1 Name of Medicine
Recombinant Respiratory Syncytial Virus pre-fusion F protein (RSVPreF3) (AS01E adjuvanted vaccine).
2 Qualitative and Quantitative Composition
After reconstitution, 1 dose (0.5 mL) contains 120 micrograms of RSVPreF31 antigen adjuvanted with AS01E2.
1 Respiratory syncytial virus (RSV) glycoprotein F stabilised in the pre-fusion conformation (RSVPreF3) produced by recombinant DNA technology in Chinese Hamster Ovary (CHO) cells.
2 The GlaxoSmithKline proprietary AS01E Adjuvant System is composed of the plant extract Quillaja saponaria saponin (QS-21) (25 micrograms) and 3-O-desacyl-4'-monophosphoryl lipid A (MPL) from Salmonella minnesota (25 micrograms).
List of excipients with known effect.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Powder and suspension for suspension for injection.
The powder is white.
The suspension is an opalescent, colourless to pale brownish liquid.
4.1 Therapeutic Indications
Arexvy is indicated for active immunisation for the prevention of lower respiratory tract disease caused by respiratory syncytial virus (RSV) in:
adults 60 years of age and older;
adults 50 through 59 years of age who are at increased risk for RSV disease.
The use of this vaccine should be in accordance with official recommendations.
4.2 Dose and Method of Administration
Dosage.
Arexvy is administered as a single dose of 0.5 mL.
Currently there is not enough evidence to determine the need for revaccination.
Method of administration.
Arexvy is for intramuscular injection only, preferably in the deltoid muscle.
For instructions on reconstitution of the medicinal product before administration, see Use and handling.
Use and handling.
The powder and suspension should be inspected visually for any foreign particulate matter and/or variation of appearance. If either is observed, do not reconstitute the vaccine.
How to prepare Arexvy.
Arexvy must be reconstituted prior to administration.
1. Withdraw the entire contents of the vial containing the suspension into a syringe.
2. Add the entire contents of the syringe into the vial containing the powder.
3. Gently swirl until the powder is completely dissolved.
The reconstituted vaccine is an opalescent, colourless to pale brownish liquid.
The reconstituted vaccine should be inspected visually for any foreign particulate matter and/or variation of appearance. If either is observed, do not administer the vaccine.
After reconstitution, the vaccine should be used promptly; if not possible, the vaccine should be stored in the refrigerator (2°C - 8°C) or at room temperature up to 25°C. If not used within 4 hours it should be discarded.
Before administration.
1. Withdraw 0.5 mL of the reconstituted vaccine into the syringe.
2. Change the needle so that you are using a new needle.
Administer the vaccine intramuscularly.
Arexvy is for single use in one patient only. Discard any residue.
Any unused medicinal product or waste material should be disposed of in accordance with local requirements.4.3 Contraindications
Hypersensitivity to the active substances or to any component of the vaccine (see Section 2 Qualitative and Quantitative Composition; Section 6.1 List of Excipients).
4.4 Special Warnings and Precautions for Use
Prior to immunisation.
As with all injectable vaccines, appropriate medical treatment and supervision should always be readily available in case of an anaphylactic event following the administration of the vaccine.
As with other vaccines, vaccination with Arexvy should be postponed in individuals suffering from an acute severe febrile illness. However, the presence of a minor infection, such as a cold, should not result in the deferral of vaccination.
As with any vaccine, a protective immune response may not be elicited in all vaccinees.
Syncope (fainting) can occur following, or even before, any vaccination as a psychogenic response to the needle injection. It is important that procedures are in place to avoid injury from faints.
Precautions for use.
Do not administer the vaccine intravascularly or intradermally. No data are available on subcutaneous administration of Arexvy.
As with other vaccines administered intramuscularly, Arexvy should be given with caution to individuals with thrombocytopenia or any coagulation disorder since bleeding may occur following an intramuscular administration to these individuals.
Systemic immunosuppressive medications and immunodeficiency.
Safety and immunogenicity data on Arexvy are not available for immunocompromised individuals. Patients receiving immunosuppressive treatment or patients with immunodeficiency may have a reduced immune response to Arexvy.
Guillain-Barré syndrome.
Guillain-Barré syndrome has been reported very rarely following vaccination with Arexvy in individuals ≥ 60 years (see Section 4.8 Adverse Effects (Undesirable Effects)).
Healthcare professionals should closely monitor signs and symptoms of Guillain-Barré syndrome in all Arexvy recipients to ensure correct diagnosis and to rule out other causes. If Guillain-Barré syndrome is diagnosed, prompt management with adequate supportive care and treatment is recommended.
