What is in this leaflet
This leaflet answers some common questions about APX-ESCITALOPRAM.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have benefits and risks. Your doctor has weighed the risks of you taking APX-ESCITALOPRAM against the benefits they expect it will have for you.
Talk to your doctor or pharmacist if you have any concerns about taking this medicine.
Keep this leaflet with your medicine. You may need to read it again.
What APX-ESCITALOPRAM is used for
APX-ESCITALOPRAM is used to treat depression.
It belongs to a group of medicines called selective serotonin reuptake inhibitors (SSRIs). These medicines work by their action on brain chemicals called amines which are involved in controlling mood.
Depression is longer lasting and/or more severe than the "low moods" everyone has from time to time due to the stress of everyday life. It is thought to be caused by a chemical imbalance in parts of the brain. This imbalance affects your whole body and can cause emotional and physical symptoms like feeling low in spirit, loss of interest in activities, being unable to enjoy life, poor appetite or overeating, disturbed sleep, often waking up early, loss of sex drive, lack of energy and feeling guilty over nothing. APX-ESCITALOPRAM corrects this chemical imbalance and may help relieve the symptoms of depression.
APX-ESCITALOPRAM may also be used to treat patients who may avoid and/or are fearful of social situations.
APX-ESCITALOPRAM may also be used to treat patients who have excessive anxiety and worry.
APX-ESCITALOPRAM may also be used to treat irrational fears or obsessional behaviour (obsessive-compulsive disorder). Obsessive-compulsive disorder involves having both obsessions and compulsions. Obsessions are unwanted thoughts that occur over and over again. Compulsions are the ongoing need to repeat certain actions as a result of these thoughts.
Your doctor may have prescribed this medicine for another reason. Ask your doctor if you have any questions about why it has been prescribed for you.
APX-ESCITALOPRAM is available only with a doctor's prescription.
There is no evidence that this medicine is addictive, however you may get side effects if you suddenly stop taking it.
Tell your doctor if you get any side effects after stopping APX-ESCITALOPRAM.
Before you take it
When you must not take it
Do not take APX-ESCITALOPRAM if you are allergic to medicines containing:
- escitalopram
- citalopram
- any of the ingredients listed at the end of this leaflet.
Symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty breathing, swelling of the face, lips, tongue or other parts of the body, or rash, itching or hives on the skin.
Do not take APX-ESCITALOPRAM if you are taking other medications including:
- pimozide, a medicine used to treat mental disorders
- monoamine oxidase inhibitors (MAOIs), such as phenelzine, tranylcypromine and moclobemide, which are also used for the treatment of depression.
- monoamine oxidase inhibitors such as linezolid which is an antibiotic and selegiline which is used in the treatment of Parkinson's Disease
One day must elapse after you have finished taking moclobemide before you start taking APX-ESCITALOPRAM. If you have taken any other MAOI you will need to wait 14 days. After stopping APX-ESCITALOPRAM you must allow 14 days before taking any MAOI including moclobemide.
Taking APX-ESCITALOPRAM with MAOIs may cause a serious reaction with a sudden increase in body temperature, extremely high blood pressure and severe convulsions.
Your doctor will know when it is safe to start APX-ESCITALOPRAM after the MAOI has been stopped.
Do not take it if the expiry date (Exp.) printed on the pack has passed. If you take it after the expiry date has passed, it may not work as well. The expiry date refers to the last day of the month
Do not take it if the packaging is torn or shows signs of tampering.
Before you start to take it
Tell your doctor if:
- you have allergies to any other medicines, foods, dyes or preservatives.
- you are pregnant or intend to become pregnant.
Medicines like APX-ESCITALOPRAM have been shown to reduce the quality of sperm in animal studies, which theoretically could affect fertility. If you are intending to start a family, ask your doctor for advice.
Do not take LEXAPRO if you are pregnant unless you and your doctor have discussed the risks and benefits involved.
Make sure your doctor and/or midwife know you are on APX-ESCITALOPRAM.
