Notes
Distributed by Arrotex Pharmaceuticals Pty Ltd.
1 Name of Medicine
Liraglutide.
2 Qualitative and Quantitative Composition
ARX-Liraglutide contains liraglutide, a human glucagon-like peptide-1 (GLP-1) analogue that binds to and activates the GLP-1 receptor (GLP-1R).
ARX-Liraglutide is a solution for injection in a pre-filled pen. One mL contains 6 mg salt-free anhydrous liraglutide. One pre-filled pen contains 18 mg liraglutide in 3 mL.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
ARX-Liraglutide is a solution for injection. It is a sterile, aqueous, clear, colourless or almost colourless solution, pH=8.15.
4.1 Therapeutic Indications
ARX-Liraglutide is indicated as an adjunct to a reduced-calorie diet and increased physical activity for weight management in adult patients with an initial Body Mass Index (BMI) of ≥ 30 kg/m2 (obese) or ≥ 27 kg/m2 to < 30 kg/m2 (overweight) in the presence of at least one weight related comorbidity, such as dysglycaemia (pre-diabetes and type 2 diabetes mellitus), hypertension, dyslipidaemia, or obstructive sleep apnoea.
Treatment with ARX-Liraglutide should be discontinued after 12 weeks on the 3.0 mg/day dose if a patient has not lost at least 5% of their initial body weight.
4.2 Dose and Method of Administration
Liraglutide has not been studied in patients taking insulin. ARX-Liraglutide and insulin should not be used together (see Section 4.4 Special Warnings and Precautions for Use).
ARX-Liraglutide and other products containing liraglutide or any other GLP-1 receptor agonist should not be used at the same time.
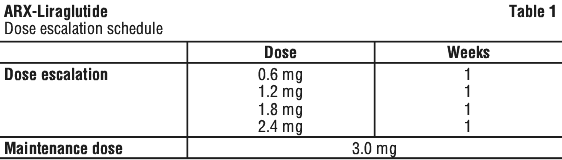
Dosage.
The starting dose is 0.6 mg once daily. The dose should be increased to 3.0 mg daily in increments of 0.6 mg with at least one week intervals to improve gastro-intestinal tolerability (see Table 1). If escalation to the next dose step is not tolerated for two consecutive weeks, consider discontinuing treatment. Daily doses higher than 3.0 mg are not recommended.
 The need for continued treatment should be re-evaluated whenever a new prescription is written and at least annually.
The need for continued treatment should be re-evaluated whenever a new prescription is written and at least annually.
Method of administration.
ARX-Liraglutide is for subcutaneous use only. It must not be administered intravenously or intramuscularly.
ARX-Liraglutide is administered once daily at any time, independent of meals. ARX-Liraglutide pen is for use by one person only. It should be injected in the abdomen, thigh or upper arm. Injection sites should always be rotated within the same region in order to reduce the risk of cutaneous amyloidosis [see Section 4.8 Adverse Effects (Undesirable Effects)]. The injection site and timing can be changed without dose adjustment. However it is preferable that ARX-Liraglutide is injected around the same time of the day, when the most convenient time of the day has been chosen.
If a dose is missed within 12 hours from when it is usually taken, the patient should take the dose as soon as possible. If there is less than 12 hours to the next dose, the patient should not take the missed dose and resume the once-daily regimen with the next scheduled dose. An extra dose or increase in dose should not be taken to make up for the missed dose.
ARX-Liraglutide should not be mixed with other injectable medicinal products (e.g. infusion fluids [see Section 4.4 Special Warnings and Precautions for Use]).
Dosage adjustment.
Patients with type 2 diabetes. When initiating ARX-Liraglutide, consider reducing the dose of concomitantly administered insulin or insulin secretagogues (such as sulfonylureas) to reduce the risk of hypoglycaemia [see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)].
ARX-Liraglutide is not a substitute for insulin.
Specific patient groups. Elderly (> 65 years old).
No dose adjustment is required based on age. Therapeutic experience with patients ≥ 75 years of age is limited and use in these patients is not recommended. ARX-Liraglutide should be used with caution in patients aged 65-74 years. [See Section 4.4 Special Warnings and Precautions for Use; Section 5.2 Pharmacokinetic Properties.]
Patients with hepatic impairment.
ARX-Liraglutide is not recommended in patients with hepatic impairment [see Section 4.4 Special Warnings and Precautions for Use].
Patients with renal impairment.
No dose adjustment is required for patients with mild or moderate renal impairment (creatinine clearance ≥ 30 mL/min). There is limited experience in patients with severe renal impairment (creatinine clearance < 30 mL/min). ARX-Liraglutide is currently not recommended for use in patients with severe renal impairment including patients with end-stage renal disease [see Section 4.4 Special Warnings and Precautions for Use; Section 5.2 Pharmacokinetic Properties].
Children and adolescents.
The safety and efficacy of liraglutide in children and adolescents below 18 years of age have not been established [see Section 5.1 Pharmacodynamic Properties]. No data are available. ARX-Liraglutide is not indicated for use in paediatric patients.
Special precautions for disposal and other handling.
ARX-Liraglutide should not be used if it does not appear clear and colourless or almost colourless.
ARX-Liraglutide should not be used if it has been frozen.
The pen is designed to be used with 32G disposable needles up to a length of 8 mm. Injection needles are not included.
The patient should be advised to discard the injection needle after each injection and store the pen without a needle attached. This may prevent blocked needles, contamination, infection, leakage of solution and inaccurate dosing.4.3 Contraindications
ARX-Liraglutide is not to be used in patients with hypersensitivity to liraglutide or any of its excipients.
4.4 Special Warnings and Precautions for Use
General.
ARX-Liraglutide must not be used as a substitute for insulin in patients with diabetes mellitus.
ARX-Liraglutide and insulin should not be used together. Liraglutide has not been studied in patients taking insulin.
ARX-Liraglutide is not indicated for the treatment of type 2 diabetes mellitus.
ARX-Liraglutide is not indicated in patients with obesity secondary to endocrinological or eating disorders or to treatment with medicinal products that may cause weight gain.
ARX-Liraglutide is not recommended in combination with other medicinal products intended for weight loss, including prescription medicines, over-the-counter medicines, and complementary medicines/herbal preparations. Efficacy and safety have not been established.
Aspiration in association with general anaesthesia or deep sedation.
Cases of pulmonary aspiration have been reported in patients receiving GLP-1 RAs undergoing general anaesthesia (GA) or deep sedation despite reported adherence to preoperative fasting recommendations. Therefore, the increased risk of residual gastric content because of delayed gastric emptying should be considered prior to performing procedures with GA or deep sedation.
Cardiovascular events.
Increase in heart rate.
An increase in heart rate with liraglutide was observed in clinical trials [see Section 4.8 Adverse Effects (Undesirable Effects)].
Heart rate should be monitored at regular intervals consistent with good clinical practice. Patients should be informed of the symptoms of increased heart rate (palpitations or feelings of a racing heartbeat while at rest). For patients who experience a sustained increase in resting heart rate, ARX-Liraglutide should be discontinued.
The effect on the heart rate of co-administration of liraglutide with other medicines that increase heart rate (e.g. sympathomimetic drugs) has not been evaluated. Consequently, co-administration of ARX-Liraglutide with these medicines should be undertaken with caution.
Dehydration, renal impairment and acute renal failure.
Patients treated with ARX-Liraglutide should be advised of the potential risk of dehydration in relation to gastrointestinal side effects and take precautions to avoid fluid depletion.
In patients treated with GLP-1 receptor agonists, including liraglutide, there have been reports of acute renal injury/failure and worsening of chronic renal failure, sometimes requiring haemodialysis [see Section 4.8 Adverse Effects (Undesirable Effects)]. Some of these events were reported in patients without known underlying renal disease. A majority of the reported events occurred in patients who had experienced nausea, vomiting, and diarrhoea leading to volume depletion. Some of the reported events occurred in patients receiving one or more medications known to affect renal function and volume status. Altered renal function has been reversed in many of the reported cases with supportive treatment and discontinuation of potentially causative agents, including liraglutide. Use caution when initiating or escalating doses of ARX-Liraglutide in patients with renal impairment.