Use in the elderly.
There are no special precautions for use in the elderly.
Paediatric use.
The safety and efficacy of Arexvy have not been established in children and adolescents.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
Use with other vaccines.
Arexvy can be given concomitantly with inactivated seasonal influenza vaccines (standard dose unadjuvanted, high dose unadjuvanted, or standard dose adjuvanted). The safety profile of Arexvy when co-administered with inactivated seasonal influenza vaccines was comparable to when Arexvy was administered alone. There was no evidence of interference in the immune response to the antigens contained in the co-administered vaccines (see Section 5.1 Pharmacodynamic Properties, Clinical trials). For adverse effects, please see Section 4.8 Adverse Effects (Undesirable Effects).
Data are currently not available for concomitant administration with other vaccines.
If Arexvy is to be given at the same time as another injectable vaccine, the vaccines should always be administered at different injection sites.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
There are no data on the effects of Arexvy on human fertility.
Animal studies do not indicate direct or indirect harmful effects with respect to fertility in females.
In rabbits, no effects on female fertility were observed following intramuscular injection of Arexvy (120 micrograms of RSVPreF3 adjuvanted with AS01E, 10 times the clinical dose on a microgram/m2 body surface area basis) per animal.
(Category B2)
There are no data from the use of Arexvy in pregnant women. Arexvy is not recommended during pregnancy.
After administration of an investigational unadjuvanted RSVPreF3 vaccine to 3557 pregnant women in a single clinical study, an increase in preterm births was observed compared to placebo.
Results from animal studies do not indicate direct or indirect harmful effects with respect to developmental and reproductive toxicity.
In a reproductive and developmental toxicity study, female rats were administered AS01B adjuvant alone by intramuscular injection 28 and 14 days prior to mating, on gestation days 3, 8, 11, and 15, and on lactation Day 7. The total dose was similar to the dose of AS01E in Arexvy. No adverse effects on preweaning development up to post-natal day 25 were observed. There was no vaccine related fetal malformations or variations.
In another reproductive and developmental toxicity study, female rabbits were administered the AS01B adjuvant by intramuscular injection 28 and 14 days prior to mating, on gestation days 3, 11, 16, and 24, and on lactation Day 7. The total dose was 2 times the dose of AS01E in Arexvy. AS01B produced no adverse effects on embryofetal or pre- and post-natal survival, growth or development of the offspring up to day 35 of age.
There are no data on the excretion of Arexvy in human or animal milk. Arexvy is not recommended in breastfeeding/lactating women.4.7 Effects on Ability to Drive and Use Machines
No studies on the effects of Arexvy on the ability to drive and use machines have been performed. However, some of the effects mentioned under Section 4.8 Adverse Effects (Undesirable Effects) (e.g. fatigue) may temporarily affect the ability to drive or use machines.
4.8 Adverse Effects (Undesirable Effects)
Clinical trial data.
The safety profile presented below is based on a placebo-controlled Phase III clinical study RSV OA=ADJ-006 (conducted in Europe, North America, Asia and Southern hemisphere) in adults ≥ 60 years of age in which 12,467 adults received one dose of Arexvy and 12,499 received placebo with a follow-up period of approximately 12 months.
Adverse drug reactions (ADRs) are listed (see Table 1) by MedDRA system organ class and by frequency.
Very common ≥ 1/10; common ≥ 1/100 to < 1/10; uncommon ≥ 1/1,000 to < 1/100; rare ≥ 1/10,000 to < 1/1,000; very rare < 1/10,000.
 Safety data from RSV OA=ADJ-006.
Safety data from RSV OA=ADJ-006. Solicited adverse reactions.
A subset of study participants (solicited safety set) was monitored for solicited adverse reactions using standardised paper diary cards during the 4 days (i.e. day of vaccination and the next 3 days) following a dose of Arexvy or placebo. The other study participants did not prospectively record solicited reactions on a diary card but may have reported them as unsolicited adverse reactions.
The reported frequencies of specific solicited local (administration site) and systemic adverse reactions (per participant) are presented in Table 2.
 In the solicited safety set, the local administration site adverse reactions reported with Arexvy had a median duration of 2 days, and the systemic adverse reactions reported with Arexvy had a median duration ranging between 1 and 2 days.
In the solicited safety set, the local administration site adverse reactions reported with Arexvy had a median duration of 2 days, and the systemic adverse reactions reported with Arexvy had a median duration ranging between 1 and 2 days.
Unsolicited adverse events.
In all participants from RSV OA=ADJ-006, unsolicited adverse events were monitored using paper diary cards during the 30-day period following vaccination (day of vaccination and the next 29 days).