When taken during pregnancy, particularly in the last three months of pregnancy, medicines like APX-ESCITALOPRAM may affect the general condition of your newborn baby and may increase the risk of a serious condition in babies, called persistent pulmonary hypertension of the newborn (PPHN), making the baby breathe faster and appear bluish. These symptoms usually begin during the first 24 hours after the baby is born. If this happens to your baby you should contact your doctor and/or midwife immediately.
If used during pregnancy APX-ESCITALOPRAM should never be stopped abruptly.
- you are breast-feeding or planning to breast-feed.
Do not take APX-ESCITALOPRAM if you are breast-feeding unless you and your doctor have discussed the risks and benefits involved. It is not recommended that you breast-feed while taking APX-ESCITALOPRAM as it is excreted in breast milk. - you have, or have had, the following medical conditions:
- a tendency to bleed or bruise easily
- diabetes
- heart disease
- kidney disease
- liver disease
- manic depression (bipolar disorder)
- a history of seizures or fits
- restlessness and/or a need to move often
- raised intraocular pressure (fluid pressure in the eye), or if you are at risk of angle-closure glaucoma. - you are receiving electroconvulsive therapy
Do not give APX-ESCITALOPRAM to a child or adolescent. There is no experience with its use in children or adolescents under 18 years old.
APX-ESCITALOPRAM can be given to elderly patients over 65 years of age with a reduced dose. The effects of APX-ESCITALOPRAM in elderly patients are similar to those in other patients.
If you have not told your doctor about any of the above, tell them before you use APX-ESCITALOPRAM.
Taking other medicines
Tell your doctor if you are taking any other medicines, including those you buy without a prescription from a pharmacy, supermarket or health food shop.
Some medicines may be affected by APX-ESCITALOPRAM or may affect how well it works. These include:
- bupropion, a medicine used to help stop smoking
- medicines used to treat reflux and ulcers, such as cimetidine, omeprazole, esomeprazole and lansoprazole
- medicines known to prolong bleeding, such as aspirin or other non-steroidal anti-inflammatory drugs (NSAIDs)
- ticlopidine and warfarin, medicines used to prevent blood clots
- fluconazole, an anti-fungal medicine
- mefloquine, an anti-malaria medicine
- sumatriptan, used to treat migraine
- tramadol, used to relieve pain
- medicines affecting the chemicals in the brain
- some heart medications, such as flecainide, propafenone, metoprolol
- tryptophan, an amino acid
- lithium, used to treat mood swings and some types of depression
- antipsychotics, medicines to treat mental disorders, such as risperidone, thioridazine and haloperidol
- tricyclic antidepressants, eg imipramine, desipramine
- St. John’s Wort (Hypericum perforatum), an herbal remedy
- Any other medicines for depression, anxiety, obsessive compulsive disorder or premenstrual dysphoric disorder.
These medicines may be affected by APX-ESCITALOPRAM, or may affect how well it works. You may need to use different amounts of your medicines, or take different medicines. Your doctor will advise you.
Some combinations of medicines may increase the risk of serious side effects and are potentially life threatening.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking APX-ESCITALOPRAM.
How to take it
How much to take
Your doctor will tell you how much APX-ESCITALOPRAM to take.
The usual starting dose is 10 mg daily. Your doctor may increase the dose, depending on your response. The maximum recommended dose is 20 mg per day.
In elderly patients, the maximum recommended dose is 10 mg per day.
In patients with liver disease, an initial dose of 5 mg daily is recommended for the first two weeks. Your doctor may then increase the dose to 10 mg daily.
Your doctor may have prescribed a different dose.
Ask your doctor or pharmacist if you are unsure of the correct dose for you. They will tell you exactly how much to take.
Follow the instructions they give you. If you take the wrong dose, APX-ESCITALOPRAM may not work as well and your condition may not improve.
How to take APX-ESCITALOPRAM
Swallow the tablets whole with a glass of water.
The tablets can be halved, but it is best not to crush or chew.
