What is in this leaflet
This leaflet answers some common questions about APREPITANT SCP.
It does not contain all of the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have benefits and risks. Your doctor has weighed the risks of you taking APREPITANT SCP against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, talk to your doctor or pharmacist.
Keep this leaflet with your medicine. You may need to read it again.
What APREPITANT SCP is used for
Chemotherapy Induced Nausea and Vomiting
APREPITANT SCP, in combination with other medicines, is used to prevent nausea (feeling sick) and vomiting associated with cancer chemotherapy.
Post-Operative Nausea and Vomiting
APREPITANT SCP is used to prevent nausea (feeling sick) and vomiting which can occur after surgery.
APREPITANT SCP belongs to a group of medicines called neurokinin 1 (NK1) receptor antagonists. It works by blocking the actions of substances in your brain, called substance P neurokinins, that cause nausea and vomiting.
APREPITANT SCP is available only with a doctor's prescription.
Your doctor may have prescribed APREPITANT SCP for another reason.
Ask your doctor if you have any questions about why APREPITANT SCP has been prescribed for you.
The safety and effectiveness of APREPITANT SCP in children and teenagers under the age of 18 years have not been established.
APREPITANT SCP is not addictive.
Before you take APREPITANT SCP
When you must not take it
Do not take APREPITANT SCP if you have an allergy to APREPITANT SCP or any of the ingredients listed at the end of the leaflet.
Do not take APREPITANT SCP if you are taking:
- cisapride, used to treat stomach reflux
- pimozide, used to treat psychotic conditions
- terfenadine (Teldane*) and astemizole (Hismanal*), antihistamines used for allergic conditions, including hayfever
* not available in Australia - St Johns Wort - a herb used to treat depression
Taking APREPITANT SCP with these medicines may cause serious or life threatening reactions.
Do not take APREPITANT SCP if you have a rare hereditary problem of fructose intolerance, glucose-galactose malabsorption, or sucrose-isomaltase insufficiency.
Do not take APREPITANT SCP if you are breast-feeding or plan to breast-feed.
It is not known if APREPITANT SCP passes into breast milk. You and your doctor should discuss whether you should stop breast-feeding or not take APREPITANT SCP.
Do not take APREPITANT SCP if the expiry date (Exp.) printed on the pack has passed.
Do not take APREPITANT SCP if the packaging is torn or shows signs of tampering. If you take this medicine after the expiry date has passed, it may not work.
If you are not sure whether you should start taking APREPITANT SCP, talk to your doctor.
Before you take it
Tell your doctor if:
- you have or have had any medical conditions
- you are pregnant or intend to become pregnant
APREPITANT SCP has not been studied in pregnant women. APREPITANT SCP should be used during pregnancy only if clearly needed. - you have any allergies to any other medicine or any other substances, such as foods, preservative or dyes.
If you have not told your doctor about any of the above, tell the doctor before you take any APREPITANT SCP.
Taking other medicines
Tell your doctor if you are taking any other medicines, including any that you buy without a prescription from a pharmacy, supermarket or health food shop.
Some medicines should not be taken with APREPITANT SCP. These include:
- cisapride, used to treat stomach reflux
- pimozide, used to treat psychotic conditions
- terfenadine (Teldane*) and astemizole (Hismanal*), antihistamines used for allergic conditions, including hayfever
*not available in Australia - St John’s Wort – a herb used to treat depression
Taking APREPITANT SCP with these medicines may cause serious or life threatening reactions.
Some medicines and APREPITANT SCP may interfere with each other. These include:
- warfarin, used to prevent blood clots.