Among participants in the solicited safety set, (Arexvy, n = 879 or placebo, n = 878), unsolicited adverse events occurring within 30 days after vaccination were reported in 14.9% and 14.6% of participants who received Arexvy and placebo, respectively.
In the exposed set, 24,966 participants 60 years of age and older, received at least 1 dose of Arexvy (n = 12,467) or placebo (n = 12,499). Unsolicited adverse events occurring within 30 days of vaccination were reported in 33.0% and 17.8% of participants, respectively. The higher frequency of reported unsolicited adverse events among participants who received Arexvy, compared to participants who received placebo, was primarily attributed to events that are consistent with adverse reactions solicited among participants in the reactogenicity subset.
Within 30 days after vaccination, atrial fibrillation was reported in 10 participants who received Arexvy and 4 participants who received placebo (of which 7 events in Arexvy arm and 1 event in placebo arm were serious); the onset of symptoms ranged from 1 to 30 days post vaccination. The currently available information on the atrial fibrillation is insufficient to determine a causal relationship to the vaccine.
Serious adverse events.
In RSV OA=ADJ-006, participants were monitored for all serious adverse events (SAEs) that occurred during the 6-month period following administration of Arexvy (n = 12,467) or placebo (n = 12,499).
SAEs with onset within 6 months following vaccination were reported at similar rates in participants who received Arexvy (4.2%) or placebo (4.0%).
Deaths.
From vaccination through the first analysis of the ongoing RSV OA=ADJ-006, adverse events leading to death were reported for 49 participants (0.4%) who received Arexvy (n = 12,467) and 58 participants (0.5%) who received placebo (n = 12,499). Based on available information, there is no evidence of causal relationship to Arexvy. Causes of death among participants were consistent with those generally reported in adult and elderly populations.
Potential immune-mediated diseases.
In RSV OA=ADJ-006, participants were monitored for all potential immune-mediated diseases (pIMDs) that occurred during the 6-month period following administration of Arexvy (n = 12,467) or placebo (n = 12,499).
New onset pIMDs or exacerbation of existing pIMDs within 6 months following vaccination were reported for 0.3% of participants who received Arexvy and 0.3% of participants who received placebo. There were no notable imbalances between study groups in individual pIMDs reported.
Safety data from RSV OA=ADJ-007. In the open-label Phase III RSV OA=ADJ-007 clinical study, participants 60 years of age and older received 1 dose of Arexvy and inactivated unadjuvanted seasonal influenza vaccine (Flu Quadrivalent containing a combined total of 60 micrograms Hemagglutinin (HA) per dose) either concomitantly at Day 1 (N = 442), or 1 dose of Flu Quadrivalent at Day 1 followed by a dose of Arexvy 1 month later (N = 443).
The reported frequencies of specific solicited local (administration site) and systemic adverse reactions are presented in Table 3.
 Safety data from RSV OA=ADJ-018. Additionally, in a placebo-controlled Phase III clinical study, 769 participants 50 through 59 years of age (including 386 participants with pre-defined, stable, chronic medical conditions leading to an increased risk for RSV disease) and 381 participants 60 years of age and older received one dose of Arexvy.
Safety data from RSV OA=ADJ-018. Additionally, in a placebo-controlled Phase III clinical study, 769 participants 50 through 59 years of age (including 386 participants with pre-defined, stable, chronic medical conditions leading to an increased risk for RSV disease) and 381 participants 60 years of age and older received one dose of Arexvy.
Solicited adverse reactions.
The reported frequencies of specific solicited, local (administration site), and systemic adverse reactions among participants 50 through 59 years of age are presented in Table 4.
 The rates of solicited local and systemic adverse reactions (Table 4) were similar in participants 50 through 59 years of age either with or without pre-defined, stable, chronic medical conditions associated with an increased risk for LRTD caused by RSV.
The rates of solicited local and systemic adverse reactions (Table 4) were similar in participants 50 through 59 years of age either with or without pre-defined, stable, chronic medical conditions associated with an increased risk for LRTD caused by RSV.
Overall, the median duration of solicited local adverse reactions and solicited systemic adverse reactions after Arexvy vaccination was 2-3 days and 1-2 days, respectively.
Unsolicited adverse events.
Unsolicited adverse events within 30 days after vaccination were reported in 13.8% of participants, 50 through 59 years of age, who received Arexvy (N = 769) compared to 12.0% of participants who received placebo (N = 383). Within 30 days after vaccination, there were no cases of atrial fibrillation reported in participants 50 through 59 years of age.
Serious adverse events.
