What is in this leaflet
This leaflet answers some common questions about risperidone. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using this medicine against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may want to read it again.
What this medicine is used for
The name of your medicine is APO-Risperidone. It contains the active ingredient risperidone.
Risperidone belongs to a group of medicines called antipsychotic agents, which improve the symptoms of certain types of mental illness.
Risperidone is used for:
- treatment of sudden (acute) and long-term (chronic) schizophrenia and other types of related psychoses. These are disorders related to thought, feeling and/or action
- short term treatment of acute mania associated with Bipolar 1 Disorder – symptoms of this condition may include elevated, expansive or irritable mood, inflated self-esteem, decreased need for sleep, pressured speech, racing thoughts, distractibility or poor judgment including disruptive or aggressive behaviours
- short term treatment of behavioural problems in patients with a decline in mental ability (dementia) – these problems include aggression through words or actions, morbid suspiciousness, agitation or wandering
- conduct and other disruptive disorders such as aggression, impulsiveness and self-injury in adults, adolescents and children aged over 5 years old who are intellectually disabled
- certain behaviours seen in children and adolescents with autism
How it works
Risperidone helps to correct a chemical imbalance in the brain associated with the above conditions.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed this medicine for another reason.
This medicine is available only with a doctor's prescription.
There is no evidence this medicine is addictive.
Before you take this medicine
When you must not use it
Do not use this medicine if you have an allergy to:
- risperidone
- any of the ingredients listed at the end of this leaflet
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue, throat or other parts of the body
- skin rash, itching or hives on the skin
Do not take this medicine if you are pregnant. Do not take risperidone whilst pregnant until you and your doctor have discussed the risks and benefits involved.
Newborn babies of mother taking risperidone in their last trimester may be at risk of having difficulty feeding or breathing, shaking, muscle stiffness and/or weakness, sleepiness or agitation.
Do not breastfeed if you are taking this medicine. Risperidone passes into breast milk and there is a possibility that your baby may be affected.
Do not use this medicine after the expiry date printed on the pack or the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- Irregular heart rhythm, abnormalities in electrical activity of the heart, high or low blood pressure, or you have had a heart attack or stroke in the past.
- unusual excessive sweating or diarrhoea, dehydration or problems with your body temperature regulation
- prone to dizziness when standing up from a lying or sitting position
- kidney or liver problems
- Parkinson's disease (a progressive movement and thinking disorder that tends to affect older people)
- dementia or Lewy Body dementia older people suffering dementia may be at increased risk of stroke or death with Risperidone
- sugar diabetes
- unusual thirst, tiredness, blurred vision, upset stomach or need to urinate – common signs of high blood sugars
- epilepsy or seizures or fits
- restlessness or difficulty sitting still
- continuous and/or painful erections (called 'priapism')
- intraoperative iris syndrome (a complication that may occur during cataract extraction)
- Involuntary movements or unusual restlessness or difficulty sitting still
- suicidal thoughts or past suicide attempts
- low volume of blood in your body (hypovolaemia) - seen by cold hands and feet, light headedness, infrequent urination, increased heart rate, and weakness
- low blood potassium levels (hypokalaemia)
- breast cancer
- cancer of the pituitary gland
- diabetes, high or low blood sugar (you may need to monitor your blood glucose levels closely)
- tardive dyskinesia (a reaction to some medicines with uncontrollable twitching or jerking movements of the tongue, face, mouth, jaw, arms and legs)
- neuroleptic malignant syndrome (a serious reaction to some medicines with a sudden increase in body temperature, very fast heartbeat, extremely high or low blood pressure and severe muscle stiffness or fits)
- blood clots, Tell your doctor if you or someone else in your family has a history of blood clots. Blood clots in the lungs and legs can occur with Risperidone. Blood clots in the lungs can result in death.
- low white blood cell count
Tell your doctor if you are pregnant or plan to become pregnant or are breastfeeding. Do not take risperidone until you and your doctor have discussed the risks and benefits involved.
Tell your doctor if you are planning to have surgery (especially of the eye), dental treatment or an anaesthetic. If you are having eye surgery, your doctor will need to assess whether you are at risk of a surgical complication (called 'Intraoperative Floppy Iris Syndrome).
Tell your doctor if you will be in a hot environment or do a lot of vigorous exercise. Risperidone may make you sweat less, causing your body to overheat.
