What is in this leaflet
This leaflet answers some common questions about B. BRAUN FENTANYL injection. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you receiving B. BRAUN FENTANYL against the benefits this medicine is expected to have for you.
If you have any concerns about receiving B. BRAUN FENTANYL, ask your doctor or pharmacist. Keep this leaflet with your medicine. You may need to read it again.
What B. BRAUN FENTANYL is used for
B. BRAUN FENTANY L injection is used to provide short-term pain relief and to help anaesthesia when you have an operation. It is a strong painkiller for use in hospitals.
B. BRAUN FENTANYL injection contains a medicine called fentanyl. It belongs to a group of medicines known as narcotic analgesics or opioids. Fentanyl relieves pain by blocking the nerves in the brain that recognise pain messages from the body.
Your doctor may have prescribed B. BRAUN FENTANYL for another reason. Ask your doctor if you have any questions about why this medicine has been prescribed for you.
B. BRAUN FENTANYL may be addictive. Addiction is unlikely in patients who receive it under medical supervision.
You may be at more risk of addiction if:
- You or someone in your family have a history of drug and alcohol abuse or mental illness
- You require repeated injections of B. BRAUN FENTANYL
- You need increasingly larger doses of B. BRAUN FENTANYL to control your pain.
Before you are given B. BRAUN FENTANYL
When you must not be given B. BRAUN FENTANYL
B. BRAUN FENTANYL should not be used:
- if you have an allergy to B. BRAUN FENTANYL, other strong pain killers or any of the ingredients. See Product Description at the end of this leaflet for a list of ingredients.
- if you suffer from asthma or if you are particularly prone to breathing difficulties (for example, in the case of head injury, coma or brain tumour).
- if you have taken a type of medicine known as monoamine oxidase inhibitors (MAOIs) within the last 14 days. These include phenelzine, moclobemide and tranylcypromine (for depression) and selegiline (for Parkinson's disease).
- if you suffer from a condition known as myasthenia gravis which causes constant weakness of muscles.
- in children less than two years old.
B. BRAUN FENTANYL should not be used if the packaging is torn or shows signs of tampering. It should not be used beyond the expiry date (month and year) printed on the pack.
Before you are given B. BRAUN FENTANYL
You must tell your doctor if:
- you are pregnant or planning to become pregnant
- you are breast feeding or wish to breastfeed. Fentanyl is excreted in human milk
- you have a lung disease or breathing problems such as severe asthma, severe bronchitis or emphysema
- you have a brain disorder
- you know you have a slow or irregular heart beat or heart problems
- you have or have ever had kidney or liver diseases
- you or someone in your family have a history of drug and alcohol abuse or mental illness
- you have an underactive thyroid gland.
- if you take any medicine that slows down your reactions (CNS depressants), especially benzodiazepines or related drugs.
If you have not told your doctor or pharmacist about any of the above, tell them before you are given B. BRAUN FENTANYL.
Your doctor will decide whether or not to treat you with B. BRAUN FENTANYL or whether to adjust the dose or alter your treatment.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including medicines you can buy without a prescription from a pharmacy, supermarket or health food shop.
In particular, tell your doctor or pharmacist if you are taking any of the following:
- medicines which make you feel drowsy or slow to react (CNS depressants), such as benzodiazepines or related drugs sleeping tablets, tranquillisers, alcohol, strong pain-killers or or some illegal drugs.
- The dose of B. BRAUN FENTANYL may have to be decreased. Also, if you receive a strong painkiller or other CNS depressant after receiving B. BRAUN FENTANYL during surgery, the dose of the painkiller or other CNS depressants may need to be lowered to reduce the risk of potentially serious side effects such as breathing difficulties, with slow or shallow breathing, severe drowsiness and decreased awareness, coma and death.
- medicines for mental disorders or a group of medicine called monoamine oxidase inhibitors (MAOIs). These include phenelzine, moclobemide and tranylcypromine (for depression) and selegiline (for Parkinson's disease). They should be stopped for 14 days before B. BRAUN FENTANYL is given.
- medicines for depression known as selective serotonin re-uptake inhibitors (SSRIs) or serotonin norepinephrine re-uptake inhibitors (SNRIs) These include fluoxetine, paroxetine, sertraline and venlafaxine
- ritonavir (a protease inhibitor used to treat HIV).
- medicines for infections such as voriconazole or fluconazole.
These medicines may be affected by B. BRAUN FENTANYL or may affect how well B. BRAUN FENTANYL works.
Your doctor or pharmacist can tell you what to do if you are taking any of these medicines.
Effect on driving and operating machinery
B. BRAUN FENTANYL can have a negative effect on your alertness and ability to drive and operate machinery. Do not drive or operate machinery until your doctor says it is safe.
Effect of alcohol
B. BRAUN FENTANYL can increase the effect of alcohol.
Addiction
You can become addicted to B. BRAUN FENTANYL even if you take it exactly as prescribed. B. BRAUN FENTANYL may become habit forming causing mental and physical dependence. If abused it may become less able to reduce pain.