Take APX-ESCITALOPRAM as a single dose either in the morning or in the evening, with or without food.
How long to take it
Continue taking APX-ESCITALOPRAM even if it takes some time before you feel any improvement in your condition. As with other medicines for the treatment of these conditions, it may take a few weeks before you feel any improvement.
Individuals vary greatly in their response to APX-ESCITALOPRAM. Your doctor will check your progress at regular intervals.
The duration of treatment may vary but is usually at least 6 months.
In some cases, the doctor may decide a longer treatment period.
Keep taking this medicine for as long as your doctor recommends, even if you begin to feel better. The underlying illness may persist for a long time. If you stop your treatment too soon, your symptoms may return.
Do not stop taking it suddenly. If APX-ESCITALOPRAM is stopped suddenly you may experience mild, but usually temporary, symptoms such as dizziness, pins and needles, electric shock sensations, sleep disturbances (vivid dreams, inability to sleep), feeling anxious or agitated, headaches, feeling sick (nausea), vomiting, sweating, tremor (shaking), feeling confused, feeling emotional or irritable, diarrhoea, visual disturbances, or fast or irregular heartbeats.
When you have completed your course of treatment, the dose of APX-ESCITALOPRAM is gradually reduced over a couple of weeks rather than stopped abruptly.
Your doctor will tell you how to reduce the dosage so that you do not get unwanted side effects.
If you forget to take it
If you miss a dose and remember in less than 12 hours, take it straight away, and then go back to taking your tablets as you would normally the next day.
Otherwise, skip that day's dose but be sure to take the next day's dose when it is due.
Do not take a double dose to make up for the dose you missed.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to take your medicine, ask your pharmacist for hints.
If you take too much (overdose)
Immediately telephone your doctor, or the Poisons Information Centre (telephone 13 11 26) or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have taken too much APX-ESCITALOPRAM. Do this even if there are no signs of discomfort or poisoning.
You may need urgent medical attention.
If you take too much APX-ESCITALOPRAM, you may have the following symptoms:
- nausea
- vomiting
- dizziness
- low blood pressure
- drowsiness
- agitation
- tremor
- convulsions or coma.
While you are taking it
Things you must do
Before starting any new medicine, tell your doctor or pharmacist that you are taking APX-ESCITALOPRAM.
Tell all the doctors, dentists and pharmacists who are treating you that you are taking this medicine.
If you become pregnant while taking it, tell your doctor immediately.
Persons taking APX-ESCITALOPRAM may be more likely to think about killing themselves or actually trying to do so, especially when APX-ESCITALOPRAM is first started or the dose is changed.
Tell your doctor immediately if you have any suicidal thoughts or other mental/mood changes or if you are close to or care for someone using APX-ESCITALOPRAM who talks about or shows signs of killing him or herself. All mentions of suicide or violence must be taken seriously.
Occasionally, the symptoms of depression may include thoughts of suicide or self-harm. It is possible that these symptoms continue or get worse until the full antidepressant effect of the medicine becomes apparent. This is more likely to occur if you are a young adult, i.e. 18 to 24 years of age, and you have not used antidepressant medicines before.
If you or someone you know is showing any of the following warning signs of suicide-related behaviour while taking APX-ESCITALOPRAM, it is important to contact a health care provider right away or even go to the nearest hospital for treatment:
- thoughts or talk of death or suicide
- thoughts or talk of self-harm or harm to others
- any recent attempts of self-harm
- increase in aggressive behaviour, irritability or agitation.
Do not stop taking this medicine or change the dose without consulting your doctor, even if you experience increased anxiety at the beginning of treatment. At the beginning of treatment, some patients may experience increased anxiety which will disappear during continued treatment.
Tell your doctor immediately if you experience symptoms such as restlessness or difficulty in sitting or standing still. These symptoms can occur during the first weeks of treatment.
Contact your doctor as soon as possible if you suddenly experience an episode of mania. Some patients with bipolar disorder (manic depression) may enter into a manic phase. This is characterised by profuse and rapidly changing ideas, exaggerated gaiety and excessive physical activity.