If you have not told your doctor about any of the above, tell them before you take this medicine.
Taking other medicines
Tell your doctor and pharmacist if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interact with risperidone. These include:
- sleeping tablets, tranquillisers, strong pain-killers or antihistamines which may make you drowsy, certain antidepressants and alcohol
- medicines to treat Parkinson's disease or a tremor
- medicines that increase the activity of the central nervous system (e.g. psychostimulants such as methylphenidate)
- medicines to treat epilepsy such as carbamazepine (also used to treat trigeminal neuralgia)
- medicines to treat depression, panic disorder, anxiety, obsessive-compulsive disorder or premenstrual dysphoric disorder, such as tricyclic antidepressants (TCAs) and selective serotonin reuptake inhibitors (SSRIs) e.g. fluoxetine and paroxetine
- other medicines to treat mental illness or psychotic conditions
- diuretics, used to treat high blood pressure or swelling caused by the build-up of too much fluid e.g. frusemide – this combination may increase the risk of side effects or death in elderly people
- rifampicin, an antibiotic
- Verapamil, a medicine used to treat high blood pressure and/or abnormal heart rhythm.
- medicines used to treat heart problems or blood pressure
- medicines to relieve severe nausea and vomiting
- protease inhibitors, used to treat HIV infection (e.g. tipranavir, ritonavir)
- cimetidine and ranitidine, used for treating stomach ulcers or excess acid
- medicines which have an effect on the heart called QT prolongation
- medicines which affect the liver so that more of an enzyme called CYP3A4 is produced
- itraconazole and ketoconazole, medicines used to treat fungal infections.
These medicines may be affected by risperidone or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
Other medicines not listed above may also interact with risperidone.
How to take this medicine
Follow all directions given to you by your doctor or pharmacist carefully. They may differ to the information contained in this leaflet.
If you do not understand the instructions on the label, ask your doctor or pharmacist for help.
Taking it for the first time
At the start of treatment, you may have a fall in blood pressure making you feel dizzy on standing up, or your heart may beat faster. Standing up slowly, especially when you get up from bed or chairs, will help your body get used to the change in position and blood pressure.
These symptoms should go away after a few days. Tell your doctor if they continue or worry you.
How much to take
Your doctor or pharmacist will tell you how much of this medicine you should take. This will depend on your condition and whether you are taking any other medicines.
Risperidone may be taken as a single dose, once a day, or it may be taken in divided doses twice a day (in the morning and in the evening).
It is very important that you take the correct amount of risperidone, but this will vary from person to person. Your doctor will adjust the number and strength of the tablets until the desired effect is obtained.
For schizophrenia and related psychoses in adults, adolescents and children aged over 15 years old:
The usual starting dose is one 1 mg tablet taken twice a day. This will be gradually increased by your doctor to suit your needs.
From then on, the dose can be taken once a day or twice a day according to your doctor's instructions.
For long-term treatment, 4 to 6 mg per day is usually sufficient, but your doctor will determine the dose most suitable for you.
Important Note: Never take more tablets than your doctor tells you to take.
The effects of high doses are not yet known. Please double check with your doctor if your doctor prescribes more than 5 mg twice a day.
Risperidone cannot be recommended for use in children with schizophrenia under 15 years as there is little experience with the product in this group.
For schizophrenia and related psychoses in elderly patients:
The usual starting dose is one 0.5 mg tablet taken twice a day.
The dose may be increased by 0.5 mg twice daily to 1 to 2 mg twice a day.
Patients who have impaired kidney or liver function:
The usual starting dose is one 0.5 mg tablet taken twice a day. The dose may be increased by 0.5 mg twice daily to 1 to 2 mg twice a day.
For acute mania in patients with Bipolar 1 Disorder:
The usual starting dose is one 2 mg tablet once a day. This dose can be adjusted by dose increases of 1 mg when needed every 24 hours. Most people feel better with doses between 2 mg and 6 mg a day. Your doctor may also decide you should take another drug called a mood stabiliser as well as risperidone.
For behavioural problems in people with dementia:
The usual starting dose is 0.25 mg (half a 0.5 mg tablet) taken twice daily. This may be gradually increased by your doctor to suit your needs.
From then on, the dose can be taken once a day or twice a day according to your doctor's instructions. For long-term treatment, 1 mg daily is the usual dose, but your doctor will determine the dose most suitable for you.
