, increase in BUN, uric acid, triglycerides and a decrease in HDL cholesterol and platelet count. These adverse events were in most cases observed at a similar frequency in placebo-treated patients in controlled-studies.
Other adverse events reported during US clinical trials with an incidence of at least 1% include: hypercholesterolaemia, hyperuricaemia, increase in BUN, uric acid, triglycerides and a decrease in HDL cholesterol and platelet count. These adverse events were in most cases observed at a similar frequency in placebo-treated patients in controlled-studies. In the SENIORS study, 165 (15.5%) of nebivolol patients and 136 (12.8%) of placebo patients experienced at least one adverse event that led to permanent treatment discontinuation. The higher discontinuation rate due to adverse events in the nebivolol group, in particular due to bradycardia, is not unexpected given the pharmacology of the drug.
In the SENIORS study, 165 (15.5%) of nebivolol patients and 136 (12.8%) of placebo patients experienced at least one adverse event that led to permanent treatment discontinuation. The higher discontinuation rate due to adverse events in the nebivolol group, in particular due to bradycardia, is not unexpected given the pharmacology of the drug.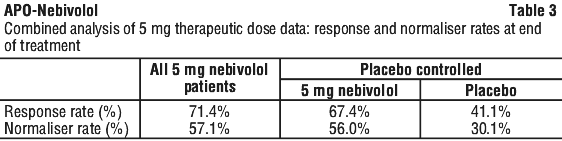 The blood pressure lowering effect becomes evident after 2 weeks of treatment. Occasionally, the optimal effect is reached only after 4 weeks.
The blood pressure lowering effect becomes evident after 2 weeks of treatment. Occasionally, the optimal effect is reached only after 4 weeks. The study period encompassed an up-titration period from between 4 and 16 weeks, with a dose escalation every 1-2 weeks. The maintenance period continued for up to 40 months, resulting in a median observation period of 20 months (in quartile range 14 to 29 months) and a median treatment exposure of 17 months. The trial concluded with a down-titration phase involving halving the dose every week.
The study period encompassed an up-titration period from between 4 and 16 weeks, with a dose escalation every 1-2 weeks. The maintenance period continued for up to 40 months, resulting in a median observation period of 20 months (in quartile range 14 to 29 months) and a median treatment exposure of 17 months. The trial concluded with a down-titration phase involving halving the dose every week. Nebivolol, on top of standard therapy, induced a statistically and clinically significant benefit over placebo by prolonging the time to occurrence of either death from any cause or cardiovascular hospitalisation (time to first event, primary end-point for efficacy) with a relative risk reduction of 14% and absolute risk reduction of 4.2%. This corresponds to a number needed to treat (NNT) of 28 patients to prevent one death or one cardiac hospitalisation. The benefit over placebo is evident after about 6 month's treatment and was maintained for the duration of follow-up.
Nebivolol, on top of standard therapy, induced a statistically and clinically significant benefit over placebo by prolonging the time to occurrence of either death from any cause or cardiovascular hospitalisation (time to first event, primary end-point for efficacy) with a relative risk reduction of 14% and absolute risk reduction of 4.2%. This corresponds to a number needed to treat (NNT) of 28 patients to prevent one death or one cardiac hospitalisation. The benefit over placebo is evident after about 6 month's treatment and was maintained for the duration of follow-up.