Your doctor may order additional blood tests to check the effect of warfarin after you have taken APREPITANT SCP. - rifampicin, an antibiotic used to treat tuberculosis and other infections
- ketoconazole, used to treat fungal infections
- oral contraceptive pills (also known as the pill). Alternative or "back-up" measures of contraception should be used during treatment with APREPITANT SCP and for one month following the last dose of APREPITANT SCP
- paroxetine, used to treat depression, and obsessive compulsive and panic disorders
- diltiazem, used to treat angina and high blood pressure
- midazolam, triazolam, or alprazolam, used as sedatives or to treat anxiety or panic disorder
- dexamethasone or methylprednisolone, steroid medicines used for a variety of conditions
- certain cancer chemotherapy agents, including etoposide, vinorelbine, paclitaxel
- tolbutamide, used to treat diabetes
- phenytoin, used to treat epilepsy
These medicines may be affected by APREPITANT SCP or may affect how well it works. You may need different amounts of your medicine, or you may need to take different medicines.
Your doctor can tell you what to do if you are taking any of these medicines.
If you are not sure whether you are taking any of these medicines, check with your doctor or pharmacist.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking APREPITANT SCP.
How to take APREPITANT SCP
How much to take
Take APREPITANT SCP only when prescribed by your doctor.
Chemotherapy Induced Nausea and Vomiting
APREPITANT SCP may be given to you in one of two ways:
- APREPITANT SCP 1-Day Regimen Day 1 (day of chemotherapy):
One 165 mg capsule of APREPITANT SCP will be given to you to take by mouth 1 hour before you start your chemotherapy treatment on Day 1 only.
OR
- APREPITANT SCP 3-Day Regimen Day 1 (day of chemotherapy):
APREPITANT SCP 125 mg will be given to you by mouth 1 hour before you start your chemotherapy treatment on Day 1.
Day 2 and Day 3 (the two days after chemotherapy):
Take one 80 mg capsule of APREPITANT SCP each morning for the 2 days following your chemotherapy treatment.
Post-Operative Nausea and Vomiting
The recommended does APREPITANT SCP to prevent nausea and vomiting caused by surgery is one 40 mg capsule within 3 hours before your surgery.
Follow all directions given to you by your doctor and pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the carton, ask your doctor or pharmacist for help.
How to take APREPITANT SCP
Chemotherapy Induced Nausea and Vomiting
Swallow each capsule of APREPITANT SCP with a glass of water.
It does not matter if you take APREPITANT SCP before, with or after food. However, if you are taking the 165 mg capsule, take it either without food or with a light meal.
Post-Operative Nausea and Vomiting
Your doctor will give you one 40 mg capsule of APREPITANT SCP 3 hours before your surgery.
APREPITANT SCP can be taken with or without food. Follow your doctor’s instructions about eating or drinking before surgery.
How long to take APREPITANT SCP for
Chemotherapy Induced Nausea and Vomiting
APREPITANT SCP may be given in one of two ways. APREPITANT SCP 1-Day regimen is a 165 mg capsule given only on the day of chemotherapy treatment.
APREPITANT SCP 3-Day regimen is usually taken for 3 days.
Post-Operative Nausea and Vomiting
APREPITANT SCP is given as 1 dose before your surgery.
If you are not sure how long to take APREPITANT SCP, talk to your doctor.
If you forget to take APREPITANT SCP
If you forget to take your capsules, contact your doctor for instructions.
If you take too much APREPITANT SCP (overdose)
Immediately telephone your doctor, or the Poisons Information Centre (telephone 13 11 26), or go to Accident and Emergency at the nearest hospital, if you think you or anyone else may have taken too much APREPITANT SCP. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are using APREPITANT SCP
Things you must do
Women taking oral contraceptive pills for birth control should also use other methods of contraception during treatment with APREPITANT SCP and for one month following the last dose of APREPITANT SCP. This is because oral contraceptive pills may not work as well when taking APREPITANT SCP.
If you become pregnant while taking APREPITANT SCP, tell your doctor immediately.
If you are about to be started on any new medicine, tell your doctor and pharmacist that you are taking APREPITANT SCP.
Things you must not do
Do not give APREPITANT SCP to anyone else, even if they have the same condition as you.