In RSV OA=ADJ-018 participants were monitored for all SAEs that occurred during the 6-month period following administration of Arexvy (N = 769) or placebo (N = 383). Among participants 50 through 59 years of age, SAEs with onset within 6 months post vaccination were reported in 2.3% of those who received Arexvy and 2.1% of those who received placebo.
Deaths.
There were no adverse events leading to death reported within 6 months following vaccination.
Potential immune-mediated diseases.
In RSV OA=ADJ-018, participants were monitored for all pIMDs that occurred during the 6-month period following administration of Arexvy (N = 769) or placebo (N = 383). Among participants 50 through 59 years of age, new onset pIMDs or exacerbation of existing pIMDs with onset within 6 months post vaccination were reported in 0.5% of those who received Arexvy and 0.3% of those who received placebo. There were no notable imbalances between study groups in individual pIMDs reported.
Serious adverse events reported from other studies. Study RSV OA=ADJ-004 (NCT04732871).
One case of Guillain-Barré syndrome beginning 9 days after Arexvy vaccination was reported in a participant enrolled in a study site in Japan.
Post-marketing experience.
See Table 5.
 Description of selected adverse events.
Description of selected adverse events. Guillain-Barré syndrome.
The results of a post-marketing observational study over 1 RSV season in patients 65 years or older suggest an increased risk of Guillain-Barré syndrome (estimated 7 excess cases per million doses administered) during the 42 days following vaccination with Arexvy. However, available evidence is insufficient to establish a causal relationship.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
Insufficient data are available.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
The RSVPreF3 antigen in Arexvy is derived from the laboratory-adapted RSV-A A2 strain, stabilised in the pre-fusion conformation of the naturally occurring F protein for which both RSV-A and B subtypes share high amino acid sequence homology. The risk of developing RSV-associated LRTD increases with age and with presence of underlying comorbidities. Arexvy induces the functional humoral immune responses against the RSV-A and RSV-B subtypes and the antigen-specific cellular immune responses which contribute to protect against RSV-associated LRTD (see Immunogenicity of Arexvy).
In a Phase I/II clinical trial, formulation adjuvanted with AS01E showed the ability to induce RSVPreF3-specific CD4+ T cells in adults 60 to 80 years of age to levels similar to those observed in young adults, despite lower baseline levels in the older adults.
Non-clinical data show that AS01E induces a local and transient activation of the innate immune system. The adjuvant effect of AS01E is the result of interactions between MPL and QS-21 formulated in liposomes. This facilitates the recruitment and activation of antigen presenting cells carrying vaccine-derived antigens in the draining lymph node, which in turn leads to the generation of RSVPreF3-specific CD4+ T cells and induction of RSV-A and RSV-B neutralising titres. In addition, RSVPreF3 formulated with AS01E can elicit specific binding antibodies directed to site O/, a highly neutralising sensitive epitope, exposed only on the pre-fusion conformation of the F protein.
Clinical trials.
Efficacy of Arexvy. Efficacy of Arexvy against RSV-associated LRTD in adults 60 years and older was evaluated for up to 3 RSV seasons in RSV OA=ADJ-006, a Phase III, randomised, placebo-controlled, observer-blind clinical study conducted in 17 countries from Northern and Southern Hemispheres.
The study excluded participants who were immunocompromised. Participants with pre-existing, chronic, stable disease such as diabetes, hypertension, or cardiac disease were allowed to participate in the study if considered by the investigator as medically stable at the time of vaccination.
The primary population for efficacy analysis (referred to as the modified Exposed Set, included adults 60 years of age and older receiving 1 dose of Arexvy or placebo and who did not report an RSV-confirmed acute respiratory illness (ARI) prior to Day 15 after vaccination).
The primary efficacy analysis set over the first RSV season included 24,960 participants who received 1 dose of Arexvy (N = 12,466) or placebo (N = 12,494).
Pre-season 2, participants who received Arexvy were re-randomised to receive placebo (n = 4,991) or a second dose of Arexvy (n = 4,966). Participants who received placebo before Season 1 received a second dose of placebo before Season 2. The participants were followed up to the end of the third RSV season (median follow-up time 30.6 months).
The median age of participants was 69 years (range: 59 to 102 years), with approximately 74% over 65 years of age, approximately 44% over 70 years of age and approximately 8% over 80 years of age. Approximately 52% were female. At baseline, 39.3% of participants had at least one comorbidity of interest; 19.7% of participants had an underlying cardiorespiratory condition (COPD, asthma, any chronic respiratory/pulmonary disease, or chronic heart failure) and 25.8% of participants had endocrinometabolic conditions (diabetes, advanced liver or renal disease). Among participants in the modified Exposed Set for the analysis of efficacy over 2 and over 3 RSV seasons, demographic and baseline characteristics were similar to those in the modified Exposed Set for the analysis of efficacy over the first RSV season.