Use in hepatic impairment.
The safety and efficacy of liraglutide in patients with hepatic insufficiency has not been studied. ARX-Liraglutide is not recommended in patients with hepatic insufficiency.
Use in renal impairment.
The safety and efficacy of liraglutide in patients with severe renal impairment have not been established. ARX-Liraglutide is not recommended for use in patients with severe renal impairment, including end-stage renal disease.
Use in the elderly.
In liraglutide clinical trials, 232 (6.9%) of the liraglutide-treated patients were 65 years of age and over, and 17 (0.5%) of the liraglutide-treated patients were 75 years of age and over. Patients ≥ 65 years may experience more gastrointestinal adverse reactions with liraglutide than younger patients [see Section 4.4 Special Warnings and Precautions for Use, Dehydration, renal impairment and acute renal failure]. No overall differences in safety or effectiveness were observed between these patients and younger patients. Use caution in patients aged 65-74 years. ARX-Liraglutide is not recommended in patients 75 years or older.
Paediatric use.
The efficacy and safety of liraglutide has not been studied in paediatric patients. ARX-Liraglutide is not indicated for use in paediatric patients.
Pancreatitis.
Acute pancreatitis has been observed with the use of GLP-1 receptor agonists. After initiation of ARX-Liraglutide, observe patients carefully for signs and symptoms of pancreatitis. Patients should be informed of the characteristic symptoms of acute pancreatitis. If pancreatitis is suspected, ARX-Liraglutide should be discontinued and appropriate management initiated. If acute pancreatitis is confirmed, ARX-Liraglutide should not be restarted.
In liraglutide clinical trials, acute pancreatitis was confirmed by adjudication more commonly in liraglutide-treated patients versus placebo-treated patients [see Section 4.8 Adverse Effects (Undesirable Effects)].
It is unknown whether patients with a history of pancreatitis are at increased risk for pancreatitis while using liraglutide, since these patients were excluded from clinical trials.
ARX-Liraglutide is not recommended for use in patients with a history of pancreatitis.
In the absence of other signs and symptoms of acute pancreatitis, elevations in pancreatic enzymes alone are not predictive of acute pancreatitis.
Cholelithiasis and cholecystitis.
In the liraglutide clinical trials, cholelithiasis or cholecystitis was reported more commonly in liraglutide-treated patients than in placebo-treated patients [see Section 4.8 Adverse Effects (Undesirable Effects)]. The majority of liraglutide-treated patients with cholelithiasis or cholecystitis required cholecystectomy. Substantial or rapid weight loss can increase the risk of acute gallbladder disease; however, the incidence was greater in liraglutide-treated patients versus placebo-treated patients even after accounting for weight loss. Patients should be informed of the characteristic symptoms of cholelithiasis and cholecystitis.
Inflammatory bowel disease and diabetic gastroparesis.
There is limited experience in patients with inflammatory bowel disease and diabetic gastroparesis. ARX-Liraglutide is not recommended in these patients because it is associated with gastrointestinal adverse reactions, including nausea, vomiting and diarrhoea.
Hypoglycaemia with concomitant use of anti-diabetic therapy.
The risk of serious hypoglycaemia is increased when liraglutide is used in combination with insulin secretagogues (e.g. sulfonylureas) in patients with type 2 diabetes [see Section 4.8 Adverse Effects (Undesirable Effects), Table 2]. The risk of hypoglycaemia can be lowered by a reduction in the dose of sulfonylurea.
The addition of liraglutide in patients treated with insulin has not been evaluated. The SCALE-Diabetes trial excluded patients on insulin [see Section 5.1 Pharmacodynamic Properties, Clinical trials]. ARX-Liraglutide and insulin should not be used together.
Liraglutide can lower blood glucose. Monitor blood glucose parameters before starting and during ARX-Liraglutide treatment in patients with type 2 diabetes. If needed, adjust co-administered anti-diabetic medicines based on glucose monitoring and risk of hypoglycaemia.
Malignancies.
In the clinical development program for weight loss, there was no imbalance for all neoplasms, combined. However, when subgroup analyses were done by individual types of cancer, imbalances were identified, including invasive breast cancer in women and colorectal neoplasms (mainly adenomas) [see Section 4.8 Adverse Effects (Undesirable Effects)].
Thyroid C-cell tumours.
Liraglutide caused thyroid C-cell adenomas and carcinomas in two-year studies in mice and rats. Such medullary thyroid cancers are extremely rare cancers in humans. C-cell neoplasia was observed in mice at subcutaneous doses ≥ 1 mg/kg/day (relative exposure based on plasma AUC, ≥ 8) and in rats at all doses tested (≥ 0.075 mg/kg/day subcutaneously; relative exposure, ≥ 0.5). No tumours or other C-cell proliferative changes were seen in monkeys treated with liraglutide for 20 months (≤ 5 mg/kg/day subcutaneously; relative exposure, ≤ 70). The findings in mice and rats are mediated by a specific GLP-1 receptor-mediated mechanism to which rodents are particularly sensitive. The relevance for humans is likely to be low but cannot presently be completely excluded.
Thyroid disease.
In clinical trials in type 2 diabetes, thyroid adverse events such as goitre have been reported, in particular in patients with pre-existing thyroid disease. Cases of increased blood calcitonin were also observed in the weight management clinical trials. ARX-Liraglutide should be used with caution in patients with thyroid disease.
Hypersensitivity reactions.
There have been reports of serious hypersensitivity reactions (e.g. anaphylactic reactions and angioedema) in patients treated with liraglutide. If a hypersensitivity reaction occurs, then the patient should discontinue ARX-Liraglutide and other suspect medicines and promptly seek medical advice.
Angioedema has been reported with other GLP-1 receptor agonists. Do not use ARX-Liraglutide in patients with a history of angioedema with another GLP-1 receptor agonist because such patients may be predisposed to angioedema with liraglutide.
Suicidal behaviour and ideation.
Patients treated with ARX-Liraglutide should be monitored for the emergence of depression, suicidal thoughts or behaviour, or any unusual changes in mood or behaviour. Discontinue ARX-Liraglutide in patients who experience suicidal thoughts or behaviours or who develop other symptoms of depression [see Section 4.8 Adverse Effects (Undesirable Effects)].
Patients with a history of major depressive disorder or other major psychiatric disorder were excluded from the liraglutide clinical trials. Because of the lack of data on efficacy and safety in patients with a history of major depressive disorder or other major psychiatric disorder, ARX-Liraglutide is not recommended in these patients.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
No clinically significant drug interactions have been demonstrated with liraglutide.
In vitro assessment of drug-drug interaction.
Liraglutide has very low potential for pharmacokinetic drug-drug interactions related to cytochrome P450 (CYP) and plasma protein binding.
In vivo assessment of drug-drug interaction.
The drug-drug interaction studies were performed at steady state with liraglutide 1.8 mg/day. The effect on rate of gastric emptying (paracetamol AUC0-5h) was equivalent between liraglutide 1.8 mg and 3.0 mg [see Section 5.1 Pharmacodynamic Properties]. Administration of the interacting drugs was timed so that Cmax of liraglutide (8-12 h) would coincide with the absorption peak of the co-administered drugs.
Oral medications.
The delay of gastric emptying caused by liraglutide may impact absorption of concomitantly administered oral medicinal products. Interaction studies did not show any clinically relevant delay of absorption of the compounds that were studied, however clinically relevant interactions with other compounds where the effect is dependent on Cmax and tmax, drugs with narrow therapeutic index, or medications associated with local gastrointestinal irritation (e.g. bisphosphonates, potassium chloride) cannot be excluded.
Few patients treated with liraglutide reported at least one episode of severe diarrhoea. Diarrhoea may affect the absorption of concomitant oral medicinal products.
Paracetamol.
Liraglutide did not change the overall exposure (AUC) of paracetamol following a single dose of paracetamol 1000 mg, administered 8 hours after the dose of liraglutide at steady state. Paracetamol Cmax was decreased by 31% and median tmax was delayed up to 15 min. No dose adjustment for concomitant use of paracetamol is required.