Things to be careful of
Be careful driving or operating machinery until you know how APREPITANT SCP affects you.
APREPITANT SCP generally does not cause any problems with your ability to drive a car or operate machinery.
However, as with many medicines, it may cause certain side effects in some people, including tiredness and dizziness.
Things that may be helpful to manage your chemotherapy induced nausea and vomiting
Small, frequent meals or eating a snack before your chemotherapy treatment may help you to tolerate it better.
Talk to your doctor, pharmacist or nurse for more information.
Things that may be helpful to manage your nausea and vomiting caused by your surgery
Talk to your doctor about measures to manage your nausea and vomiting after surgery.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking APREPITANT SCP.
APREPITANT SCP helps most people with nausea and vomiting associated with cancer chemotherapy, but it may have unwanted side effects in few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice or have any of the following and they worry you:
- tiredness
- generally feeling unwell
- muscle weakness
- headache dizziness
- constipation, diarrhoea
- indigestion, heartburn, loss of appetite
- gas from the stomach or bowel, wind
- hiccups/hiccoughs
- vomiting
- disorientation
- chills
- hot flushes
- bloating
- pain on urination
- change to your walking pattern
- acne
These are the common side effects of APREPITANT SCP. For the most part these have been mild.
Tell your doctor immediately if you notice any of the following:
- slow, fast or irregular heart beat
- severe upper stomach pain
- symptoms of severe sunburn, such as redness, itching, pain, swelling or blistering
- signs of anaemia such as, being short of breath when exercising, looking pale
- frequent signs of infections such as fever, severe chills, sore throat or mouth ulcers
These may be serious side effects. You may need urgent medical attention. These side effects are rare.
If any of the following happen tell your doctor immediately or go emergency department at the nearest hospital:
- swelling of the face, lips, mouth, throat or tongue which may cause difficulty in breathing or swallowing
- pinkish, itchy swellings on the skin, also called hives or nettlerash
- severe skin reactions, including the inside of the nose or mouth
These may be serious side effects. If you have them, you may be having a serious allergic reaction to APREPITANT SCP. You may need urgent medical attention. These side effects are rare.
Other side effects not listed above may also occur in some patients. Tell your doctor if you notice anything that is making you feel unwell.
After using APREPITANT SCP
Storage
Keep APREPITANT SCP where children cannot reach it. A locked cupboard at least one- and-a-half metres above the ground is a good place to store medicines.
Keep your capsules in a cool dry place where the temperature stays below 25°C.
Do not store APREPITANT SCP or any other medicine in the bathroom or near a sink.
Do not leave APREPITANT SCP in the car or on window sills. Heat and dampness can destroy some medicines.
Disposal
If your doctor tells you to stop taking APREPITANT SCP, or your capsules have passed its expiry date, ask your pharmacist what to do with any that are left over.
Product description
What it looks like
APREPITANT SCP 40 mg:
- opaque hard gelatin capsules with a yellow cap and white body, printed with “40mg” on the body
APREPITANT SCP 80 mg:
- opaque hard gelatin capsules with a white cap and white body, printed with “80mg” on the body
APREPITANT SCP 125 mg:
- opaque hard gelatin capsules with a pink cap and white body, printed with “125mg” on the body
APREPITANT SCP 165 mg:
- opaque hard gelatin capsules with a blue cap and white body, printed with “165mg” on the body
Available in OPA/ALU/PVC - Aluminium foil blister packs of 1 capsules for each strength. And pack of two capsules for the 80 mg strength.
Ingredients
Active ingredient:
APREPITANT SCP 40 mg
Each capsules contains 40 mg of aprepitant.
APREPITANT SCP 80 mg
Each capsules contains 80 mg of aprepitant.
APREPITANT SCP 125 mg
Each capsules contains 125 mg of aprepitant.
APREPITANT SCP 165 mg
Each capsules contains 165 mg of aprepitant.