Using the Gait speed test, 38.3% of participants were ranked as pre-frail (0.4-0.99m/s walking speed) and 1.5% as frail (< 0.4 m/s walking speed or who were not able to perform the test).
Confirmed RSV cases were determined by quantitative Reverse Transcription Polymerase Chain Reaction (qRT-PCR) on nasopharyngeal swab during all ARI episodes. ARI was defined by the presence of at least 2 respiratory symptoms/signs for at least 24 hours (nasal congestion, sore throat, lower respiratory symptoms/signs, as described below), or at least 1 respiratory symptom/sign + 1 systemic symptom/sign (fever or feverishness, fatigue, body aches, headache, decreased appetite) for at least 24 hours.
LRTD was defined based on the following criteria: the participant must have experienced at least 2 lower respiratory symptoms/signs including at least 1 lower respiratory sign for at least 24 hours, or experienced at least 3 lower respiratory symptoms for at least 24 hours. Lower respiratory symptoms included: new or increased sputum, new or increased cough, new or increased dyspnea (shortness of breath). Lower respiratory signs included: new or increased wheezing, crackles/rhonchi, respiratory rate ≥ 20 respirations/min, low or decreased oxygen saturation (O2 saturation < 95% or ≤ 90% if baseline is < 95%) or need for oxygen supplementation.
Severe RSV-associated LRTD was defined as qRT-PCR confirmed RSV-associated LRTD with at least 2 lower respiratory signs, or preventing normal, everyday activities or requiring supportive therapy.
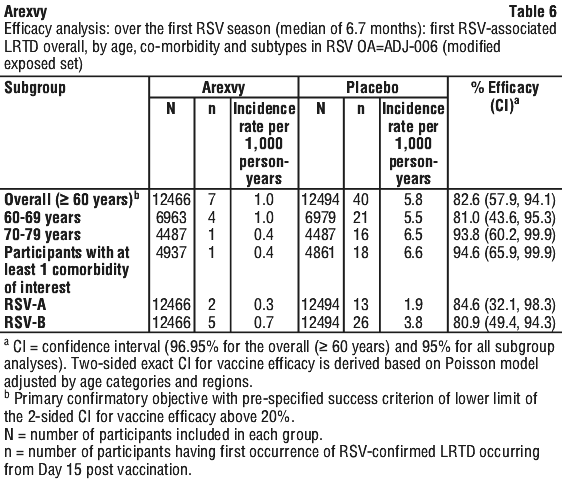
Efficacy against RSV-associated LRTD over the first RSV season (median of 6.7 months).
The primary objective was to demonstrate the efficacy of Arexvy in the prevention of a first episode of confirmed RSV-A and/or B associated LRTD during the first season.
Compared with placebo, Arexvy significantly reduced the risk of developing RSV-associated LRTD by 82.6% (96.95% CI: [57.9, 94.1]) in participants 60 years of age and older, which met the pre-specified success criterion for the primary study objective (see Table 2). High vaccine efficacy against RSV-LRTD is observed through the median follow-up period of 6.7 months.
The vaccine efficacy against RSV A-associated LRTD and RSV B-associated LRTD was 84.6% (95% CI [32.1, 98.3]) and 80.9% (95% CI [49.4, 94.3]), respectively. See Table 6.
 Compared with placebo, Arexvy significantly reduced the risk of developing RSV-associated LRTD by 84.4% (95% CI: [46.9, 97.0]) in participants 70 years of age and older.
Compared with placebo, Arexvy significantly reduced the risk of developing RSV-associated LRTD by 84.4% (95% CI: [46.9, 97.0]) in participants 70 years of age and older.
Compared with placebo, Arexvy significantly reduced the risk of developing RSV-associated LRTD in pre-frail participants by 92.9% (95% CI [53.4, 99.8]). The vaccine efficacy in the frail subgroup (189 participants in Arexvy vs 177 participants in placebo) cannot be reliably estimated due to the low number of total cases accrued (2 cases).
Efficacy against severe RSV-associated LRTD and RSV-associated ARI over the first RSV season (median of 6.7 months).
Compared with placebo, Arexvy significantly reduced the risk of developing severe RSV-associated LRTD by 94.1% (95% CI [62.4, 99.9]) in participants 60 years of age and older. One case of severe RSV-associated LRTD in the Arexvy group and 17 cases in the placebo group were reported, amongst which 2 cases required supportive therapy (oxygen supplementation).