Atorvastatin.
Liraglutide did not change the overall exposure (AUC) of atorvastatin following a single dose of atorvastatin 40 mg, administered 5 hours after the dose of liraglutide at steady state. Atorvastatin Cmax was decreased by 38% and median tmax was delayed from 1 hour to 3 hours with liraglutide. Therefore, no dose adjustment of atorvastatin is required when given with liraglutide.
Griseofulvin.
Liraglutide did not change the overall exposure (AUC) of griseofulvin following co-administration of a single dose of griseofulvin 500 mg with liraglutide at steady state. Griseofulvin Cmax increased by 37% while median tmax did not change. Dose adjustments of griseofulvin and other compounds with low solubility and high permeability are not required.
Digoxin.
A single dose of digoxin 1 mg was administered 7 hours after the dose of liraglutide at steady state. The concomitant administration with liraglutide resulted in a reduction of digoxin AUC by 16%; Cmax decreased by 31%. Digoxin median time to maximum concentration (tmax) was delayed from 1 hour to 1.5 hours. No dose adjustment of digoxin is required based on these results.
Lisinopril.
A single dose of lisinopril 20 mg was administered 5 minutes after the dose of liraglutide at steady state. The co-administration with liraglutide resulted in a reduction of lisinopril AUC by 15%; Cmax decreased by 27%. Lisinopril median tmax was delayed from 6 hours to 8 hours with liraglutide. No dose adjustment of lisinopril is required based on these results.
Oral contraceptives.
A single dose of an oral contraceptive combination product containing 0.03 mg ethinylestradiol and 0.15 mg levonorgestrel was administered under fed conditions and 7 hours after the dose of liraglutide at steady state. Liraglutide lowered ethinylestradiol and levonorgestrel Cmax by 12 and 13%, respectively. Tmax was delayed by 1.5 hours with liraglutide for both compounds. There was no clinically relevant effect on the overall exposure (AUC) of ethinylestradiol. Liraglutide increased the levonorgestrel AUC0-∞ by 18%. The contraceptive effect is therefore anticipated to be unaffected when co-administered with liraglutide.
Warfarin and other coumarin derivatives.
No interaction study has been performed. A clinically relevant interaction with active substances with poor solubility or narrow therapeutic index such as warfarin cannot be excluded. Upon initiation of ARX-Liraglutide treatment in patients on warfarin or other coumarin derivatives, more frequent monitoring of INR (International Normalised Ratio) is recommended.
Insulin.
No pharmacokinetic interaction was observed between liraglutide and insulin detemir when separate subcutaneous injections of insulin detemir 0.5 units/kg (single-dose) and liraglutide 1.8 mg (steady state) were administered in patients with type 2 diabetes.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No adverse effects on fertility were observed in male and female rats given subcutaneous doses of liraglutide at ≤ 1 mg/kg/day, yielding exposure to liraglutide (plasma AUC) 12-14 times higher than that of patients at the maximum recommended human dose.
(Category B3)
Increased embryofetal death and minor fetal skeletal abnormalities (kinked ribs) were observed in rats given liraglutide at 1 mg/kg/day by subcutaneous injection (yielding 12-times the plasma AUC in humans at the maximum recommended clinical dose). In rabbits treated at doses ≥ 0.01 mg/kg/day (relative exposure, ≥ 0.2), there was retardation of fetal growth and an increased incidence of several minor skeletal and visceral abnormalities. Postnatal body weight gain was reduced in the offspring of rats treated with liraglutide during gestation and lactation. These findings may have occurred secondary to reduced maternal food consumption. Placental transfer of liraglutide and/or its metabolites was demonstrated in the animal species.
There are limited data from the use of liraglutide in pregnant women. ARX-Liraglutide should not be used during pregnancy. If a patient wishes to become pregnant, or pregnancy occurs, treatment with ARX-Liraglutide should be discontinued.
It is not known whether liraglutide is excreted in human milk. Studies in lactating rats have shown that the transfer of liraglutide and metabolites of close structural relationship into milk is low. Non-clinical studies have shown a treatment related reduction of neonatal growth in suckling rat pups. Due to lack of experience, ARX-Liraglutide must not be used during breast-feeding.4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed. Patients with type 2 diabetes should be advised to take precautions to avoid hypoglycaemia while driving and using machines, in particular when ARX-Liraglutide is used in combination with a sulfonylurea.
4.8 Adverse Effects (Undesirable Effects)
Summary of safety profile.
Clinical trials were conducted with the reference medicine Saxenda. Overall, gastrointestinal reactions were the most frequently reported adverse reactions during treatment with liraglutide: nausea, vomiting, diarrhoea and constipation reported by > 10% of subjects, see Description of selected adverse reactions below.
Tabulated summary of adverse reactions.
The data below reflect exposure to liraglutide in four randomised, double-blind, placebo controlled, multicentre Phase 3 clinical trials, one of 32-weeks duration and three of 56-weeks duration, and one Phase 2 supportive trial in 469 adult patients.
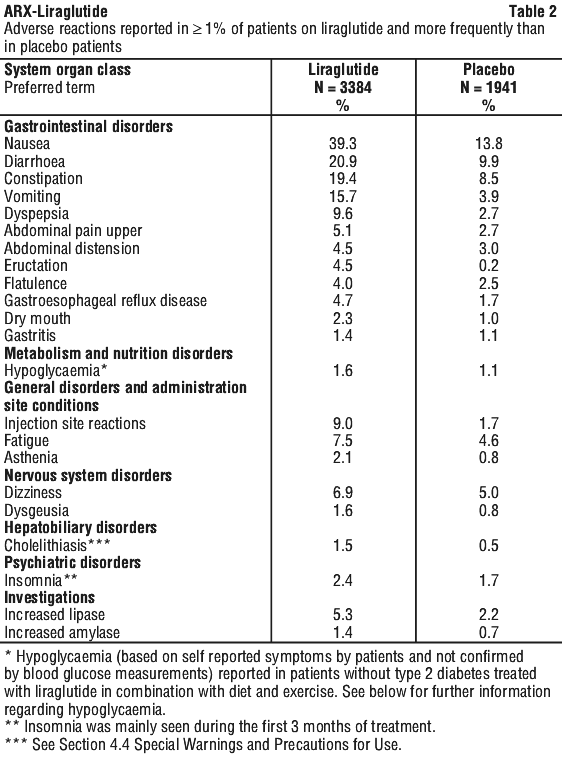
In clinical trials, 9.8% of patients treated with liraglutide prematurely discontinued treatment due to adverse reactions, compared with 4.3% of placebo-treated patients. Adverse reactions reported in greater than or equal to 1% of liraglutide treated patients and more frequently than in placebo patients are shown in Table 2.
Nervous system disorders.
Very common (≥ 1/10) - headache.
Skin and subcutaneous tissue disorders.
Common (≥ 1/100 to < 1/10) - rash.
Less common adverse events in clinical trials (< 1%).
Adverse reactions are listed by system organ class using the frequency categories uncommon (≥ 1/1,000 to < 1/100) and rare (≥ 1/10,000 to < 1/1,000).
Gastrointestinal disorders.
Uncommon - pancreatitis, delayed gastric emptying.*
Metabolism and nutrition disorders.
Uncommon - dehydration.
General disorders and administration site conditions.
Uncommon - malaise.
Hepatobiliary disorders.
Uncommon - cholecystitis.
Immune system disorders.
Rare - anaphylactic reaction.
Cardiac disorders.
Uncommon - tachycardia.
Skin and subcutaneous tissue disorders.
Uncommon - urticaria.
Renal and urinary disorders.
Rare - acute renal failure, renal impairment.
*Delayed gastric emptying based on clinical trial and post marketing data.
Description of selected adverse reactions.
Cardiovascular events. Heart rate increase.