They also contain the inactive ingredients:
- hypromellose
- microcrystalline cellulose spheres
- poloxamer
- sucrose
Capsule shell ingredients:
- gelatin
- sodium lauryl sulfate
- titanium dioxide
- iron oxide yellow (40 mg)
- iron oxide red (125 mg)
- indigo carmine aluminium lake (165 mg).
The capsules do not contain gluten, tartrazine or any other azo dyes.
Supplier
Southern Cross Pharma Pty Ltd
Suite 5/118 Church Street
Hawthorn, Victoria 3122
Australia
Australian registration numbers:
APREPITANT SCP 40 mg
AUST R 310215
APREPITANT SCP 80 mg
AUST R 319700
APREPITANT SCP 125 mg
AUST R 310216
APREPITANT SCP 165 mg
AUST R 310220
Date of preparation: May 2023.
Published by MIMS May 2024






 In an additional active-controlled clinical study in 1169 patients receiving the 3-day oral aprepitant regimen and HEC, the adverse experience profile was generally similar to that seen in the other HEC studies with the 3-day oral aprepitant regimen.
In an additional active-controlled clinical study in 1169 patients receiving the 3-day oral aprepitant regimen and HEC, the adverse experience profile was generally similar to that seen in the other HEC studies with the 3-day oral aprepitant regimen.
 The following additional clinical adverse experiences (incidence > 0.5% and greater than ondansetron), regardless of causality, were reported in patients treated with aprepitant.
The following additional clinical adverse experiences (incidence > 0.5% and greater than ondansetron), regardless of causality, were reported in patients treated with aprepitant.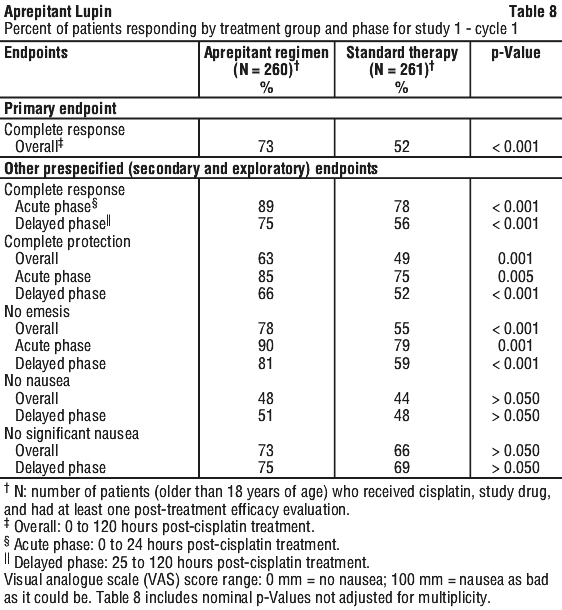
 In both studies, a statistically significant, higher proportion of patients receiving the aprepitant regimen in cycle 1 had a complete response (primary endpoint), compared with patients receiving standard therapy. A statistically significant difference in complete response in favour of the aprepitant regimen was also observed when the acute phase and the delayed phase were analysed separately.