Arexvy significantly reduced the risk of developing confirmed RSV-associated ARI in adults ≥ 60 years of age by 71.7% (95% CI [56.2, 82.3]).
Efficacy against RSV-associated LRTD over 2 RSV seasons (median of 17.8 months) and over 3 RSV seasons (median of 30.6 months).
Participants 60 years of age and older who received 1 dose of Arexvy or placebo were followed over 3 RSV seasons (up to the end of the second and third season in the Northern Hemisphere), with a median follow-up time of 17.8 months over 2 RSV seasons and 30.6 months over 3 RSV seasons. The vaccine efficacy against RSV-associated LRTD over 2 RSV seasons was 67.2% (97.5% CI [48.2, 80.0]) in participants 60 years of age and older (30 cases in the Arexvy group and 139 cases in the placebo group) and over 3 RSV seasons was 62.9% (97.5% CI [46.7, 74.8]).
The vaccine efficacy against RSV A-associated LRTD and RSV B-associated LRTD over 3 RSV seasons was 69.8% (97.5% CI [42.2, 85.7]) and 58.6% (97.5% CI [35.9, 74.1]), respectively.
The vaccine efficacy analyses by age subgroup and for participants with at least one comorbidity of interest are presented in Table 7.
 Subgroup analyses of RSV-associated LRTD vaccine efficacy over 2 RSV seasons and 3 RSV seasons showed similar efficacy point estimates. In participants 70 years of age and older, over 2 RSV seasons and over 3 RSV seasons, the vaccine efficacy against RSV-associated LRTD was 69.3% (95% CI [43.4, 84.6]) and 66.0% (95% CI [44.3, 80.2]), respectively.
Subgroup analyses of RSV-associated LRTD vaccine efficacy over 2 RSV seasons and 3 RSV seasons showed similar efficacy point estimates. In participants 70 years of age and older, over 2 RSV seasons and over 3 RSV seasons, the vaccine efficacy against RSV-associated LRTD was 69.3% (95% CI [43.4, 84.6]) and 66.0% (95% CI [44.3, 80.2]), respectively.
The vaccine efficacy against severe RSV-associated LRTD over 2 RSV seasons was 78.8% (95% CI [52.6, 92.0]) in participants 60 years of age and older (7 cases in the Arexvy group and 48 cases in the placebo group, amongst which 1 case in the Arexvy group and 5 cases in the placebo group required supportive therapy [oxygen supplementation and positive airway pressure]). The vaccine efficacy against severe RSV-associated LRTD over 3 RSV seasons was 67.4% (95% CI [42.4, 82.7]) in participants 60 years of age and older (15 cases in the Arexvy group and 75 cases in the placebo group, amongst which 2 cases in the Arexvy group and 5 cases in the placebo group required supportive therapy [oxygen supplementation and positive airway pressure]).
Efficacy against RSV-associated LRTD over the second RSV season and over the third RSV season.
The vaccine efficacy against RSV-associated LRTD over the second RSV season with median follow-up of 6.3 months was 56.1% (95% CI [28.2,74.4]) in participants 60 years of age and older (20 cases in the Arexvy group and 91 in the placebo group).
The vaccine efficacy against RSV-associated LRTD over the third RSV season with median follow-up of 7.0 months was 48.0% (95% CI [8.7, 72.0]) in participants 60 years of age and older (16 cases in the Arexvy group and 61 cases in the placebo group).
Immunogenicity of Arexvy.
An immunological correlate of protection has not been established; therefore, the level of immune response that provides protection against RSV-associated LRTD is unknown.
Adults 60 years and older.
The immune responses to Arexvy were evaluated in a Phase III immunogenicity and safety study RSV OA=ADJ-004 in adults 60 years and older. Functional humoral immune responses post-vaccination compared to pre-vaccination were evaluated with results from 940 participants for RSV-A and 941 participants for RSV-B for month 1 vs. pre-vaccination, and 928 participants for RSV-A and 929 participants for RSV-B at month 6 vs. pre-vaccination. The cell-mediated immune responses were evaluated with results from 471 participants at pre-vaccination, 410 at month 1 and 440 at month 6.
Arexvy elicited RSV-specific humoral and cellular immune responses. The geometric mean increase of the RSV-A and RSV-B neutralising titres compared to pre-vaccination were 10.5-fold (95% CI [9.9, 11.2]) and 7.8-fold (95% CI [7.4, 8.3]) at 1-month post-vaccination, respectively, and 4.4-fold (95% CI [4.2, 4.6]) and 3.5-fold (95% CI [3.4, 3.7]) at 6-months post-vaccination, respectively. The median frequency (percentile [25th, 75th]) of the RSVPreF3-specific CD4+ T-cells (per million of CD4+ T cells) was 1339.0 (829.0, 2136.0) 1-month post-vaccination and 666.0 (428.0, 1049.5) 6-months post-vaccination as compared to 191.0 (71.0, 365.0) pre-vaccination.