Sometimes you may be unaware of the above-mentioned symptoms and therefore you may find it helpful to ask a friend or relative to help you to observe the possible signs of change in your behaviour.
Things you must not do
Do not give the tablets to anyone else, even if they have the same condition as you.
Do not take APX-ESCITALOPRAM to treat any other complaints unless your doctor tells you to.
Do not stop taking APX-ESCITALOPRAM, or lower the dose, without checking with your doctor.
Do not let yourself run out of tablets over the weekend or on holidays.
Suddenly stopping APX-ESCITALOPRAM may cause unwanted discontinuation symptoms such as dizziness, sensory disturbances, sleep disturbances, agitation or anxiety tremor, confusion, sweating, headache, diarrhoea, palpitations, emotional instability, irritability, visual disturbances and nausea. Your doctor will tell you when and how APX-ESCITALOPRAM should be discontinued. Your doctor will gradually reduce the amount you are using, usually over a period of one to two weeks, before stopping completely.
Things to be careful of
Be careful driving or operating machinery until you know how APX-ESCITALOPRAM affects you. It may cause visual disturbances (such as blurred vision), nausea, fatigue and dizziness in some people, especially early in the treatment. If you have any of these symptoms, do not drive, operate machinery, or do anything else that could be dangerous.
Avoid alcohol while you are taking this medicine. It is not recommended that you drink alcohol while you are being treated for depression.
Side effects
All medicines may have some unwanted side effects. Sometimes they are serious, but most of the time they are not. Your doctor has weighed the risks of using this medicine against the benefits he/she expects it will have for you.
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking APX-ESCITALOPRAM. It helps most people with depression, social anxiety disorder (social phobia), generalised anxiety disorder and obsessive-compulsive disorder, but it may have unwanted side effects in a few people.
The side effects of APX-ESCITALOPRAM are, in general, mild and disappear after a short period of time.
Tell your doctor if you notice any of the following and they worry you:
- decreased or loss of appetite
- dry mouth
- diarrhoea
- nausea (feeling sick)
- sleeplessness
- feeling sleepy, drowsiness, fatigue, yawning
- increased sweating
- sexual disturbances (decreased sex drive, in men - delayed ejaculation or problems with erection; in women – difficulty in getting orgasm).
*The side effects marked with an asterisk (*) are a number of rare side effects that are known to occur with medicines that work in a similar way to APX-ESCITALOPRAM.
Tell your doctor as soon as possible if you notice any of the following:
- agitation, confusion, panic attacks*, anxiety, restlessness*
- dizziness
- dizziness when you stand up due to low blood pressure*
- Fast heart rate or decrease in heart rate or irregular heartbeat
- low levels of sodium in the blood (the symptoms are feeling sick and unwell with weak muscles or confused) *
- abnormal liver function test (increased amounts of liver enzymes in the blood) *
- difficulty urinating*
- unusual secretion of breast milk*
- bleeding disorders including skin mucous bleeding (e.g. bruising*) and low levels of blood platelets*
- rash, itching, hives, patches of circumscribed swellings
- An increased risk of bone fractures has been observed in patients taking this type of medicine*
These are serious side effects and you may need urgent medical attention.
Tell your doctor immediately, or go to Accident and Emergency at the nearest hospital if you notice any of the following:
- Thoughts of harming yourself or thoughts of suicide*, see also section “Things you must do”
- Serious allergic reaction, the symptoms of which may include swelling of the face, lips, mouth or throat which can cause difficulty in swallowing or breathing
- high fever, agitation, confusion, trembling and abrupt contractions of muscles, symptoms of a rare condition called serotonin syndrome
- mania* (i.e.: elevated mood and associated symptoms)
- hallucinations
- seizures, tremors, movement disorders (involuntary movements of the muscles) *
- fast, irregular heartbeat with feelings of dizziness or difficulty breathing.