Dependence
As with all other opioid containing products, your body may become used to you taking B. BRAUN FENTANYL. Taking it may result in physical dependence. Physical dependence means that you may experience withdrawal symptoms if you stop taking B. BRAUN FENTANYL suddenly, so it is important to take it exactly as directed by your doctor.
Tolerance
Tolerance to B. BRAUN FENTANYL may develop, which means that the effect of the medicine may decrease. If this happens, more may be needed to maintain the same effect.
Withdrawal
Continue taking your medicine for as long as your doctor tells you. If you stop having this medicine suddenly, your pain may worsen and you may experience some or all of the following withdrawal symptoms:
- nervousness, restlessness, agitation, trouble sleeping or anxiety
- body aches, weakness or stomach cramps
- loss of appetite, nausea, vomiting or diarrhoea
- increased heart rate, breathing rate or pupil size
- watery eyes, runny nose, chills or yawning
- increased sweating.
B. BRAUN FENTANYL given to the mother during labour can cause breathing problems and signs of withdrawal in the newborn.
Using B. BRAUN FENTANYL
How it is given
B. BRAUN FENTANYL is given by your doctor as an injection into a muscle or a vein. Your doctor will decide how much B. BRAUN FENTANYL you need. This will depend on your age, body weight, medical conditions and history.
Adults
The usual dose of B. BRAUN FENTANYL ranges from 50 micrograms to 100 micrograms depending on what it is being used for. Repeat doses may be given in some cases.
If you are elderly, you may be given a lower dose of B. BRAUN FENTANYL.
Children 2 to 12 years old
The usual dose ranges from 20 micrograms to 30 micrograms per 10 kg of body weight.
B. BRAUN FENTANYL is not recommended for use in children under 2 years.
If you are given too much (overdose)
Immediate medical care is required. The main symptom is markedly weakened breathing.
The doctor or the anaesthetist giving you B. BRAUN FENTANYL will be experienced in its use, so it is unlikely that you will be given too much.
In the unlikely event that an overdose occurs, your doctor or the anaesthetist will take the necessary actions. The symptoms of overdose could include:
- Slow, unusual or difficult breathing
- Drowsiness, dizziness or unconsciousness
- Slow or weak heartbeat
- Nausea or vomiting
- Convulsions or fits
- Muscle stiffness
- Lowering of blood pressure
- Lowering of heart rate
If these symptoms occur, you may be administered another medicine (e.g. naloxone) to help reverse the effects.
If you think you or anybody else has taken too much B. BRAUN FENTANYL, contact your doctor, pharmacist or the Poisons Information Centre who will advise you what to do.
You can contact the Poisons Information Centre by dialing:
- Australia: 13 11 26
While you are given B. BRAUN FENTANYL
Things you must do
- Always follow your doctor's instructions carefully.
- Tell your doctor if you become pregnant.
- Tell your doctor or pharmacist if you are about to start taking a new medicine.
- Tell your doctor or nurse if you do not feel well after being given B. BRAUN FENTANYL (see SIDE EFFECTS).
Things you must not do
- Do not use B. BRAUN FENTANYL to treat any other complaint unless your doctor says so.
- Do not give this medicine to anyone else, even if their symptoms seem similar to yours.
- Do not drive or operate machinery until your doctor says it is safe to do so.
If you do not understand the instructions provided with this medicine, ask your doctor or pharmacist for help.
Side Effects
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some side effects. Do not be alarmed by this list of possible side effects. You may not experience any of them. Ask your doctor or pharmacist to answer any questions you may have.
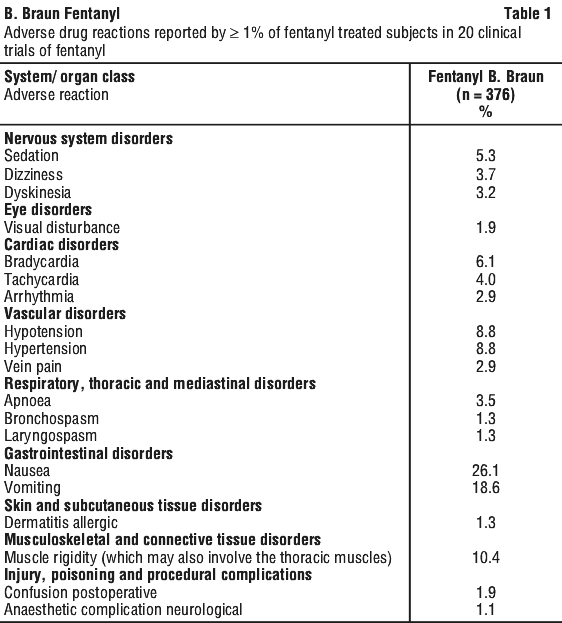
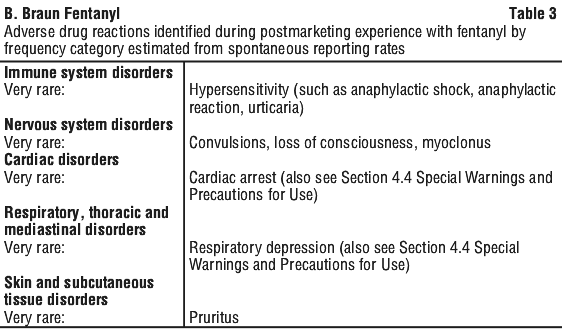
Tell your doctor immediately if you notice any of the following as you may need urgent medical care:
- difficulty in breathing (abnormally slow and/or weak breathing or increased breathing rate or a temporary cessation of breathing)
- muscle stiffness or involuntary muscle movements, including slow, stiff or jerking movements
- slow, fast or irregular heart beat or cardiac arrest
- a feeling of choking which is caused by the spasm of the muscles around the voice box
- allergic reactions such as skin rash, redness and swelling of the face, neck or throat.