Adults 50 through 59 years of age at increased risk for RSV disease.
The non-inferiority of the immune response to Arexvy in adults 50 through 59 years of age compared to adults 60 years of age and older, where vaccine efficacy against RSV-associated LRTD was demonstrated, was evaluated in a Phase III, observer-blind, randomised, placebo-controlled study (RSV OA=ADJ-018).
Cohort 1 consisted of participants 50 through 59 years of age separated in 2 sub-cohorts according to their medical history. Sub-cohort 1 consisted of participants with pre-defined, stable, chronic medical conditions leading to an increased risk for RSV disease (Arexvy, N=386; placebo, N=191) such as chronic pulmonary disease, chronic cardiovascular disease, diabetes, chronic kidney or liver disease. Sub-cohort 2 consisted of participants without pre-defined, stable, chronic medical conditions (Arexvy, N=383; placebo, N=192). Cohort 2 consisted of participants 60 years of age and older (Arexvy, N=381).
The primary immunogenicity objective was to demonstrate non inferiority of the humoral immune response (in terms of RSV-A and RSV-B neutralising titres) following the administration of Arexvy at 1-month post-vaccination in participants 50-59 years of age with pre-defined, stable, chronic medical conditions leading to an increased risk for RSV disease, compared to participants 60 years of age and older.
The prespecified criteria for non-inferiority of the immune responses were defined as the 2-sided (95%) CI upper limits (UL) on the group geometric mean titre (GMT) ratios ≤ 1.50 and the UL of the 2-sided (95%) CIs on the Seroresponse Rate (SRR) difference ≤ 10% for the RSV-A and RSV-B neutralising titres in participants 60 years of age and older relative to participants 50 through 59 years of age with pre-defined, stable, chronic medical conditions leading to an increased risk for RSV disease. See Table 8.
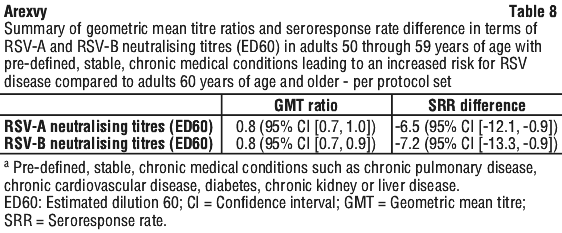 The non-inferiority criteria of the immune responses for the RSV-A and RSV-B neutralising titres were met. The efficacy of Arexvy, in adults 50 through 59 years of age at increased risk for RSV disease, can be inferred following comparison of the immune response in adults 50 through 59 years of age with the immune response in adults 60 years of age and older in which vaccine efficacy was demonstrated.
The non-inferiority criteria of the immune responses for the RSV-A and RSV-B neutralising titres were met. The efficacy of Arexvy, in adults 50 through 59 years of age at increased risk for RSV disease, can be inferred following comparison of the immune response in adults 50 through 59 years of age with the immune response in adults 60 years of age and older in which vaccine efficacy was demonstrated.
Immunogenicity following concomitant vaccination.
In three open-label Phase III clinical studies, participants were randomised to receive 1 dose of Arexvy administered either concomitantly at day 1 or separately (1 month apart) with inactivated seasonal quadrivalent influenza vaccine (standard dose unadjuvanted, adults ≥ 60 years of age, N=885; high dose unadjuvanted, adults ≥ 65 years of age, N=1,029; or standard dose adjuvanted, adults ≥ 65 years of age, N=1,045). The prespecified criteria for non-inferiority of the immune responses were defined as the 2-sided 95% confidence interval upper limits (UL) on the group geometric mean titre (GMT) ratios ≤ 1.50 for the RSV-A neutralising titres and haemagglutinin inhibition titres against each of the Flu strains, in the separate administration versus co-administration groups.
There was no evidence of interference in the immune response to RSV-A or any of the four Flu antigens when Arexvy was co-administered with standard dose unadjuvanted or high dose unadjuvanted seasonal influenza vaccines.
Upon coadministration of Arexvy with standard dose adjuvanted seasonal influenza vaccine, there was no evidence of clinically relevant interference in the immune response to RSV-A or any of the four Flu antigens. The UL of the GMT ratio was ≤ 1.50 for RSV A and three out of four Flu strains. For Flu A/H3N2, the UL of the GMT ratio was 1.53.