Mean increases in resting heart rate of 2 to 3 beats per minute (bpm) were observed with routine clinical monitoring in liraglutide-treated patients compared to placebo in clinical trials. More patients treated with liraglutide, compared with placebo, had changes from baseline at two consecutive visits of more than 10 bpm (34% versus 19%); and 20 bmp (5% versus 2%). At least one resting heart rate exceeding 100 bpm was recorded for 6% of liraglutide-treated patients compared with 4% of placebo-treated patients, with this occurring at two consecutive study visits for 0.9% and 0.3%, respectively. Tachycardia was reported as an adverse reaction in 0.6% of liraglutide-treated patients and in 0.1% of placebo-treated patients [see Section 4.4 Special Warnings and Precautions for Use].
In a clinical pharmacology trial that monitored heart rate continuously for 24 hours, liraglutide treatment was associated with a heart rate that was 4 to 9 bpm higher than that observed with placebo.
Major adverse cardiovascular events.
Major adverse cardiovascular events (MACE) were adjudicated by an external independent group of experts and defined as non-fatal myocardial infarction, non-fatal stroke, and cardiovascular death. From the 5 double-blind controlled phase 2 and phase 3 clinical trials there were 6 (0.1%) confirmed MACE for liraglutide-treated patients and 10 (0.5%) for placebo-treated patients. The hazard ratio and 95% CI was 0.33 [0.12; 0.90] for liraglutide versus placebo. Favourable trends for cardiovascular disease in pre-market trials (that were not powered for this endpoint and who enrolled low-risk patients) did not necessarily provide reassurance of cardiovascular safety.
The Liraglutide Effect and Action in Diabetes Evaluation of Cardiovascular Outcomes Results (LEADER) study provides some supportive evidence for the use of liraglutide in cardiovascular disease. Extrapolation of the results of LEADER to ARX-Liraglutide is to be performed with caution given that the maintenance dose of ARX-Liraglutide is 3.0 mg per day (versus 1.8 mg per day in the LEADER trial), and that the patient population treated in LEADER all had type 2 diabetes mellitus, were of a high CV risk, and were significantly older than ARX-Liraglutide patients on average. The duration of exposure to liraglutide was between 3.5 and 5 years. The mean age was 64 years and the mean BMI was 32.5 kg/m2. Mean baseline HbA1c was 8.7%. Liraglutide significantly reduced the rate of major adverse cardiovascular events (primary endpoint events, MACE) vs. placebo (3.41 vs. 3.90 per 100 patient years of observation in the liraglutide and placebo groups, respectively) with a risk reduction of 13%.
Cardiac conduction disorders and PR interval prolongation.
A prolongation of the mean PR interval of up to 10 ms was reported with liraglutide treatment in a clinical trial in healthy volunteers, using lower doses than recommended for weight management.
In liraglutide clinical trials, the incidence of cardiac conduction disorders (e.g. first degree atrioventricular [AV] block) was higher with liraglutide than placebo; 11 (0.3%) of 3384 liraglutide-treated patients compared with none of the 1941 placebo-treated patients had a cardiac conduction disorder [see Section 4.4 Special Warnings and Precautions for Use].
Hypoglycaemia in patients without type 2 diabetes. In clinical trials in overweight or obese patients without type 2 diabetes treated with liraglutide in combination with diet and exercise no severe hypoglycaemic events (requiring third party assistance) were reported. Symptoms of hypoglycaemic events were reported by 1.6% of patients treated with liraglutide and 1.1% of patients treated with placebo; however, these events were not confirmed by blood glucose measurements. The majority of events were mild.
Hypoglycaemia in patients with type 2 diabetes. In a clinical trial in overweight or obese patients with type 2 diabetes treated with liraglutide in combination with diet and exercise, hypoglycaemic events were accompanied by blood glucose measurements and classified accordingly. Severe hypoglycaemia (requiring third party assistance) was reported by 0.7% of patients treated with liraglutide and only in patients concomitantly treated with sulfonylurea. Also, in these patients documented symptomatic hypoglycaemia (defined as plasma glucose ≤ 3.9 mmol/L accompanied by symptoms) was reported by 43.6% of patients treated with liraglutide and in 27.3% of patients treated with placebo. Among patients not concomitantly treated with sulfonylurea, 15.7% of patients treated with liraglutide and 7.6% of patients treated with placebo reported documented symptomatic hypoglycaemic events.
Gastrointestinal adverse reactions. In liraglutide clinical trials, 68% of liraglutide-treated patients and 39% of placebo-treated patients reported gastrointestinal disorders; the most frequently reported was nausea (39% versus 14%). The percentage of patients reporting nausea declined as treatment continued. Other common adverse reactions that occurred at higher incidence among liraglutide-treated patients included diarrhoea, constipation, vomiting, dyspepsia, abdominal pain, dry mouth, gastritis, gastroesophageal reflux, flatulence, eructation, and abdominal distension. Episodes of gastrointestinal events leading to discontinuation of therapy were: liraglutide 6.2% versus placebo: 0.8% [see Section 4.4 Special Warnings and Precautions for Use].
Most episodes of gastrointestinal events were mild to moderate, transient and the majority did not lead to discontinuation of therapy. The reactions usually occurred during the first weeks of treatment and diminished within a few days or weeks on continued treatment.
Patients older than 65 years of age may experience more gastrointestinal effects when treated with liraglutide [see Section 4.4 Special Warnings and Precautions for Use].
Patients with mild or moderate renal impairment (creatinine clearance ≥ 30 mL/min) may experience more gastrointestinal effects when treated with liraglutide.
Acute renal failure. In patients treated with GLP-1 receptor agonists, including liraglutide, there have been reports of acute renal injury/failure and worsening chronic renal failure, sometimes requiring haemodialysis. Some of these events were reported in patients without known underlying renal disease. A majority of reported events occurred in patients who had experienced nausea, vomiting, or diarrhoea leading to volume depletion [see Section 4.4 Special Warnings and Precautions for Use]. Some of the reported events occurred in patients receiving one or more medications known to affect renal function and volume status. Altered renal function has been reversed in many of the reported cases with supportive treatment and discontinuation of potentially causative agents, including liraglutide.
Malignancy. Breast cancer.
In liraglutide clinical trials, breast cancer confirmed by adjudication was reported in 17 (0.7%) of 2379 liraglutide-treated women compared with 3 (0.2%) of 1300 placebo-treated women, including invasive cancer (13 liraglutide-treated versus 2 placebo-treated women) and ductal carcinoma in situ (4 versus 1). The majority of cancers were estrogen- and progesterone-receptor positive. There were too few cases to determine whether these cases were related to liraglutide. In addition, there are insufficient data to determine whether liraglutide has an effect on pre-existing breast neoplasia.
Colorectal neoplasms.
In liraglutide clinical trials, benign colorectal neoplasms (mostly colon adenomas) confirmed by adjudication were reported in 20 (0.6%) of 3291 liraglutide-treated patients compared with 7 (0.4%) of 1843 placebo-treated patients. Six positively adjudicated cases of malignant colorectal carcinoma were reported in 5 liraglutide-treated patients and 1 in a placebo-treated patient.
Papillary thyroid cancer.
In liraglutide clinical trials, papillary thyroid carcinoma, confirmed by adjudication, was reported in 8 (0.2%) of 3291 liraglutide-treated patients compared with no cases among 1843 placebo treated patients. Four of these papillary thyroid carcinomas were less than 1 cm in greatest diameter and 4 were diagnosed in surgical pathology specimens after thyroidectomy.
Immunogenicity. Consistent with the potentially immunogenic properties of protein and peptide pharmaceuticals, patients may develop anti-liraglutide antibodies following treatment with liraglutide. In clinical trials, 2.5% of liraglutide treated patients developed anti-liraglutide antibodies. Antibody formation has not been associated with reduced efficacy of liraglutide.
Injection site reactions. Injection site reactions have been reported in patients treated with liraglutide solution for injection. These reactions have usually been mild and transitory and the majority resolved during continued treatment.
