What is in this leaflet
This leaflet answers some common questions about Atrovent Metered Aerosol.
It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using Atrovent Metered Aerosol against the benefits it is expected to have for you.
If you have any concerns about using this medicine, ask your doctor or pharmacist.
This leaflet was last updated on the date at the end of this leaflet. More recent information may be available. The latest Consumer Medicine Information is available from your pharmacist, doctor or from www.medicines.org.au and may contain important information about the medicine and its use of which you should be aware.
Keep this leaflet with the medicine. You may need to read it again.
What Atrovent Metered Aerosol is used for
Atrovent Metered Aerosol is inhaled into the lungs for the maintenance treatment of difficulty in breathing, wheezing or coughing in:
- asthma
- chronic obstructive pulmonary disease, also called COPD.
Asthma is a disease where the lining of the lungs becomes inflamed (red and swollen), making it difficult to breathe. This may be due to an allergy to house dust mites, smoke or other irritants.
COPD is a serious lung condition that can cause difficulty in breathing and constant coughing. The term COPD is associated with the conditions chronic bronchitis and emphysema. Symptoms of COPD include shortness of breath, cough, chest discomfort and coughing up phlegm.
Atrovent Metered Aerosol contains the active ingredient ipratropium bromide monohydrate. It belongs to a group of medicines called anticholinergic bronchodilators.
Atrovent Metered Aerosol opens up the air passages in people suffering from asthma and chronic obstructive pulmonary disease (COPD).
It begins to act quickly after use but may take up to 2 hours to give maximum benefit.
Ask your doctor if you have any questions about why Atrovent Metered Aerosol has been prescribed for you. Your doctor may have prescribed Atrovent Metered Aerosol for another reason.
There is no evidence that Atrovent Metered Aerosol is addictive.
Atrovent Metered Aerosol is available only with a doctor's prescription.
Before you use Atrovent Metered Aerosol
When you must not use it
Do not use Atrovent Metered Aerosol if you have an allergy to:
- ipratropium bromide
- similar medicines which contain atropine or medicines like atropine
- any of the ingredients listed at the end of this leaflet
- any other anticholinergic medicines.
Some of the symptoms of an allergic reaction may include:
- shortness of breath, wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Do not use Atrovent Metered Aerosol after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start using Atrovent Metered Aerosol, contact your doctor or pharmacist.
Before you start to use it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor or pharmacist if you are pregnant, intend to become pregnant, are breastfeeding or plan to breastfeed. Your doctor or pharmacist will discuss the possible risks and benefits of using Atrovent Metered Aerosol during pregnancy and breastfeeding.
Tell your doctor if you have or have had any of the following medical conditions:
- glaucoma (high pressure in the eye)
- difficulty or pain when passing urine
- constipation
- cystic fibrosis.
If you have not told your doctor or pharmacist about any of the above, tell them before you start using Atrovent Metered Aerosol.
Using other medicines
Tell your doctor or pharmacist if you are using any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Atrovent Metered Aerosol may interfere with each other. These include:
- medicines used to treat heart problems such as adrenaline
- medicines used to treat asthma or COPD such as theophylline, salbutamol and tiotropium.
These medicines may be affected by Atrovent Metered Aerosol, or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while using Atrovent Metered Aerosol.
How to use Atrovent Metered Aerosol
Each puff must be properly inhaled through the mouth for Atrovent Metered Aerosol to work. At the end of this leaflet you will find instructions on how to use Atrovent Metered Aerosol correctly.
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you are not sure how to use the inhaler, ask your doctor or pharmacist for help.
Children should only use their inhaler on medical advice and with the help of an adult.
If you find it difficult to breathe in and use your inhaler at the same time, talk to your doctor or pharmacist. They may be able to recommend another method.
How much to use
The usual doses for adults and children are stated below.
Adults:
2 to 4 puffs, three to four times daily (at regular intervals), up to a maximum of 16 puffs a day.
Children 6 to 12 years:
1 to 2 puffs, three to four times daily (at regular intervals), up to a maximum of 8 puffs a day.
Children under 6 years:
1 puff, three times daily (at regular intervals).
Depending on your condition and specific needs, your doctor may advise you to take a different dose.
When to use it
Use your medicine at about the same time each day. Using it at the same time each day will have the best effect. It will also help you remember when to use it.
If you forget to use it
If it is almost time for your next dose, skip the dose you missed and have your next dose when you are meant to.
Otherwise, use it as soon as you remember, and then go back to using your medicine as you would normally.
Do not use a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to use your medicine, ask your pharmacist for some hints.
How long to use it
Continue using Atrovent Metered Aerosol for as long as your doctor tells you. Atrovent Metered Aerosol helps control your condition, but does not cure it. It is important to keep using it even if you feel well.