- severe drowsiness
- convulsions
- loss of consciousness
Tell your doctor if you experience any of the following:
- dizziness
- low or high or variable blood pressure which may cause headache, weakness or dizziness
- hiccups
- blurred vision
- nausea, vomiting
- excessive sweating
- itching
- an unusual sense of well being
- sedation
- headaches
- post-operative confusion or agitation
- neurological or airway complications of anaesthesia
- vein pain or inflammation
- chills or lowered body temperature
- visual disturbance
If you become dependent on B. BRAUN FENTANYL and your injections are suddenly stopped or greatly reduced, you may experience withdrawal symptoms such as nausea, vomiting, diarrhoea, anxiety and shivering.
Other side effects not listed above may also occur in some people. Tell your doctor if you notice any other effects.
Storage and disposal
B. BRAUN FENTANYL should be kept in the pack until use, protected from light, in a cool dry place where the temperature stays below 25°C.
B. BRAUN FENTANYL will be kept in a locked cupboard in the hospital pharmacy or operating theatre.
B. BRAUN FENTANYL should not be used after the date (month and year) printed after "EXP". The anaesthetist will inspect B. BRAUN FENTANYL before use to determine that it is still within its use by date.
The hospital staff looking after you will dispose of any remaining B. BRAUN FENTANYL appropriately.
Product Description
What it looks like
B. BRAUN FENTANYL injection is a clear, colourless solution. It is available in 2 mL, 5 mL and 10 mL glass ampoules. The 2 mL ampoule is available in boxes of 10, the 5mL ampoule is available in boxes of 10 and the 10 mL ampoule is available in boxes of 5 and 10, for hospital use only.
Registration numbers
100 micrograms/2 mL ampoule: AUST R 227122
250 micrograms/5 mL ampoule: AUST R 227123
500 micrograms/10 mL ampoule: AUST R 227124
Ingredients
B. BRAUN FENTANYL contains 50 micrograms of fentanyl per mL (as fentanyl citrate). The 2 mL ampoule contains 100 micrograms of fentanyl, the 5 mL ampoule contains 250 micrograms of fentanyl, and the 10 mL ampoule contains 500 micrograms of fentanyl.
B. BRAUN FENTANYL also contains sodium chloride and water.
Sponsor
B. Braun Australia Pty Ltd
Level 5, 7-9 Irvine Place
Bella Vista
NSW 2153
Australia
This leaflet was prepared in March 2021.
Published by MIMS September 2021

 Additional ADRs that occurred in < 1% of fentanyl treated subjects in the 20 clinical trials are listed in Table 2.
Additional ADRs that occurred in < 1% of fentanyl treated subjects in the 20 clinical trials are listed in Table 2.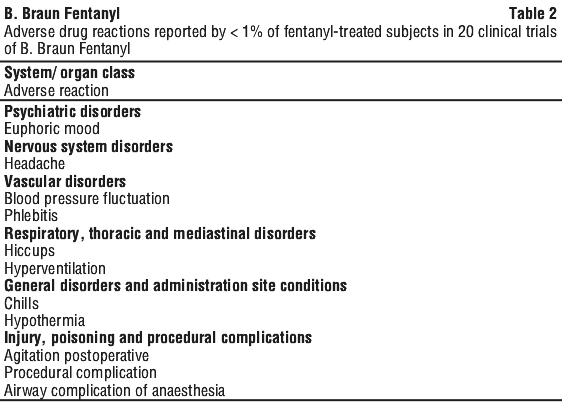
 When a neuroleptic is used with fentanyl, the following adverse reactions may be observed: chills and/or shivering; restlessness, postoperative hallucinatory episodes; and extrapyramidal symptoms.
When a neuroleptic is used with fentanyl, the following adverse reactions may be observed: chills and/or shivering; restlessness, postoperative hallucinatory episodes; and extrapyramidal symptoms.
 N-phenyl-N-[1-(2-phenylethyl) piperidin-4-yl]propanamide dihydrogen 2-hydroxypropane-1,2,3-tricarboxylate.
N-phenyl-N-[1-(2-phenylethyl) piperidin-4-yl]propanamide dihydrogen 2-hydroxypropane-1,2,3-tricarboxylate.