Pancreatitis. Few cases of acute pancreatitis have been reported during long-term clinical trials with liraglutide [see Section 4.4 Special Warnings and Precautions for Use]. In liraglutide clinical trials, acute pancreatitis was confirmed by adjudication in 9 (0.3%) of 3291 liraglutide-treated patients versus 2 (0.1%) of 1843 placebo-treated patients. In addition, there were 2 cases of acute pancreatitis in liraglutide-treated patients who prematurely withdrew from the clinical trials, occurring 74 and 124 days after the last dose. There were two additional cases in liraglutide-treated patients, one during an off-treatment follow-up period within 2 weeks of discontinuing liraglutide, and one that occurred in a patient who completed treatment and was off treatment for 106 days.
Allergic reactions. Few cases of anaphylactic reactions with symptoms such as hypotension, palpitations, dyspnoea or oedema have been reported with marketed use of liraglutide. Anaphylactic reactions may potentially be life threatening.
Suicidal behaviour and ideation. In the liraglutide clinical trials, 9 (0.3%) of 3384 liraglutide-treated patients and 2 (0.1%) of the 1941 placebo-treated patients reported suicidal ideation; one of the liraglutide-treated patients attempted suicide [see Section 4.4 Special Warnings and Precautions for Use].
Hypotension. Adverse reactions related to hypotension (i.e. reports of hypotension, orthostatic hypotension, circulatory collapse, and decreased blood pressure) were reported more frequently with liraglutide (1.1%) compared with placebo (0.5%) in liraglutide clinical trials. Systolic blood pressure decreases to less than 80 mmHg were observed in 4 (0.1%) liraglutide-treated patients compared with no placebo-treated patients. One of the liraglutide-treated patients had hypotension associated with gastrointestinal adverse reactions and renal failure [see Section 4.4 Special Warnings and Precautions for Use].
Laboratory abnormalities. Liver enzymes.
Increases in alanine aminotransferase (ALT) greater than or equal to 10 times the upper limit of normal were observed in 5 (0.15%) liraglutide-treated patients (two of whom had ALT greater than 20 and 40 times the upper limit of normal) compared with 1 (0.05%) placebo-treated patient during the liraglutide clinical trials. Because clinical evaluation to exclude alternative causes of ALT and aspartate aminotransferase (AST) increases was not done in most cases, the relationship to liraglutide is uncertain. Some increases in ALT and AST were associated with other confounding factors (such as gallstones).
Serum calcitonin.
Calcitonin, a biological marker of MTC, was measured throughout the clinical development program [see Section 4.4 Special Warnings and Precautions for Use]. More patients treated with liraglutide in the clinical trials were observed to have high calcitonin values during treatment, compared with placebo. The proportion of patients with calcitonin greater than or equal to 2 times the upper limit of normal at the end of the trial was 1.2% in liraglutide-treated patients and 0.6% in placebo-treated patients. Calcitonin values greater than 20 ng/L at the end of the trial occurred in 0.5% of liraglutide-treated patients and 0.2% of placebo-treated patients; among patients with pre-treatment serum calcitonin less than 20 nanogram/L, none had calcitonin elevations to greater than 50 nanogram/L at the end of the trial.
Post-marketing adverse effects.
The following adverse reactions have been reported during post approval use of liraglutide. Because these reactions are reported voluntarily from a population of uncertain size it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal disorders.
Acute pancreatitis, haemorrhagic and necrotising pancreatitis.
Intestinal obstruction including ileus.
General disorders and administration site conditions.
Allergic reactions: urticaria, rash and pruritus.
Malaise.
Immune system disorders.
Angioedema and anaphylactic reactions.
Metabolism and nutrition disorders.
Dehydration resulting from nausea, vomiting and diarrhoea.
Renal and urinary disorders.
Increased serum creatinine, acute renal failure or worsening of chronic renal failure, sometimes requiring haemodialysis.
Cardiac disorders.
Increased heart rate.
Skin and subcutaneous tissue disorders.
Cutaneous amyloidosis.
Patients must be instructed to perform continuous rotation of the injection site to reduce the risk of developing cutaneous amyloidosis. There may be a potential risk of change in liraglutide absorption or effect following ARX-Liraglutide injections at sites with cutaneous amyloidosis.
Infections and infestations.
Urinary tract infection.
Hepatobiliary.
Elevation of liver enzymes.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important.It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at http://www.tga.gov.au/reporting-problems.4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
From clinical trials and post-market use of liraglutide, deliberate or accidental administration of doses up to 24 times the recommended maintenance dose (72 mg) have been reported, including one case of a 6-fold overdose (18 mg daily) given for 7 months. These included instances where patients needed hospitalisation either due to severe events of vomiting, nausea and diarrhoea, or as a precaution. In some reports glucose infusion was administered. Severe hypoglycaemia has also been observed. All patients were reported to have recovered from the events without complications.
In the event of overdosage, appropriate supportive treatment should be initiated according to the patient's clinical signs and symptoms. The patient should be observed for clinical signs of dehydration and blood glucose should be monitored.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Liraglutide is an acylated human GLP-1 analogue with 97% amino acid sequence homology to endogenous human GLP-1. Like endogenous GLP-1, liraglutide binds to and activates the GLP-1R. Liraglutide is relatively stable against metabolic degradation and has a plasma half-life of 13 hours after subcutaneous administration.
Unlike native GLP-1, liraglutide has a pharmacokinetic and pharmacodynamic profile in humans suitable for once daily administration. Following subcutaneous administration, the protracted action profile is based on three mechanisms: self-association (which results in slow absorption), binding to albumin and enzymatic stability towards the dipeptidyl peptidase (DPP-IV) and neutral endopeptidase (NEP) enzymes, resulting in a long plasma half-life.
GLP-1 is a physiological regulator of appetite and food intake and GLP-1R is present in several areas of the brain involved in appetite regulation as well as the intestine. In animal studies, peripheral administration of liraglutide led to uptake in specific brain regions including the hypothalamus, where liraglutide, via specific activation of the GLP-1R, increased key satiety and decreased key hunger signals. Transient inhibition of gastric emptying was also observed.
GLP-1 receptors are also expressed in specific locations in the heart, vasculature, immune system, and kidneys. In mouse models of atherosclerosis liraglutide prevented aortic plaque progression and reduced the expression of genes related to inflammation in aortic tissue. In addition, liraglutide had a beneficial effect on plasma lipids, decreasing plasma triglyceride, total cholesterol, LDL and VLDL, and increasing HDL. Liraglutide did not reduce the plaque size of already established plaques.
Liraglutide lowers body weight through decreased food intake and loss of predominantly fat mass. Liraglutide does not increase 24-hour energy expenditure. Liraglutide affects the four main components of appetite. Liraglutide regulates appetite by increasing feelings of fullness and satiety, while lowering feelings of hunger and prospective food consumption.
Liraglutide also has effects on glucose homeostasis, resulting in lowering of fasting and post-prandial glucose. Liraglutide stimulates insulin secretion, lowers inappropriately high glucagon secretion in a glucose-dependent manner and improves beta-cell function. The mechanism of blood glucose lowering also may involve a minor delay in gastric emptying. [See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions].
Clinical trials.
Clinical trials were conducted with the reference medicine Saxenda. The safety and efficacy of liraglutide for weight management in conjunction with reduced caloric intake and increased physical activity were studied in four phase 3 randomised, double-blind, placebo-controlled trials which included a total of 5,358 patients.
SCALE-Obesity and pre-diabetes (NN8022-1839).
In this trial, a total of 3,731 patients with obesity (BMI ≥ 30 kg/m2), or overweight (BMI ≥ 27 kg/m2) with dyslipidaemia and/or hypertension, were stratified according to pre-diabetes status at screening and BMI at baseline (≥ 30 kg/m2 or < 30 kg/m2). All 3,731 patients were randomised to 56 weeks of treatment and the 2,254 patients with pre-diabetes at screening were randomised to 160 weeks of treatment followed by a 12-week off medication/placebo observational follow up period. Lifestyle intervention in the form of an energy-restricted diet and exercise counselling was background therapy for all patients. The 56 week part of this trial assessed body weight loss in all the 3,731 randomised patients (2,590 completers). The 160 week part of this trial assessed time to onset of type 2 diabetes in the 2,254 randomised patients with pre-diabetes (1,128 completers).
SCALE-Diabetes (NN8022-1922).