These are very serious side effects. You may need urgent medical attention or hospitalisation.
Tell your doctor if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some patients.
An increased risk of bone fractures has been observed in patients taking medicines like APX-ESCITALOPRAM.
Do not be alarmed by this list of side effects. You may not experience any of them.
After using it
Storage
Keep APX-ESCITALOPRAM where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Keep your tablets in the blister pack until it is time to take them.
Keep your tablets in a cool dry place where the temperature stays below 25°C.
Do not store APX-ESCITALOPRAM or any other medicine in the bathroom or near a sink.
Do not leave it in the car or on windowsills. Heat and dampness can destroy some medicines.
Disposal
If your doctor tells you to stop taking it, or your tablets have passed their expiry date, ask your pharmacist what to do with any that are left over.
Return any unused medicine to your pharmacist.
Product description
What it looks like
APX-ESCITALOPRAM comes in two strengths of tablets:
- 10 mg tablet - white, oval convex shaped, coated tablet with ‘E’ bisect ‘1’ on one side and plain on the other side
- 20 mg tablet - white, oval convex shaped, coated tablet with ‘E’ bisect ‘2’ on one side and plain on the other side.
Each pack contains 28 tablets.
Ingredients
The active ingredient in APX-ESCITALOPRAM is escitalopram (as oxalate):
- each APX-ESCITALOPRAM 10 tablet contains 10 mg of escitalopram
- each APX-ESCITALOPRAM 20 tablet contains 20 mg of escitalopram.
The tablets also contain:
- microcrystalline cellulose
- colloidal anhydrous silica
- purified talc
- croscarmellose sodium
- magnesium stearate
- Opadry II complete film coating system 85F28751 WHITE (ARTG No 1027).
The tablets do not contain gluten, lactose, sucrose, tartrazine or any other azo dyes.
Manufacturer
Arrow Pharma Pty Ltd
15-17 Chapel Street
Cremorne VIC 3121
Australia
Australian registration numbers:
APX-ESCITALOPRAM 10 - AUST R 333535
APX-ESCITALOPRAM 20 - AUST R 333536
This leaflet was revised in May 2021.
Published by MIMS October 2021





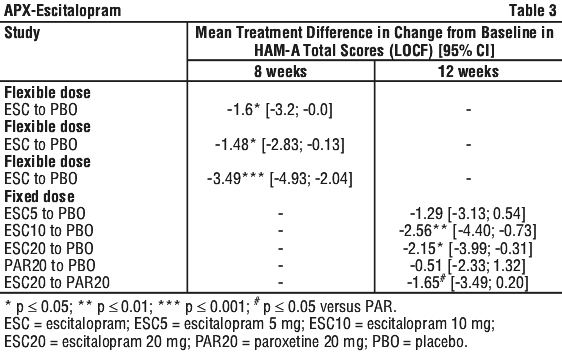 In the pooled analysis of these three placebo-controlled, flexible-dose studies of similar design, the mean change from baseline in HAM-A total score improved statistically significantly (LOCF) over time in the escitalopram group relative to the placebo group. The separation from placebo was first observed at week 1 and continued through to the end of the study (week 8). The treatment difference to placebo at week 8 was -2.3 in favour of escitalopram (p ≤ 0.01).
In the pooled analysis of these three placebo-controlled, flexible-dose studies of similar design, the mean change from baseline in HAM-A total score improved statistically significantly (LOCF) over time in the escitalopram group relative to the placebo group. The separation from placebo was first observed at week 1 and continued through to the end of the study (week 8). The treatment difference to placebo at week 8 was -2.3 in favour of escitalopram (p ≤ 0.01). The beneficial effect of long-term treatment with escitalopram was also reflected in the analyses of responders and remitters in this study. The analyses showed a further increase both in the proportion of responders and in the proportion of remitters from week 12 to week 24, especially in the escitalopram 20 mg group. At week 24, a statistically significantly greater proportion of responders and remitters were seen in all three escitalopram dose groups (except for the proportion of responders in the 10 mg group) than in the placebo group (p ≤ 0.01) (see Tables 5 and 6).