For conduct and other disruptive disorders in adults, adolescents and children aged over 5 years old who are intellectually disabled:
For people who weigh 50 kg or more, the usual starting dose is one 0.5 mg tablet taken once a day. The dose may be increased by 0.5 mg once every two days, to the usual dose of 0.5 to 1.5 mg once a day.
For people who weigh less than 50 kg, the usual starting dose is 0.25 mg (half a 0.5 mg tablet) taken once a day. The dose may be increased by 0.25 mg once every two days, to the usual dose of 0.25 to 0.75 mg once a day.
Your doctor will advise you on how much risperidone you need.
Risperidone cannot be recommended for use in children under 5 years or age with disruptive behaviour disorders as there is little experience with the product in this group.
For behavioural disorders associated with autism in children and adolescents:
For people weighing less than 20 kg the usual starting dose is 0.25 mg (half a 0.5 mg tablet) daily. On Day 4, this dose can be increased to 0.5 mg.
For people weighing 20 kg or more the usual starting dose is 0.5 mg (0.5 mg tablet or half a 1 mg tablet) daily. On Day 4, this dose can be increased to 1 mg.
After 14 days, the doctor should check that the tablets are working. The doctor will say whether a higher dose is needed if the tablets do not seem to be have the desired effect. Your doctor will advise you on how much risperidone you need.
In clinical trials, people with autism weighing less than 20 kg did not need more than 1.5 mg/day. People weighing over 20 kg did not need more than 2.5 mg of risperidone a day, and people weighing over 45 kg did not need more than 3.5 mg a day.
If you feel sleepy, then the doctor may split up your daily dose so that you take half of it in the morning and half in the evening.
How to take it
Swallow the tablets whole with a full glass of water.
When to take it
Take your medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
It does not matter if you take this medicine before or after food.
How long to take it
Continue taking your medicine for as long as your doctor tells you.
This medicine helps to control your condition but does not cure it. It is important to keep taking your medicine even if you feel well.
If you forget to take it
If it is almost time to take your next dose, skip the dose you missed and take the next dose when you are meant to.
Otherwise take it as soon as you remember and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for missed doses. This may increase the chance of you experiencing side effects.
If you forget to take risperidone for 5 days or more, tell your doctor before starting your medicine again.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints to help you remember.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at your nearest hospital, if you think that you or anyone else may have taken too much of this medicine. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
Symptoms of an overdose may include drowsiness, sleepiness, excessive trembling, excessive muscle stiffness, increased heart rate, and very low blood pressure causing fainting or unconsciousness.
While you are taking this medicine
Things you must do
If you are about to be started on any new medicine, tell your doctor or pharmacist that you are taking risperidone.
Tell any other doctors, dentists and pharmacists who are treating you that you are taking this medicine.
If you become pregnant or start breastfeeding while you are taking risperidone, tell your doctor immediately.
If you are going to have surgery, tell the surgeon or anaesthetist that you are using this medicine.
Keep all of your doctor's appointments so that your progress can be checked. Your doctor may occasionally do tests to make sure risperidone is working and to prevent side effects.
If a child is taking risperidone, tell the doctor if they do not seem to be developing normally.
Tell your doctor immediately if you notice any unusual movements of the tongue, or other uncontrolled movements of the tongue, mouth, cheeks or jaw which may progress to the arms and legs. These are symptoms of a condition called tardive dyskinesia, which may develop in people taking antipsychotic medicines, including risperidone. This condition is more likely to occur during long term treatment with risperidone, especially in elderly women. In very rare cases, this may be permanent. However, if detected early, these symptoms are usually reversible.
Pre-menopausal women should tell their doctor if they do not have a period for more than six months while taking risperidone.
Be careful during strenuous exercise or exposure to extreme heat. Try to drink plenty of water.
Things you must not do
Do not take your medicine to treat any other complaints unless your doctor tells you to.
Do not give this medicine to anyone else, even if they have the same condition as you.
Do not stop taking your medicine, or change the dosage, without first checking with your doctor. Do not use more than the recommended dose.
Do not drink alcohol whilst you are taking this medicine. Risperidone can increase the effects of alcohol.
Things to be careful of
Be careful when driving or operating machinery until you know how this medicine affects you.
Risperidone may cause dizziness, drowsiness or light-headedness in some people, especially after the first dose.