A 56-week trial assessing body weight loss in 846 randomised (628 completers) obese and overweight patients with insufficiently controlled type 2 diabetes (HbA1c range 7-10%). The background treatment at trial start was either diet and exercise alone, metformin, a sulfonylurea, a glitazone as single agents or any combination hereof.
SCALE-Sleep apnoea (NN8022-3970).
A 32 week trial assessing sleep apnoea severity and body weight loss in 359 randomised (276 completers) obese patients with moderate or severe obstructive sleep apnoea (OSA).
SCALE-Maintenance (NN8022-1923).
A 56-week trial assessing body weight maintenance and weight loss in 422 randomised (305 completers) obese or overweight patients, with hypertension or dyslipidaemia, after a preceding ≥ 5% weight loss induced by a low caloric diet.
In all studies, patients received one-on-one instruction for a reduced calorie diet (approximately 500 kcal/day (2090 kJ/day) deficit) and exercise counselling (recommended increase in physical activity of minimum 150 mins/week) that began with the first dose of study medication or placebo and continued throughout the trial.
Body weight.
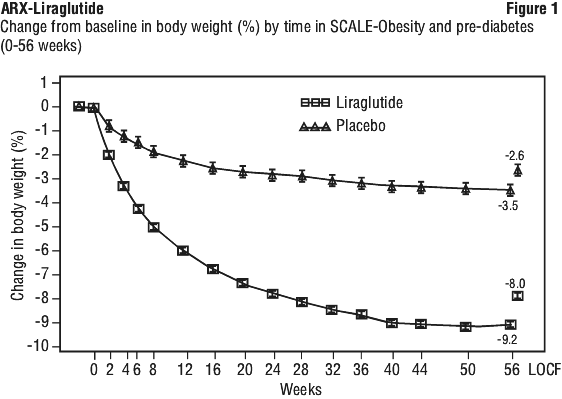
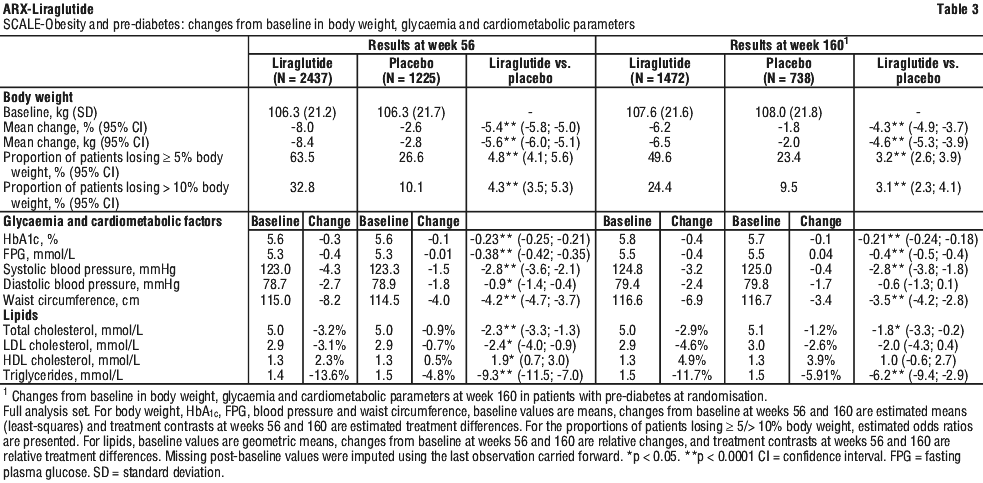
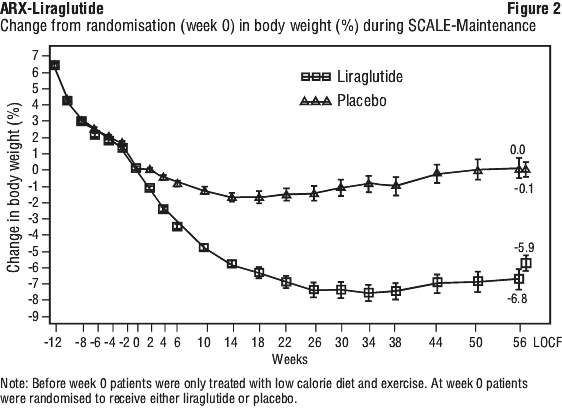
Superior weight loss was achieved with liraglutide compared to placebo in obese/overweight patients in all groups studied including those with and without pre-diabetes, type 2 diabetes and moderate or severe obstructive sleep apnoea. Across the trial populations, greater proportions of the patients achieved ≥ 5% and > 10% weight loss with liraglutide than with placebo (Tables 3-5). A significant body weight reduction was also observed in SCALE-Maintenance, where patients had achieved a mean weight loss of 6.0% on a low-calorie diet during a 12 week run-in period prior to treatment with liraglutide. In SCALE-Maintenance, more patients maintained the weight loss achieved prior to treatment initiation with liraglutide than with placebo (81.4% and 48.9%, respectively). Specific data on weight loss, responders and time course for all 4 trials are presented in Tables 3-6 and Figures 2-3.
In SCALE-Obesity and Pre-diabetes, patients treated with liraglutide achieved a greater weight loss, as compared to placebo. The weight loss occurred mainly in the first year. The mean percent change in body weight and the proportions of patients achieving greater than or equal to 5% and greater than 10% weight loss from baseline to week 160 were also significant compared to placebo in this trial (Table 3).
Weight loss response after 12 weeks with liraglutide 3.0 mg treatment.
Early responders were defined as patients who achieved a weight loss of ≥ 5% after 12 weeks on maintenance dose of liraglutide (4 weeks of dose escalation and 12 weeks on maintenance dose). In the 56-week part of SCALE-Obesity and Pre-diabetes, 67.5% of the patients achieved ≥ 5% weight loss after 12 weeks. In SCALE-Diabetes, 50.4% of patients achieved ≥ 5% weight loss after 12 weeks. With continued treatment with liraglutide, 86.2% of these early responders achieved a weight loss of ≥ 5% and 51% achieved a weight loss of ≥ 10% after one year of treatment. The mean weight loss in early responders who completed 1 year of treatment was 11.2% of their baseline body weight. For patients who achieved a weight loss of < 5% after 12 weeks on maintenance dose and completed 1 year of treatment, the mean weight loss was 3.8% after 1 year.
Glycaemic control.
Treatment with liraglutide significantly improved glycaemic parameters across sub-populations with normoglycaemia, pre-diabetes and type 2 diabetes.
In the 56-week part of SCALE-Obesity and Pre-diabetes, fewer patients treated with liraglutide had developed type 2 diabetes compared to patients treated with placebo (0.2% vs. 1.1%). More patients with pre-diabetes at baseline had reversed their pre-diabetes compared to patients treated with placebo (69.2% vs. 32.7%). In the 160 week part of SCALE-Obesity and Pre-diabetes, the primary efficacy endpoint was the proportion of patients with onset of type 2 diabetes evaluated as time to onset. At week 160, while on treatment, 3% treated with liraglutide and 11% treated with placebo were diagnosed with type 2 diabetes. More patients in the liraglutide 3.0 mg group (65.9%) than the placebo group (36.3%) had regressed their pre-diabetes to normoglycaemia by week 160 (odds ratio 3.6 [95% CI: 3.0 to 4.4], p < 0.001).
The estimated time to onset of type 2 diabetes for patients treated with liraglutide 3.0 mg was 2.7 times' longer (with a 95% confidence interval of [1.9, 3.9]), and the hazard ratio for risk of developing type 2 diabetes was 0.2 for liraglutide versus placebo.
During the entire 160 week treatment period, HbA1c was lower in the liraglutide group than in the placebo group with a statistically significant estimated treatment difference of -0.21% [-0.24; -0.18]95% C.I, p < 0.0001 at 160 weeks. After being off treatment for 12 weeks (week 172) a steep increase in HbA1c was observed in the liraglutide group, while a more modest increase was seen in the placebo group. The same pattern was observed with fasting plasma glucose (FPG). During the entire trial FPG was lower with liraglutide than with placebo, and immediately after treatment cessation FPG reversed to baseline level in the liraglutide group, while no change was observed in the placebo group.