In both studies, a statistically significant, higher proportion of patients receiving the aprepitant regimen in cycle 1 had a complete response (primary endpoint), compared with patients receiving standard therapy. A statistically significant difference in complete response in favour of the aprepitant regimen was also observed when the acute phase and the delayed phase were analysed separately.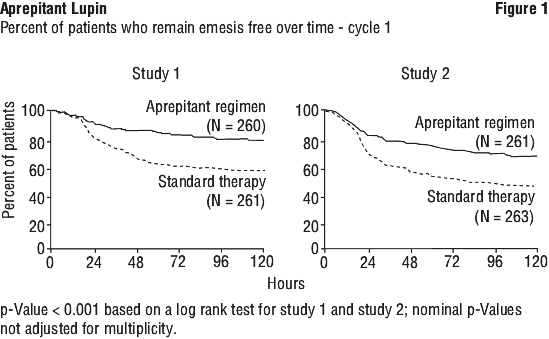

 The antiemetic activity of aprepitant was evaluated during the acute phase (0 to 24 hours post-chemotherapy treatment), the delayed phase (25 to 120 hours post-chemotherapy treatment) and overall (0 to 120 hours post-chemotherapy treatment) in cycle 1. Efficacy was based on evaluation of the following composite measures:
The antiemetic activity of aprepitant was evaluated during the acute phase (0 to 24 hours post-chemotherapy treatment), the delayed phase (25 to 120 hours post-chemotherapy treatment) and overall (0 to 120 hours post-chemotherapy treatment) in cycle 1. Efficacy was based on evaluation of the following composite measures: In this study, a statistically significantly (p = 0.015) higher proportion of patients receiving the aprepitant regimen (51%) in cycle 1 had a complete response (primary endpoint) during the overall phase compared with patients receiving standard therapy (42%). The unadjusted absolute difference in complete response (8.3%) represents a 20% relative improvement (relative risk ratio = 1.2, aprepitant regimen over standard therapy).
In this study, a statistically significantly (p = 0.015) higher proportion of patients receiving the aprepitant regimen (51%) in cycle 1 had a complete response (primary endpoint) during the overall phase compared with patients receiving standard therapy (42%). The unadjusted absolute difference in complete response (8.3%) represents a 20% relative improvement (relative risk ratio = 1.2, aprepitant regimen over standard therapy). In this study, a statistically significantly higher proportion of patients receiving the aprepitant regimen in cycle 1 reported no impact of nausea and vomiting on daily life, as measured by a FLIE total score > 108, compared with patients receiving standard therapy.
In this study, a statistically significantly higher proportion of patients receiving the aprepitant regimen in cycle 1 reported no impact of nausea and vomiting on daily life, as measured by a FLIE total score > 108, compared with patients receiving standard therapy. In a second multicentre, randomised, double-blind, parallel-group, clinical study, the aprepitant regimen was compared with standard therapy in 848 patients receiving a chemotherapy regimen that included any IV dose of oxaliplatin, carboplatin, epirubicin, idarubicin, ifosfamide, irinotecan, daunorubicin, doxorubicin; cyclophosphamide IV (< 1500 mg/m2); or cytarabine IV (> 1 g/m2). Patients who were randomised to receive the aprepitant regimen consisted of 76% women and 24% men. Patients receiving the aprepitant regimen were receiving chemotherapy for a variety of tumour types including 52% with breast cancer, 21% with gastrointestinal cancers including colorectal cancer, 13% with lung cancer and 6% with gynaecological cancers. The aprepitant regimen consisted of aprepitant 125 mg on day 1 and 80 mg/day on days 2 and 3 in combination with ondansetron 8 mg orally twice on day 1 plus dexamethasone 12 mg orally on day 1. Standard therapy consisted of placebo in combination with ondansetron 8 mg orally (twice on day 1, and every 12 hours on days 2 and 3) plus dexamethasone 20 mg orally on day 1.