Make sure you know how you react to risperidone before you drive a car, operate machinery, or do anything else that could be dangerous if you feel drowsy or dizzy.
Avoid excessive eating and try to eat a moderate diet. Risperidone can cause weight gain.
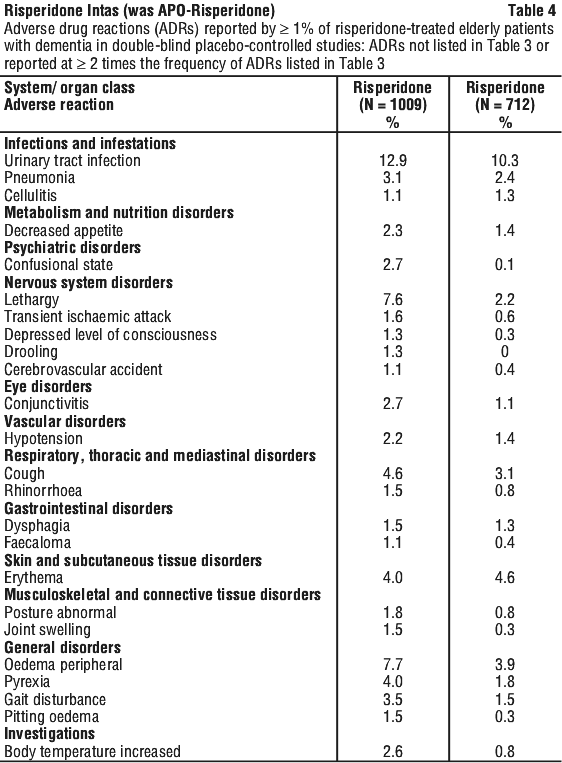
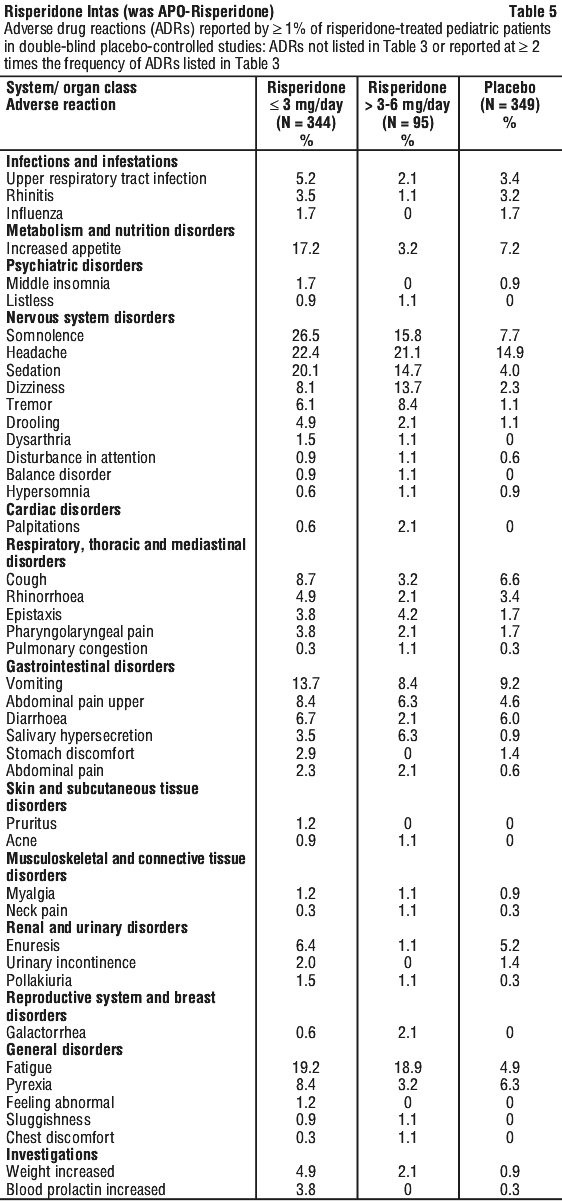
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking risperidone. Your doctor will decide whether any change in your treatment is needed.
This medicine helps most people, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not.
Do not be alarmed by this list of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following side effects and they worry you:
- difficulty thinking, working or carrying out your usual daily activities because of:
- headache
- trembling, muscle weakness, unsteadiness on your feet, lack of coordination or slow, shuffling walk (symptoms of Parkinsonism).