In SCALE-Diabetes, 69.2% of obese patients with type 2 diabetes treated with liraglutide achieved an HbA1c < 7% (ADA) target compared to 27.2% for placebo and 56.5% of obese patients with type 2 diabetes treated with liraglutide achieved an HbA1c ≤ 6.5% (IDF) target compared to 15.0% for placebo.
Cardiometabolic risk factors.
Treatment with liraglutide significantly improved systolic blood pressure, waist circumference and fasting lipids compared with placebo (Tables 3 and 4).
Apnoea-hypopnoea index (AHI).
Treatment with liraglutide significantly reduced the severity of obstructive sleep apnoea as assessed by change from baseline in the AHI compared with placebo (Table 5).
SCALE-Obesity and pre-diabetes: weight management in obese and overweight patients with or without pre-diabetes.
See Figure 1 and Table 3.
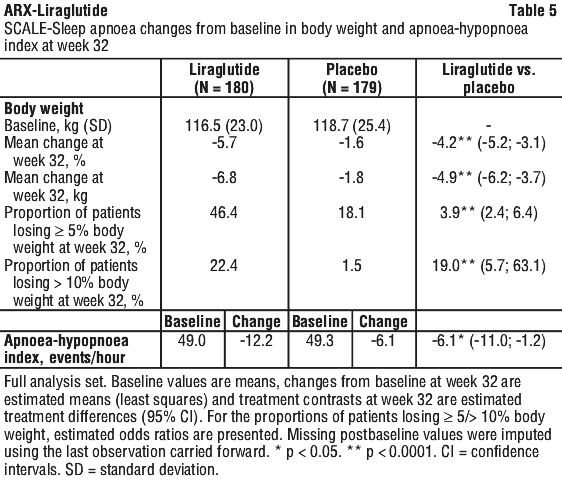
SCALE-Diabetes: weight management in obese and overweight patients with type 2 diabetes.
See Table 4.
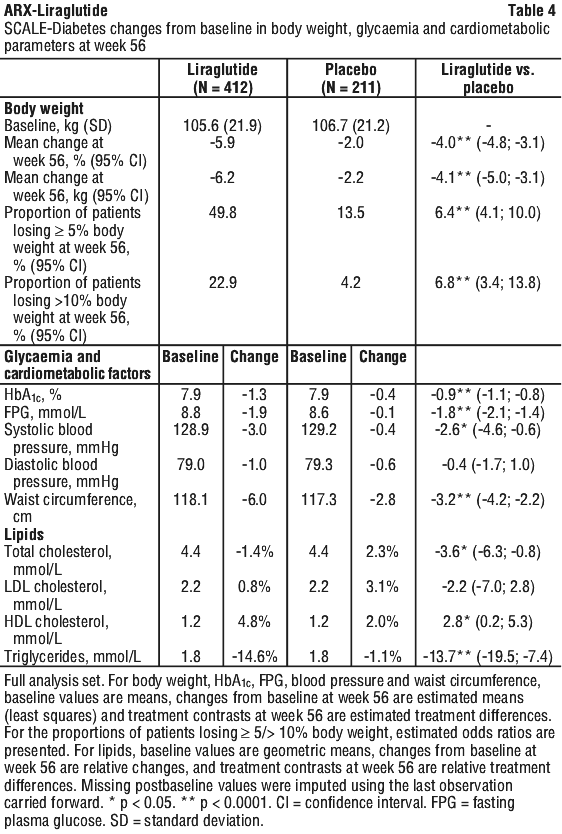
SCALE-Sleep apnoea: weight management in obese patients with moderate or severe obstructive sleep apnoea.
See Table 5.
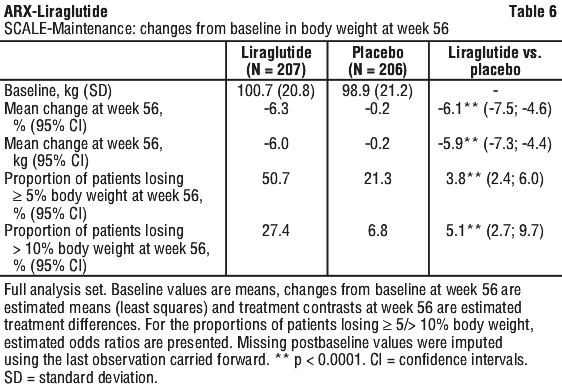
SCALE-Maintenance: weight loss in obese and overweight patients with at least one comorbid condition after initial ≥ 5% weight loss on low caloric diet.
See Table 6 and Figure 2.
Concomitant medication.
Liraglutide was more likely than placebo to reduce the use of antihypertensive and lipid lowering drugs after one year of treatment and in patients with type 2 diabetes, liraglutide was also more likely than placebo to reduce the use of oral antidiabetic drugs after one year of treatment.
Patient reported outcomes.
Liraglutide improved several patient reported outcomes compared to placebo. Significant improvements were seen in the IWQoL-Lite total score (SCALE-Obesity and Pre-diabetes and SCALE-Diabetes) and in all domains of the SF-36 (SCALE-Obesity and Pre-diabetes), indicating favourable effects on physical function and mental health.
Pharmacodynamics.
In long term clinical trials involving overweight and obese patients liraglutide, in conjunction with reduced calorie intake and increased physical activity, significantly lowered body weight.
Distribution of weight loss.
In a sub-study of obese (BMI 30-40 kg/m2), non-diabetic patients, DEXA analysis and CT scans were performed at baseline and at Week 20 for 15 patients on liraglutide and 14 patients on placebo. In the sub-study, weight loss was predominantly from fat mass rather than from lean body mass for both treatment groups. Mean visceral and subcutaneous adipose tissue area was reduced after 20 weeks of treatment compared to baseline. Moreover, with liraglutide, relative reductions in visceral fat were greater than in subcutaneous fat.
Effects on appetite sensations, calorie intake and energy expenditure, gastric emptying, and fasting and postprandial glycaemia.
A five week clinical pharmacology trial was conducted in 49 obese (BMI 30-40 kg/m2) non-diabetic patients to investigate the pharmacodynamic effects of liraglutide.
Appetite sensations, calorie intake, and energy expenditure.
The weight loss effect of liraglutide is considered to be mediated by regulation of appetite and food intake. Appetite sensations were assessed before and up to five hours after a standardised breakfast meal, and ad libitum food intake was assessed during the subsequent lunch meal. Compared to placebo, liraglutide increased post-prandial satiety and fullness ratings, reduced hunger and prospective food consumption ratings and decreased ad libitum food intake. No treatment-related increase in 24-hour energy expenditure was observed as assessed in a respiratory chamber.
Gastric emptying.
Liraglutide caused a minor delay in gastric emptying during the first hour after the meal, thereby reducing the rate as well as the total level of postprandial glucose that appeared in the circulation.
Fasting and postprandial glucose, insulin and glucagon.
Fasting and postprandial glucose, insulin and glucagon concentrations were assessed before and up to five hours after a standardised meal test. Compared to placebo, liraglutide reduced fasting glucose and postprandial glucose (AUC0-60 min) in the first hour after the meal, and also reduced 5-hour glucose AUC and incremental glucose (AUC0-300 min). In addition, liraglutide decreased postprandial glucagon (AUC0-300 min) and postprandial insulin (AUC0-60 min) and incremental insulin (iAUC0-60 min) after the meal compared with placebo.
Fasting and incremental glucose and insulin concentrations were also assessed during a 75-g oral glucose tolerance test (OGTT) before and after 56 weeks of treatment in 3,731 overweight and obese patients with and without pre-diabetes [see Section 5.1 Pharmacodynamic Properties, Clinical trials, SCALE-Obesity and pre-diabetes]. Compared to placebo, liraglutide reduced fasting and incremental glucose concentrations (Figure 1). The effect was more pronounced in patients with pre-diabetes. In addition, liraglutide reduced fasting insulin and increased incremental insulin concentrations compared to placebo.