In a second multicentre, randomised, double-blind, parallel-group, clinical study, the aprepitant regimen was compared with standard therapy in 848 patients receiving a chemotherapy regimen that included any IV dose of oxaliplatin, carboplatin, epirubicin, idarubicin, ifosfamide, irinotecan, daunorubicin, doxorubicin; cyclophosphamide IV (< 1500 mg/m2); or cytarabine IV (> 1 g/m2). Patients who were randomised to receive the aprepitant regimen consisted of 76% women and 24% men. Patients receiving the aprepitant regimen were receiving chemotherapy for a variety of tumour types including 52% with breast cancer, 21% with gastrointestinal cancers including colorectal cancer, 13% with lung cancer and 6% with gynaecological cancers. The aprepitant regimen consisted of aprepitant 125 mg on day 1 and 80 mg/day on days 2 and 3 in combination with ondansetron 8 mg orally twice on day 1 plus dexamethasone 12 mg orally on day 1. Standard therapy consisted of placebo in combination with ondansetron 8 mg orally (twice on day 1, and every 12 hours on days 2 and 3) plus dexamethasone 20 mg orally on day 1. In this study, a statistically significantly (p < 0.0001) higher proportion of patients receiving the aprepitant regimen (76%) in cycle 1 had no vomiting (primary endpoint) during the overall phase compared with patients receiving standard therapy (62%). In addition, a higher proportion of patients receiving the aprepitant regimen in cycle 1 had a complete response in the overall phase (0-120 hours), compared with patients receiving standard therapy. Aprepitant was numerically superior versus standard therapy regardless of age, gender, or tumor type (breast, gastrointestinal, lung or other) as assessed by the no vomiting and complete response endpoints.
In this study, a statistically significantly (p < 0.0001) higher proportion of patients receiving the aprepitant regimen (76%) in cycle 1 had no vomiting (primary endpoint) during the overall phase compared with patients receiving standard therapy (62%). In addition, a higher proportion of patients receiving the aprepitant regimen in cycle 1 had a complete response in the overall phase (0-120 hours), compared with patients receiving standard therapy. Aprepitant was numerically superior versus standard therapy regardless of age, gender, or tumor type (breast, gastrointestinal, lung or other) as assessed by the no vomiting and complete response endpoints. In this study, a statistically significantly higher proportion of patients receiving the aprepitant regimen in cycle 1 reported no impact of nausea and vomiting on daily life, as measured by a FLIE total score > 108, compared with patients receiving standard therapy.
In this study, a statistically significantly higher proportion of patients receiving the aprepitant regimen in cycle 1 reported no impact of nausea and vomiting on daily life, as measured by a FLIE total score > 108, compared with patients receiving standard therapy.
 Results presented in the table were obtained by use of a logistics model with terms for treatment and investigative sites. For the no vomiting endpoint, an additional analysis was performed for time to first event, where patients who received rescue medication were censored at the time of rescue. In study 090, results of this analysis show that the reduction in risk for a vomiting episode over the 0 to 24 hour period with aprepitant 40 mg relative to ondansetron 4 mg was 61.9% (95% C.I.: 32.1%, 78.6%). In study 091, results of this analysis show that reduction in risk for a vomiting episode over the 0 to 24 hour period with aprepitant 40 mg relative to ondansetron 4 mg was 48.7% (95% C.I.: 23.6%, 65.5%).
Results presented in the table were obtained by use of a logistics model with terms for treatment and investigative sites. For the no vomiting endpoint, an additional analysis was performed for time to first event, where patients who received rescue medication were censored at the time of rescue. In study 090, results of this analysis show that the reduction in risk for a vomiting episode over the 0 to 24 hour period with aprepitant 40 mg relative to ondansetron 4 mg was 61.9% (95% C.I.: 32.1%, 78.6%). In study 091, results of this analysis show that reduction in risk for a vomiting episode over the 0 to 24 hour period with aprepitant 40 mg relative to ondansetron 4 mg was 48.7% (95% C.I.: 23.6%, 65.5%). Oral administration of the 165 mg dose of aprepitant with a standard light (low-fat) breakfast and a high-fat breakfast resulted in up to an 8% and 47% increase in AUC0-∞ of aprepitant, respectively. It is recommended that aprepitant 165 mg be taken fasted or with a light (low fat) meal to minimise the potential for drug interactions see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
Oral administration of the 165 mg dose of aprepitant with a standard light (low-fat) breakfast and a high-fat breakfast resulted in up to an 8% and 47% increase in AUC0-∞ of aprepitant, respectively. It is recommended that aprepitant 165 mg be taken fasted or with a light (low fat) meal to minimise the potential for drug interactions see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.