- Lack of energy, drowsiness or excessive sleeping during the day, sleeplessness or difficulty in concentrating
- Difficulty speaking
- Blurred vision
- Fainting
- Dizziness
- Any problems with confusion or unsteadiness
- Pains in parts of your body, e.g. in the neck, back, ear, hands or feet
- falling
- muscle joint, nerve or movement changes such as:
- shaking or trembling
- fatigue or weakness
- muscle stiffness
- restlessness in the legs or difficulty sitting still
- uncontrolled muscle spasms, twitching, jerky or writhing movements
- muscle aches or pain
- joint swelling or pain
- walking abnormally or with difficulty
- abnormal posture, such as rigid body movements and persistent abnormal positions of the body
- behavioural changes such as:
- irritability or agitation
- unusual anxiety or nervousness
- other changes such as:
- cold or flu-like symptoms e.g. cough, blocked or runny nose, sneezing, sore throat
- feeling of tension or fullness in the nose, cheeks and behind your eyes, sometimes with a throbbing ache, fever, stuffy nose and loss of the sense of smell (signs of sinusitis)
- tiredness, headaches, being short of breath when exercising, dizziness and looking pale (signs of decreased red blood cells)
- fever, chills, shortness of breath, cough, phlegm and occasionally blood (signs of pneumonia)
- nosebleeds
- discharge with itching of the eyes and crusty eyelids
- unexplained weight gain
- unexplained increase or decrease in appetite
- indigestion, stomach discomfort or pain, diarrhoea or constipation
- nausea or vomiting
- dry mouth or excessive thirst
- drooling
- difficulty swallowing
- acne
- dry skin
- rash, red skin or itchy skin
- thickening of the skin resulting in warts, corns, calluses
- skin infection
- swelling of any part of your body, e.g. hands, ankles or feet
- inability to or feeling burning pain when passing urine
- some loss of bladder control
- bedwetting
- frequent daytime urination in children
- sexual function disturbances – problems with ejaculation
- breast abnormalities – breast discomfort or swelling or unusual secretion of breast milk
- missed or irregular menstrual periods
- dizziness on standing up, especially when getting up from a sitting or lying down position
- shortness of breath
- chest pain or discomfort
- an increase of CPK (creatine phosphokinase) in your blood, an enzyme which is sometimes released with muscle breakdown.
These can only be detected by blood tests that your doctor may ask to be done.
Tell your doctor immediately, or go to accident and emergency at your nearest hospital if you notice any of the following:
Signs of heart or blood pressure problems including:
- Fainting, blurry vision, light-headedness or dizziness particularly on standing that persists despite sitting or lying down again
- Very fast heart rate, slowed heart rate, heart rhythm irregularities.
- Signs of lung problems including:
- Sudden shortness of breath, trouble breathing, wheezing or gasping when you breathe, light-headedness or dizziness
- Signs of high blood sugar or diabetes such as:
- Unusual thirst, tiredness, upset stomach or need to urinate more often than usual
- Body temperature changes such as:
- Fever
- Unexplained high body temperature, excessive sweating or rapid breathing
- Severe muscle stiffness or fits
- Involuntary movements of the tongue, face, mouth, jaw, arms, legs or trunk
- Severe or life-threatening rash with blisters and peeling skin that may start in and around the mouth, nose, eyes and genitals and spread to other areas of the body (Stevens-Johnson syndrome or toxic epidermal necrolysis)
- Rash, itching or hives on the skin; shortness of breath, wheezing or difficulty breathing; swelling of the face, lips, tongue or other parts of the body.
- Sudden weakness or numbness of the face, arms or legs, especially on one side, or instances of slurred speech (these are called mini-strokes)
Tell your doctor if you notice anything else that is making you feel unwell.
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation.
Other side effects not listed above may occur in some patients.
Do not hesitate to report any other side effects to your doctor or pharmacist. Do not be alarmed by this list of possible side effects. You may not experience any of them.
Storage and disposal
Storage
Keep your tablets in the pack until it is time to take them. If you take the tablets out of the pack, they may not keep well.
Keep your tablets in a cool dry place where the temperature stays below 25°C.
Do not store your medicine, or any other medicine, in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep this medicine where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
APO-Risperidone 0.5 mg tablets:
Brick red coloured, round, biconvex, film coated tablets plain on both sides
Blister pack of 20 or 60 tablets. AUST R 304250.