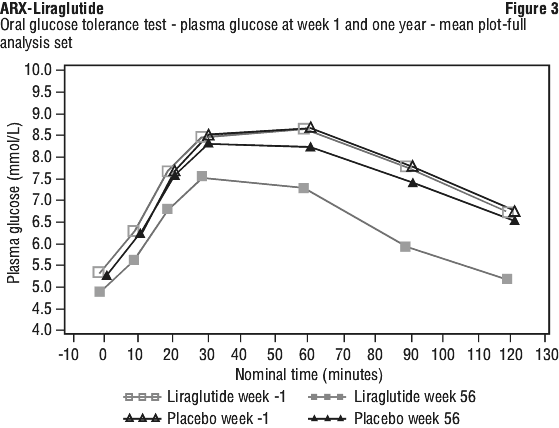
Compared to baseline levels, the week 160 post-challenge plasma glucose AUC was reduced with liraglutide, while on treatment, but remained unchanged with placebo. Additionally, post-challenge insulin AUC remained relatively stable with liraglutide during the 160-week treatment period, while declining in the placebo group. The estimated treatment effects were all statistically significant in favour of liraglutide. See Figure 3.
Effects on fasting and postprandial glucose increment in overweight and obese patients with type 2 diabetes.
Liraglutide reduced fasting glucose and mean postprandial glucose increment (90 minutes after the meal, average over 3 daily meals), compared to placebo.
Beta-cell function.
Clinical studies up to 52 weeks with liraglutide in overweight and obese patients with and without diabetes mellitus have shown a durable secretagogue effect, as well as improvements from baseline in the homeostasis model assessment for beta-cell function (HOMA-B) and the proinsulin to insulin ratio.
Cardiac electrophysiology (QTc).
In a cardiac repolarisation study, liraglutide at steady state concentrations with daily doses up to 1.8 mg did not produce QTc prolongation. The liraglutide exposure for overweight and obese subjects treated with liraglutide is comparable to the exposure evaluated in the liraglutide QTc study.
5.2 Pharmacokinetic Properties
Absorption.
The absorption of liraglutide following subcutaneous administration is slow, reaching maximum concentration approximately 11 hours post dosing. The average steady state concentration of liraglutide (AUCτ/24) reached approximately 31 nmol/L in obese (BMI 30-40 kg/m2) subjects following administration of liraglutide 3.0 mg. Liraglutide exposure increased proportionally with dose in the dose range of 0.6 to 3.0 mg. ARX-Liraglutide can be administered subcutaneously in the abdomen, thigh, or upper arm.
Distribution.
The mean apparent volume of distribution after subcutaneous administration of a liraglutide 3.0 mg is 20-25 L (for a person weighing approximately 100 kg). The mean volume of distribution after intravenous administration of liraglutide is 0.07 L/kg. Liraglutide is extensively bound to plasma protein (> 98%).
Metabolism/biotransformation.
During the 24 hours following administration of a single [3H]-liraglutide dose to healthy subjects, the major component in plasma was intact liraglutide. Two minor plasma metabolites were detected (≤ 9 % and ≤ 5% of total plasma radioactivity exposure).
Excretion.
Liraglutide is endogenously metabolised in a similar manner to large proteins without a specific organ as major route of elimination. Following a [3H]-liraglutide dose, intact liraglutide was not detected in urine or faeces. Only a minor part of the administered radioactivity was excreted as liraglutide-related metabolites in urine or faeces (6% and 5%, respectively). The urine and faeces radioactivity was mainly excreted during the first 6-8 days, and corresponded to three minor metabolites.
The apparent clearance following sub-cutaneous administration of liraglutide 3.0 mg is approximately 0.9-1.4 L/h with an elimination half-life of approximately 13 hours.
Special populations.
Elderly.
No dosage adjustment is required based on age. Age had no clinically relevant effect on the pharmacokinetics of liraglutide 3.0 mg based on a population pharmacokinetic analysis that included overweight and obese subjects (18 to 82 years).
Gender.
Based on results of population pharmacokinetic analyses, females have 24% lower weight adjusted clearance of liraglutide 3.0 mg compared to males. Based on the exposure response data, no dosage adjustment is required based on gender.
Ethnicity.
No dosage adjustment is required based on ethnicity. Ethnicity had no clinically relevant effect on the pharmacokinetics of liraglutide 3.0 mg based on the results of a population pharmacokinetic analysis which included overweight and obese patients.
Body weight.
The exposure of liraglutide decreases with an increase in baseline body weight. The 3.0 mg daily dose of liraglutide provided adequate systemic exposure over the body weight range of 60-234 kg evaluated for exposure response in the clinical trial. Liraglutide exposure was not studied in subjects with body weight > 234 kg.
Hepatic impairment.
The pharmacokinetics of liraglutide were evaluated in subjects with varying degrees of hepatic impairment in a single-dose trial (0.75 mg). Liraglutide exposure was decreased by 23% and 13% in subjects with mild or moderate hepatic impairment respectively, compared to healthy subjects. Exposure was significantly lower (44%) in subjects with severe hepatic impairment (Child Pugh score > 9).
Renal impairment.
Liraglutide exposure was mildly reduced in subjects with renal impairment compared to individuals with normal renal function in a single-dose trial (0.75 mg). Liraglutide exposure was lowered by 33%, 14%, 27% and 26%, in subjects with mild (creatinine clearance, CrCL 50-80 mL/min), moderate (CrCL 30-50 mL/min), and severe (CrCL < 30 mL/min) renal impairment and in end-stage renal disease requiring dialysis, respectively.
Paediatrics.
Liraglutide has not been studied in paediatric subjects.
5.3 Preclinical Safety Data
Genotoxicity.
Liraglutide was not mutagenic in the bacterial Ames assay, and not clastogenic in human lymphocytes in vitro, or in rat lymphocytes and bone marrow in vivo.
Carcinogenicity.
See Section 4.4 Special Warnings and Precautions for Use.6 Pharmaceutical Particulars
6.1 List of Excipients
Each mL of ARX-Liraglutide contains the following inactive ingredients: 1.42 mg dibasic sodium phosphate dihydrate, 14.0 mg propylene glycol, 5.5 mg phenol, hydrochloric acid q.s., sodium hydroxide q.s. and water for injections to 1 mL.
6.2 Incompatibilities
Substances added to ARX-Liraglutide may cause degradation of liraglutide. ARX-Liraglutide must not be mixed with other medicinal products, e.g. infusion fluids.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store in a refrigerator (2°C to 8°C). Keep away from the cooling element. Do not freeze ARX-Liraglutide and do not use ARX-Liraglutide if it has been frozen.
After first use of the ARX-Liraglutide pen, the product can be stored for 30 days at room temperature (below 30°C) or in a refrigerator (2°C to 8°C).
Keep the pen cap on when the ARX-Liraglutide pen is not in use in order to protect from light.
ARX-Liraglutide should be protected from excessive heat and sunlight.
Always remove the injection needle after each injection and store the ARX-Liraglutide pen without an injection needle attached. This may prevent blocked needles, contamination, infection, leakage of solution and inaccurate dosing.
6.5 Nature and Contents of Container
Cartridge (type 1 glass) with a plunger (bromobutyl) and line seal/ lined ferrule (metal cap with bilayer rubber disk) contained in a single-patient use, disposable multi-dose pen-injector.
Each pen contains 3 mL solution at a concentration of 6 mg/mL and is able to deliver doses of 0.6 mg, 1.2 mg, 1.8 mg, 2.4 mg and 3.0 mg.
Pack sizes of 1, 3 or 5 pre-filled pens. Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
In liraglutide, the lysine at position 34 has been replaced with arginine, and a palmitic acid has been attached via a glutamoyl spacer to lysine at position 26.
Chemical structure.
Liraglutide has the molecular formula C172H265N43O51 and a molecular weight of 3751.20 daltons.

CAS number.
204656-20-2.7 Medicine Schedule (Poisons Standard)
Prescription only medicine (Schedule 4).
Summary Table of Changes


 The need for continued treatment should be re-evaluated whenever a new prescription is written and at least annually.
The need for continued treatment should be re-evaluated whenever a new prescription is written and at least annually.