If you use too much (overdose)
Immediately telephone your doctor or pharmacist or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Emergency at the nearest hospital, if you think that you or anyone else may have used too much Atrovent Metered Aerosol. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
Symptoms of an overdose may include:
- a fast or irregular heart beat
- dry mouth
- blurred vision.
While you are using Atrovent Metered Aerosol
Things you must do
Stop using Atrovent Metered Aerosol and tell your doctor immediately if you get sudden tightness of the chest, coughing, wheezing or breathlessness immediately after using Atrovent Metered Aerosol. These may be signs of a condition called bronchospasm.
If you have an Asthma or COPD Action Plan that you have agreed with your doctor, follow it closely at all times.
If you find that the usual dose of Atrovent Metered Aerosol is not giving as much relief as before, or you need to use it more often, contact your doctor so that your condition can be checked. This is important to ensure your breathing problem is controlled properly.
Continue using Atrovent Metered Aerosol for as long as your doctor or pharmacist tells you.
Keep all your doctor's appointments so that your progress can be checked.
Make sure you keep Atrovent Metered Aerosol with you at all times.
Contact your doctor immediately if you experience irritation or a feeling of having something in the eye, or any disturbances with your sight (blurred vision, visual halos or coloured images) together with red eyes, during or after using Atrovent Metered Aerosol. This may mean that you have developed a serious eye condition called narrow-angle glaucoma. This can happen if the spray gets in your eyes.
Tell any other doctors, dentists, and pharmacists who are treating you that you are using Atrovent Metered Aerosol.
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are using Atrovent Metered Aerosol.
If you plan to have surgery, tell the surgeon or anaesthetist that you are using Atrovent Metered Aerosol. It may affect other medicines during surgery.
If you become pregnant while using Atrovent Metered Aerosol tell your doctor or pharmacist immediately.
Things you must not do
Do not take any other medicines for your breathing problems without checking with your doctor.
Do not give Atrovent Metered Aerosol to anyone else, even if they have the same condition as you.
Do not use Atrovent Metered Aerosol to treat any other complaints unless your doctor or pharmacist tells you to.
Do not stop using Atrovent Metered Aerosol, or lower the dosage, without checking with your doctor or pharmacist.
Do not allow the Atrovent Metered Aerosol spray to enter the eyes.
Things to be careful of
Be careful driving or operating machinery until you know how Atrovent Metered Aerosol affects you.
Atrovent Metered Aerosol may cause dizziness and blurred vision in some people. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous. Children should be careful when performing physical activities.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using Atrovent Metered Aerosol.
This medicine helps most people with asthma or COPD, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Do not be alarmed by the following list of possible side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
If you get any side effects, do not stop using the Atrovent Metered Aerosol without first talking to your doctor or pharmacist.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- headache
- dizziness
- dry or sore mouth
- throat irritation
- cough
- nausea, vomiting
- a change in bowel movements (e.g. constipation, diarrhoea)
- wind, indigestion, reflux (an unusual backflow of fluid).
These are the more common side effects of Atrovent Metered Aerosol. Usually they are mild and short-lived.
Tell your doctor as soon as possible if you experience difficulty passing urine. This is a serious side effect that may require medical attention.
If any of the following happen, tell your doctor immediately or go to Emergency at your nearest hospital:
- difficulty breathing or worsening of your breathing problems
- spasm of the muscles around the voice box, causing choking
- swelling of the throat
- fast or irregular heart beat, also called palpitations
- pounding heart beat
- allergic reaction (shortness of breath, wheezing or troubled breathing; swelling of the face, lips, tongue or other parts of the body, rash, itching or hives on the skin)
- irritation or a feeling of having something in the eye, dilated pupils, blurred vision, visual halos or coloured images.
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell. Other side effects not listed above may occur in some people.
After using Atrovent Metered Aerosol
Cleaning
Clean and dry your mouthpiece at least once a week. It is important to keep the mouthpiece of your inhaler clean and dry. It may not work as well if it gets dirty. Follow the instructions at the end of this leaflet for cleaning your mouthpiece.
Storage
Keep your inhaler in a cool dry place where the temperature stays below 25°C.
Do not store Atrovent Metered Aerosol or any other medicine in the bathroom or near a sink.
Do not leave it in the car on hot days or on window sills. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor or pharmacist tells you to stop using Atrovent Metered Aerosol or it has passed its expiry date, ask your pharmacist what to do with any that are left over.
Do not puncture the container or throw it into a fire.
Product description
What it looks like
Atrovent Metered Aerosol is supplied in a can containing 200 puffs.
