Notes
Distributed by Medsurge Healthcare Pty Ltd
1 Name of Medicine
Argipressin.
2 Qualitative and Quantitative Composition
Argipressin Medsurge is a sterile, aqueous solution of synthetic argipressin (8-arginine argipressin). Argipressin is a polypeptide hormone having the properties of causing the contraction of vascular and other smooth muscles and of antidiuresis. Each 1 mL vial contains 20 pressor units of argipressin.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Sterile solution for injection. A clear colourless solution.
4.1 Therapeutic Indications
Argipressin Medsurge is used in the prevention and treatment of postoperative abdominal distention; in abdominal radiography to dispel interfering gas shadows and in diabetes insipidus.
4.2 Dose and Method of Administration
Intravenous use of argipressin is not recommended. Subcutaneous or intramuscular dosage should not exceed 0.75 mL.
Dosage should be appropriately reduced in use in children.
Abdominal distention.
In the average postoperative adult patient, give 0.25 mL (5 units) by i.m. or s.c. initially, increasing to 0.5 mL (10 units) at subsequent injections if necessary. Injections may be repeated at three or four hourly intervals as required.
Argipressin used in this manner will frequently prevent, or relieve, postoperative distention. These recommendations apply also to distention complicating pneumonia or other acute toxaemias.
Abdominal radiography.
For the average case, two i.m. or s.c. injections of 0.5 mL each (10 units) are suggested. These should be given two hours and one half hour, respectively, before films are exposed. Many radiologists advise giving an enema prior to the first dose of argipressin.
Diabetes insipidus.
Argipressin may be given by i.m. or s.c. injections. The dosage by injection is 0.25 to 0.5 mL (5 to 10 units) repeated two or three times daily as needed.
Product is for single use in one patient only. Discard any residue.4.3 Contraindications
Argipressin is contraindicated in patients who are hypersensitive to the drug or any of the excipients. Chronic nephritis with nitrogen retention contraindicates the use of argipressin until reasonable nitrogen blood levels have been attained.
4.4 Special Warnings and Precautions for Use
This drug should not be used in patients with vascular disease, especially disease of the coronary arteries, except with extreme caution. In such patients, even small doses may precipitate anginal pain and with larger doses, the possibility of myocardial infarction should be considered.
Argipressin may produce water intoxication. The early signs of drowsiness, listlessness and headaches should be recognised to prevent terminal coma and convulsions.
Argipressin should not be administered intravenously; subcutaneous or intramuscular dosage should not exceed 0.75 mL.
Argipressin should be used cautiously in the presence of epilepsy, migraine, asthma, toxaemia of pregnancy, nephritis with arterial hypertension, goitre with cardiac complications, coronary thrombosis, angina pectoris and arteriosclerosis or any state in which a rapid addition to extracellular water may produce hazard for an already overburdened system.
Use in the elderly.
No data available.
Paediatric use.
No data available.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
The following drugs may potentiate the antidiuretic effect of argipressin when used concurrently: carbamazepine, urea, fludrocortisone, tricyclic antidepressants.
The following drugs may decrease the antidiuretic effect of argipressin when used concurrently: democlocycline, noradrenaline, lithium, heparin, alcohol.
Ganglionic blocking agents may produce a marked increase in sensitivity to the pressor effects of argipressin.
Isolated cases of severe bradycardia and heart block have been reported in patients receiving argipressin and H2-antagonists.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No data available.
(Category B2)
Medicines taken by only a limited number of pregnant women and women of child-bearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human foetus having been observed.
Studies in animals are inadequate or may be lacking, but available data show no evidence of an increased occurrence of foetal damage.
No data available.4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Local or systemic allergic reactions may occur in hypersensitive individuals. The following side effects have been reported following the administration of argipressin: tremor, sweating, vertigo, circumoral pallor, 'pounding' in head, abdominal cramps, passage of gas, nausea, vomiting, urticaria, bronchial constriction, arrhythmias, decreased cardiac output, angina, myocardial ischaemia, peripheral vasoconstriction, gangrene, rhabdomyolysis and cutaneous gangrene. Anaphylaxis (cardiac arrest and/or shock) has been observed shortly after injection of argipressin.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia) or in New Zealand on 0800 764 766.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
The antidiuretic action of argipressin is ascribed to its ability to increase resorption of water by the renal tubules.
Argipressin can cause contraction of smooth muscle of the gastrointestinal tract and of all parts of the vascular bed, especially the capillaries, small arterioles and venules, with less effect on the smooth musculature of the large veins. The direct effect on the contractile elements is neither antagonised by adrenergic blocking agents nor prevented by vascular denervation.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Genotoxicity.
No data available.
Carcinogenicity.
No data available.6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium acetate trihydrate, hydrochloric acid, sodium hydroxide and water for injections.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2°C to 8°C. (Refrigerate. Do not freeze). Protect from light.
6.5 Nature and Contents of Container
Argipressin Medsurge is supplied as pack of 10 x 1 mL glass vials.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
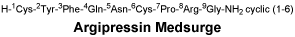 Molecular formula: C46H65N15O12S2. Molecular weight: 1084.24.
Molecular formula: C46H65N15O12S2. Molecular weight: 1084.24.
CAS number.
113-79-1.7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Summary Table of Changes


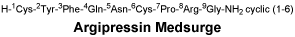 Molecular formula: C46H65N15O12S2. Molecular weight: 1084.24.
Molecular formula: C46H65N15O12S2. Molecular weight: 1084.24.