APO-Risperidone 1 mg tablets:
White to off white capsule shaped, biconvex, film coated tablets plain on both sides.
Blister pack of 60 tablets. AUST R 304251.
APO-Risperidone 2 mg tablets:
Light orange coloured, capsule shaped, biconvex, film coated tablets plain on both sides.
Blister pack of 60 tablets. AUST R 304252.
APO-Risperidone 3 mg tablets:
Light yellow coloured, oval shaped, biconvex, film coated tablets with break line on one side and plain on other side.
Blister pack of 60 tablets. AUST R 304253.
APO-Risperidone 4 mg tablets:
Green coloured, capsule shaped, biconvex, film coated tablets plain on both sides.
Blister pack of 60 tablets. AUST R 304254.
*Not all strengths, pack types and/or pack sizes may be available.
Ingredients
Each tablet contains 0.5 mg, 1 mg, 2 mg, 3 mg or 4 mg of risperidone as the active ingredient.
It also contains the following inactive ingredients:
- lactose monohydrate
- maize starch
- microcrystalline cellulose
- hypromellose
- magnesium stearate
- colloidal anhydrous silica
- sodium lauryl sulfate
- purified talc
- propylene glycol
- titanium dioxide (0.5 mg, 2 mg, 3 mg and 4 mg tablets)
- iron oxide red (0.5 mg tablets)
- sunset yellow FCF aluminium lake (2 mg tablets)
- quinoline yellow aluminium lake (3 mg and 4 mg tablets)
- indigo carmine aluminium lake (4 mg tablets)
This medicine is gluten-free, contains sugars as lactose, tartrazine-free and free of other azo dyes.
Sponsor
Arrotex Pharmaceuticals Pty Ltd
15-17 Chapel Street
Cremorne VIC 3121
www.arrotex.com.au
This leaflet was prepared in November 2023.
Published by MIMS April 2026






 For prescribers preferring to dose on a mg/kg/day basis the following guidance is provided.
For prescribers preferring to dose on a mg/kg/day basis the following guidance is provided. Patients experiencing somnolence may benefit from a switch in dosing from once daily to either once daily at bedtime or twice daily.