Ingredients
Each Atrovent Metered Aerosol dose contains 21 micrograms ipratropium bromide monohydrate as the active ingredient.
It also contains:
- citric acid
- purified water
- absolute ethanol
- norflurane.
Atrovent Metered Aerosol does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
Supplier
Atrovent Metered Aerosol is supplied in Australia by:
Boehringer Ingelheim Pty Limited
ABN 52 000 452 308
Sydney NSW
www.boehringer-ingelheim.com.au
This Consumer Medicine Information was revised October 2019.
® Atrovent is a registered trade mark of Boehringer Ingelheim
© Boehringer Ingelheim Pty Limited 2019
Australian Registration Number:
AUST R 91129
Atrovent®
Metered Aerosol
ipratropium bromide monohydrate
Directions for use
The mouthpiece has been designed for use with the Atrovent Metered Aerosol canister so that you always get the right amount of medicine.
The mouthpiece must never be used with any other metered aerosol canister. The Atrovent Metered Aerosol canister must not be used with any mouthpiece other than the one supplied with this product.
The container is under pressure and should not be opened by force or exposed to temperatures above 50°C.
How to use the metered aerosol
Note:
Before first use of a new inhaler, prime it by activating twice into the atmosphere.
Before use if the inhaler has not been used for 3 days, reprime by activating once into the atmosphere.
- Remove the protective cap from the short end of the mouthpiece.
- Hold the inhaler with the arrow on the canister pointing upwards. Do not spray into or around the eyes.
- Breathe out fully, to the end of a normal breath.
- Place the open end of the mouthpiece between the lips but not past the teeth (Figure 1).
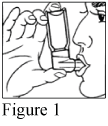
- Breathe in slowly through the mouth and at the same time firmly press the canister down once. Do not stop breathing when you press on the canister - continue to take a full, deep breath.
- Hold your breath for about ten seconds if possible, then breathe out slowly. Remove the inhaler from your mouth.
- If a second or subsequent inhalation is necessary, start again from instruction 3.
Replace the protective cap on the mouthpiece.
How to check how full your inhaler is
Your inhaler is not transparent and makes almost no sound when it is shaken, so it is difficult to know when your inhaler is nearly empty.
The inhaler will deliver 200 puffs. When the labelled number of doses have been used (usually after 3 weeks when used as recommended) the canister may still appear to contain a small amount of fluid. The inhaler should, however, be replaced so that you can be certain that you are getting the right amount of medicine in each actuation.
Care and cleaning
In order to obtain the best service from your Atrovent Metered Aerosol, these simple directions on maintenance should be carefully observed.
One of the most common causes of failure of the metering device of the inhaler to function is a dirty or blocked mouthpiece.
Always replace the protective cap on the mouthpiece after use to prevent the inside of the mouthpiece and canister from becoming dusty.
Clean your mouthpiece at least once a week. It is important to keep the mouthpiece of your inhaler clean to ensure that medicine does not build up and block the spray.
Remember to take out the canister before you wash the mouthpiece. When removing the canister from the mouthpiece pull out carefully to avoid bending the thin plastic stem.
Never press the thin plastic stem of the canister against a firm surface to check for function. This may bend the stem and lead to malfunction of the inhaler.
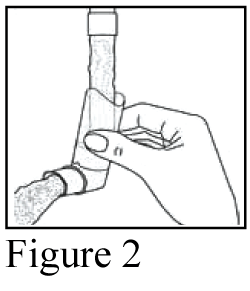
For cleaning, first take off the dust cap and remove the canister from the mouthpiece. Rinse warm water through the mouthpiece until no medication build-up and/or dirt is visible (Figure 2).
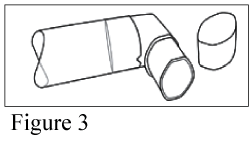
After cleaning, shake out the mouthpiece and let it air-dry without using any heating system (Figure 3). Once the mouthpiece is dry, replace the canister and the dust cap.
Using Atrovent Metered Aerosol with a spacer
Your doctor or pharmacist may suggest that you use a device called a 'spacer' with your inhaler, to help you use your inhaler more effectively.
A spacer is a holding chamber into which you spray your inhaler before inhaling.
Follow the instructions supplied with the spacer, to use your inhaler and spacer together properly.
Before first using your spacer, wash it with warm water and a small amount of detergent. Allow it to dry without rinsing, or drying with a cloth. Repeat this cleaning process at least once every month. This will help keep your spacer clean and to work properly.
Make sure you breathe in and out through the spacer after each puff from your inhaler. If you need to be shown how to use your spacer, or if you are having difficulties using your spacer, discuss this with your pharmacist.
Published by MIMS October 2022