The beneficial effect of long-term treatment with escitalopram was also reflected in the analyses of responders and remitters in this study. The analyses showed a further increase both in the proportion of responders and in the proportion of remitters from week 12 to week 24, especially in the escitalopram 20 mg group. At week 24, a statistically significantly greater proportion of responders and remitters were seen in all three escitalopram dose groups (except for the proportion of responders in the 10 mg group) than in the placebo group (p ≤ 0.01) (see Tables 5 and 6).
 In the relapse prevention study, the primary efficacy analysis showed a statistically significantly superior effect of escitalopram relative to placebo on the time to relapse of SAD (log-rank test, p ≤ 0.001). Furthermore, patients treated with escitalopram had fewer protocol-defined relapses than those treated with placebo. In addition, patients treated with escitalopram showed a further improvement in mean LSAS total score during the double-blind period, while patients treated with placebo showed deterioration.
In the relapse prevention study, the primary efficacy analysis showed a statistically significantly superior effect of escitalopram relative to placebo on the time to relapse of SAD (log-rank test, p ≤ 0.001). Furthermore, patients treated with escitalopram had fewer protocol-defined relapses than those treated with placebo. In addition, patients treated with escitalopram showed a further improvement in mean LSAS total score during the double-blind period, while patients treated with placebo showed deterioration. Furthermore, escitalopram 20 mg/day was significantly more efficacious than placebo on the Y-BOCS subscale of rituals at week 12. Both escitalopram 10 mg/day and escitalopram 20 mg/day were significantly more efficacious than placebo on the Y-BOCS subscale of obsessions as well as on the NIMH-OCS total score, CGI-I score and CGI-S score.
Furthermore, escitalopram 20 mg/day was significantly more efficacious than placebo on the Y-BOCS subscale of rituals at week 12. Both escitalopram 10 mg/day and escitalopram 20 mg/day were significantly more efficacious than placebo on the Y-BOCS subscale of obsessions as well as on the NIMH-OCS total score, CGI-I score and CGI-S score. Results after 24 weeks showed that both escitalopram 10 mg/day (p < 0.05) and escitalopram 20 mg/day (p < 0.01) were significantly more efficacious than placebo as measured by the primary outcome measure, the Y-BOCS total score, as well as on the secondary subscales of Y-BOCS (obsessions and rituals) and the NIMH-OCS score (escitalopram 10 mg/day (p < 0.01) and escitalopram 20 mg/day (p < 0.001)). See Table 9.
Results after 24 weeks showed that both escitalopram 10 mg/day (p < 0.05) and escitalopram 20 mg/day (p < 0.01) were significantly more efficacious than placebo as measured by the primary outcome measure, the Y-BOCS total score, as well as on the secondary subscales of Y-BOCS (obsessions and rituals) and the NIMH-OCS score (escitalopram 10 mg/day (p < 0.01) and escitalopram 20 mg/day (p < 0.001)). See Table 9. The beneficial efficacy of long-term treatment with escitalopram was also demonstrated by the analyses of responders and remitters in this study.
The beneficial efficacy of long-term treatment with escitalopram was also demonstrated by the analyses of responders and remitters in this study. Responders at the end of the above 16-week open-label treatment phase (escitalopram 10 mg: 30 responders; escitalopram 20 mg: 133 responders) entered the 24-week randomised, double-blind placebo-controlled relapse prevention phase. Both escitalopram 10 mg/day (p=0.014) and 20 mg/day (p < 0.001) showed significantly fewer relapses.
Responders at the end of the above 16-week open-label treatment phase (escitalopram 10 mg: 30 responders; escitalopram 20 mg: 133 responders) entered the 24-week randomised, double-blind placebo-controlled relapse prevention phase. Both escitalopram 10 mg/day (p=0.014) and 20 mg/day (p < 0.001) showed significantly fewer relapses.