Patients experiencing somnolence may benefit from a switch in dosing from once daily to either once daily at bedtime or twice daily.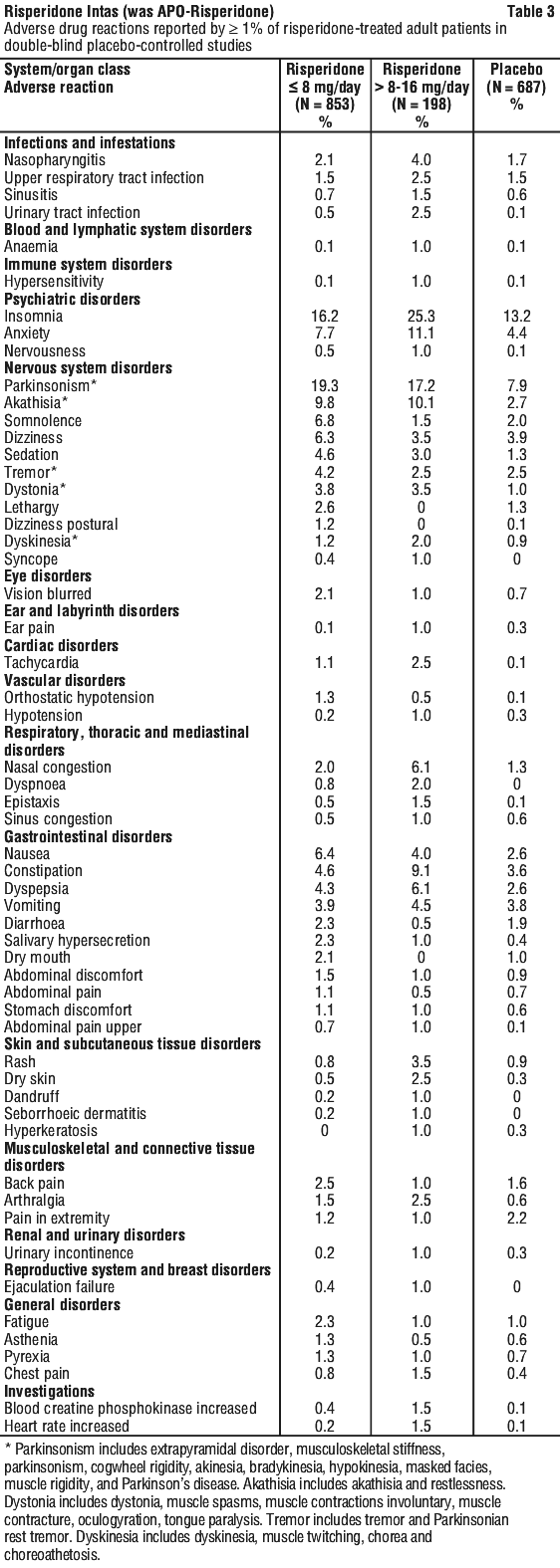
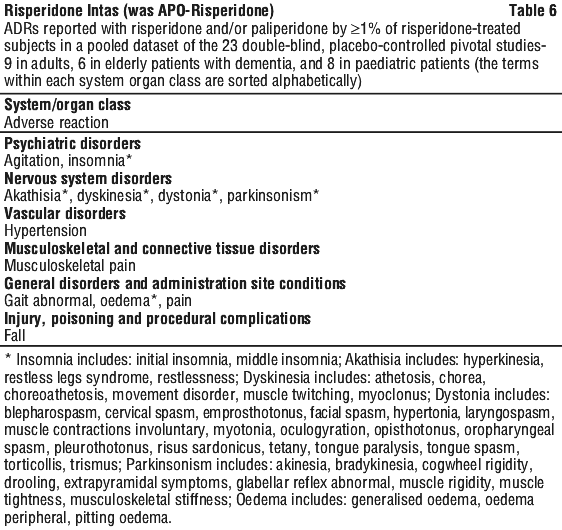 ADRs reported with risperidone and/or paliperidone by < 1% of risperidone treated subjects in a pooled dataset of 23 double blind, placebo controlled pivotal studies (9 in adults, 6 in elderly patients with dementia, and 8 in paediatric patients) are shown in Table 6B.
ADRs reported with risperidone and/or paliperidone by < 1% of risperidone treated subjects in a pooled dataset of 23 double blind, placebo controlled pivotal studies (9 in adults, 6 in elderly patients with dementia, and 8 in paediatric patients) are shown in Table 6B. ADRs reported with risperidone and/or paliperidone in other clinical trials but not reported by risperidone treated subjects in a pooled dataset of 23 double blind, placebo controlled pivotal studies are shown in Table 6C.
ADRs reported with risperidone and/or paliperidone in other clinical trials but not reported by risperidone treated subjects in a pooled dataset of 23 double blind, placebo controlled pivotal studies are shown in Table 6C.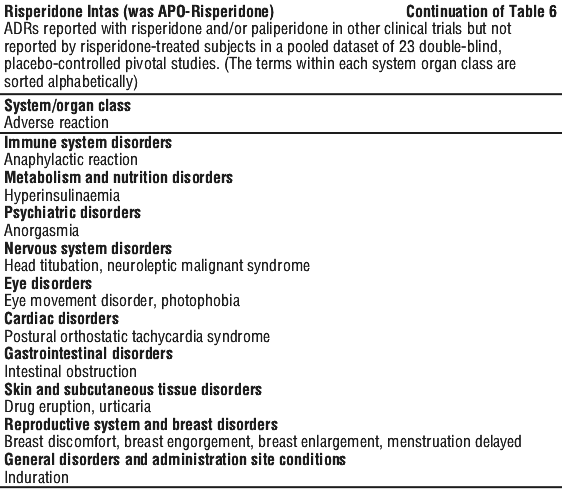
 There have also been reports of benign pituitary adenoma that were temporally related, but not necessarily causally related, to risperidone therapy.
There have also been reports of benign pituitary adenoma that were temporally related, but not necessarily causally related, to risperidone therapy.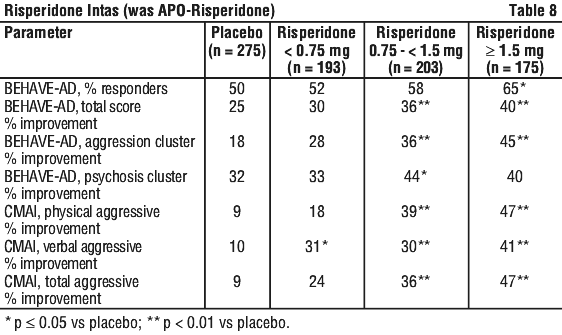 The rate of discontinuation from the pooled studies was similar for patients receiving placebo (30.2%), risperidone (33.5%) and haloperidol (29.6%). In the combined analysis, risperidone, at a daily dose above 0.75 mg, effectively reduces the severity (measured by means of the Behave-AD) and frequency (measured by the CMAI) of aggressiveness symptoms in this patient population.