Immune responses for the RSV-B neutralising titres were comparable in the separate administration versus co-administration groups in all three studies.
Currently no clinical data is available on coadministration of Arexvy with other vaccines.
5.2 Pharmacokinetic Properties
Not relevant to vaccines.
5.3 Preclinical Safety Data
Genotoxicity.
Arexvy was not tested for genotoxicity.
Carcinogenicity.
Arexvy was not tested for carcinogenicity.6 Pharmaceutical Particulars
6.1 List of Excipients
Powder (RSVPreF3 antigen).
Trehalose dihydrate, polysorbate 80, monobasic potassium phosphate, dibasic potassium phosphate.
Suspension (AS01E adjuvant system).
Dioleoylphosphatidylcholine, cholesterol, sodium chloride, dibasic sodium phosphate, monobasic potassium phosphate, water for injections.
6.2 Incompatibilities
This medicinal product must not be mixed with other medicinal products.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
For shelf-life after reconstitution of the medicinal product, see Section 4.2 Dose and Method of Administration, Use and handling.
6.4 Special Precautions for Storage
Store in a refrigerator (2°C - 8°C).
Do not freeze. Discard if the vial has been frozen.
Store in the original package in order to protect from light.
For storage conditions after reconstitution of the medicinal product, see Section 4.2 Dose and Method of Administration, Use and handling.
6.5 Nature and Contents of Container
Arexvy is presented as:
Powder for 1 dose in a vial (type I glass) with stopper (butyl rubber).
Suspension for 1 dose in a vial (type I glass) with a stopper (butyl rubber).
Arexvy is available in a pack size of 1 vial of powder plus 1 vial of suspension or in a pack size of 10 vials of powder plus 10 vials of suspension.
Not all strengths, dose forms, pack sizes, container types may be distributed in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Not relevant to vaccines.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Summary Table of Changes



 In the solicited safety set, the local administration site adverse reactions reported with Arexvy had a median duration of 2 days, and the systemic adverse reactions reported with Arexvy had a median duration ranging between 1 and 2 days.
In the solicited safety set, the local administration site adverse reactions reported with Arexvy had a median duration of 2 days, and the systemic adverse reactions reported with Arexvy had a median duration ranging between 1 and 2 days.
 The rates of solicited local and systemic adverse reactions (Table 4) were similar in participants 50 through 59 years of age either with or without pre-defined, stable, chronic medical conditions associated with an increased risk for LRTD caused by RSV.
The rates of solicited local and systemic adverse reactions (Table 4) were similar in participants 50 through 59 years of age either with or without pre-defined, stable, chronic medical conditions associated with an increased risk for LRTD caused by RSV.
 Compared with placebo, Arexvy significantly reduced the risk of developing RSV-associated LRTD by 84.4% (95% CI: [46.9, 97.0]) in participants 70 years of age and older.
Compared with placebo, Arexvy significantly reduced the risk of developing RSV-associated LRTD by 84.4% (95% CI: [46.9, 97.0]) in participants 70 years of age and older. Subgroup analyses of RSV-associated LRTD vaccine efficacy over 2 RSV seasons and 3 RSV seasons showed similar efficacy point estimates. In participants 70 years of age and older, over 2 RSV seasons and over 3 RSV seasons, the vaccine efficacy against RSV-associated LRTD was 69.3% (95% CI [43.4, 84.6]) and 66.0% (95% CI [44.3, 80.2]), respectively.
Subgroup analyses of RSV-associated LRTD vaccine efficacy over 2 RSV seasons and 3 RSV seasons showed similar efficacy point estimates. In participants 70 years of age and older, over 2 RSV seasons and over 3 RSV seasons, the vaccine efficacy against RSV-associated LRTD was 69.3% (95% CI [43.4, 84.6]) and 66.0% (95% CI [44.3, 80.2]), respectively. The non-inferiority criteria of the immune responses for the RSV-A and RSV-B neutralising titres were met. The efficacy of Arexvy, in adults 50 through 59 years of age at increased risk for RSV disease, can be inferred following comparison of the immune response in adults 50 through 59 years of age with the immune response in adults 60 years of age and older in which vaccine efficacy was demonstrated.
The non-inferiority criteria of the immune responses for the RSV-A and RSV-B neutralising titres were met. The efficacy of Arexvy, in adults 50 through 59 years of age at increased risk for RSV disease, can be inferred following comparison of the immune response in adults 50 through 59 years of age with the immune response in adults 60 years of age and older in which vaccine efficacy was demonstrated.