The rate of discontinuation from the pooled studies was similar for patients receiving placebo (30.2%), risperidone (33.5%) and haloperidol (29.6%). In the combined analysis, risperidone, at a daily dose above 0.75 mg, effectively reduces the severity (measured by means of the Behave-AD) and frequency (measured by the CMAI) of aggressiveness symptoms in this patient population.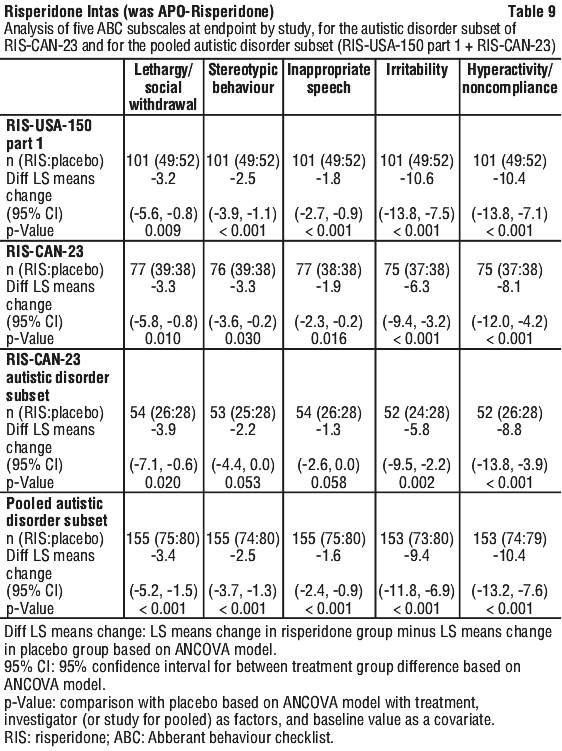 Following completion of study 1, 63 patients entered an open label extension for up to 4 additional months. 39 patients who were clinically stable and who showed a positive response to risperidone after 6 months were then randomised to receive risperidone or placebo during an 8 week, double blind withdrawal period. The relapse rate was 11/16 and 2/16 in placebo and risperidone treated patients, respectively (odds ratio 15.4, 95% confidence limits 2.50, 95.05).
Following completion of study 1, 63 patients entered an open label extension for up to 4 additional months. 39 patients who were clinically stable and who showed a positive response to risperidone after 6 months were then randomised to receive risperidone or placebo during an 8 week, double blind withdrawal period. The relapse rate was 11/16 and 2/16 in placebo and risperidone treated patients, respectively (odds ratio 15.4, 95% confidence limits 2.50, 95.05).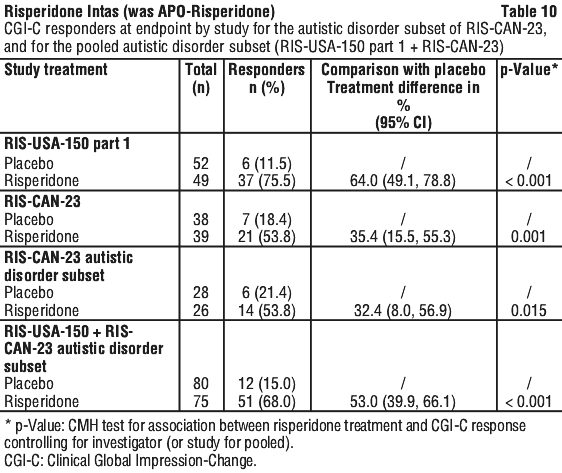 As few autistic children with an IQ > 84 are seen, there is limited clinical experience with risperidone in such children. Experience in autistic adolescents is also limited.
As few autistic children with an IQ > 84 are seen, there is limited clinical experience with risperidone in such children. Experience in autistic adolescents is also limited. Molecular formula: C23H27FN4O2.
Molecular formula: C23H27FN4O2.