1 Name of Medicine
Bendamustine hydrochloride.
2 Qualitative and Quantitative Composition
Bendamustine Sandoz 25 mg powder for injection vial.
Each 25 mg vial contains 25 mg of bendamustine hydrochloride (equivalent to 26.1 mg bendamustine hydrochloride monohydrate).
Bendamustine Sandoz 100 mg powder for injection vial.
Each 100 mg vial contains 100 mg of bendamustine hydrochloride (equivalent to 104.6 mg bendamustine hydrochloride monohydrate).
List of excipients with known effect.
Mannitol.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Powder for injection, for intravenous infusion.
Bendamustine Sandoz is a white to off-white, freeze-dried, lyophilisate powder for concentrate for solution for infusion.
4.1 Therapeutic Indications
First-line treatment of chronic lymphocytic leukaemia (Binet stage B or C). Efficacy relative to first-line therapies other than chlorambucil has not been established.
Previously untreated indolent CD20-positive, stage III-IV Non-Hodgkin's lymphoma, in combination with rituximab.
Previously untreated CD20-positive, stage III-IV Mantle Cell Lymphoma in combination with rituximab, in patients ineligible for autologous stem cell transplantation.
Relapsed/refractory indolent non-Hodgkin's lymphoma.
4.2 Dose and Method of Administration
For intravenous infusion over 30-60 minutes (see Section 4.2 Dose and Method of Administration, Special precautions for disposal and handling).
Infusion must be administered under the supervision of a physician qualified and experienced in the use of chemotherapeutic agents.
Poor bone marrow function is related to increased chemotherapy-induced haematological toxicity. Treatment should not be started if leukocyte and/or platelet values drop to < 3 x 109/L or < 75 x 109/L, respectively (see Section 4.4 Special Warnings and Precautions for Use, Myelosuppression).
Monotherapy for chronic lymphocytic leukaemia.
100 mg/m2 body surface area bendamustine hydrochloride on days 1 and 2; every 4 weeks, for up to 6 cycles.
Monotherapy for indolent non-Hodgkin's lymphomas refractory to rituximab.
120 mg/m2 body surface area bendamustine hydrochloride on days 1 and 2; every 3 weeks, for at least 6 to 8 cycles (maximum 8 cycles).
Combination therapy with rituximab for first-line non-Hodgkin's lymphoma and mantle cell lymphoma.
90 mg/m2 on days 1 and 2 of a 4-week cycle for up to 6 cycles.
Treatment should be terminated or delayed if leukocyte and/or platelet values dropped to < 3 x 109/L or < 75 x 109/L, respectively. Treatment can be continued after leukocyte values have increased to > 4 x 109/L and platelet values to > 100 x 109/L.
The leukocyte and platelet nadir is reached after 14-20 days with regeneration after 3-5 weeks. During therapy free intervals strict monitoring of the blood count is recommended (see Section 4.4 Special Warnings and Precautions for Use).
In case of non-haematological toxicity dose reductions have to be based on the worst CTC grades in the preceding cycle. A 50% dose reduction is recommended in case of CTC Grade 3 toxicity. An interruption of treatment is recommended in case of CTC Grade 4 toxicity. If the toxicity resolves and the previous dose is tolerated, the reduced dose may be increased again.
If a patient requires a dose modification the individually calculated reduced dose must be given on day 1 and 2 of the respective treatment cycle.
The powder for concentrate for solution for infusion has to be reconstituted with water for injections, diluted with sodium chloride 9 mg/mL (0.9%) solution for injection and then administered by intravenous infusion. Aseptic technique is to be used.
1. Reconstitution.
Reconstitute each vial of Bendamustine Sandoz containing 25 mg bendamustine hydrochloride in 10 mL water for injections by shaking.
Reconstitute each vial of Bendamustine Sandoz containing 100 mg bendamustine hydrochloride in 40 mL water for injections by shaking.
The reconstituted concentrate contains 2.5 mg bendamustine hydrochloride per mL and appears as a clear colourless solution.
No overfill is included.
2. Dilution.
As soon as a clear solution is obtained (usually after 10 minutes) dilute the total recommended dose of Bendamustine Sandoz immediately with 0.9% NaCl solution to produce a final volume of about 500 mL.
Bendamustine Sandoz must be diluted with 0.9% NaCl solution and not with any other injectable solution.
3. Administration.
Bendamustine is soluble in water when at room temperature. It is administered by intravenous infusion after reconstitution with 10 mL (for the 25 mg vial) or 40 mL (for the 100 mg vial) water for injections and further dilution with physiological saline (0.9%).
The solution is administered by intravenous infusion over 30-60 min.
Dosage adjustment.
Renal impairment.
On the basis of pharmacokinetic data, no dose adjustment is necessary in patients with a creatinine clearance of > 10 mL/min. Experience in patients with severe renal impairment is limited.
Hepatic impairment.
On the basis of pharmacokinetic data, no dose adjustment is necessary in patients with mild hepatic impairment (serum bilirubin < 1.2 mg/dL). A 30% dose reduction is recommended in patients with moderate hepatic impairment (serum bilirubin 1.2 - 3.0 mg/dL).
No data is available in patients with severe hepatic impairment (serum bilirubin values of > 3.0 mg/dL) (see Section 4.3 Contraindications).
Paediatric patients.
There is no experience in children and adolescents with bendamustine.
Elderly patients.
There is no evidence that dose adjustments are necessary in elderly patients.
Special precautions for disposal and handling.
When handling Bendamustine Sandoz, inhalation, skin contact or contact with mucous membranes should be avoided (wear gloves and protective clothes). Contaminated body parts should be carefully rinsed with water and soap, the eye should be rinsed with physiological saline solution. If possible it is recommended to work on special safety workbenches (laminar flow) with liquid impermeable, absorbing disposable foil. Pregnant personnel should be excluded from handling cytostatics.
The product is for single use in one patient only. Discard any residue. Any unused product or waste material should be disposed of in accordance with local requirements.4.3 Contraindications
Bendamustine Sandoz is contraindicated in patients with:
Hypersensitivity to the active substance or to any of the excipients.
Severe hepatic impairment (serum bilirubin > 3.0 mg/dL).
Jaundice.
Severe bone marrow suppression and severe blood count alteration (leukocyte and/or platelet values dropped to < 3 x 109/L or < 75 x 109/L, respectively).
Major surgery less than 30 days before start of treatment.
Infections, especially involving leukocytopaenia.
Yellow fever vaccination.
Bendamustine is also contraindicated during breastfeeding.
4.4 Special Warnings and Precautions for Use
When considering combination treatment with rituximab, please consult the Product Information for rituximab and consider this in conjunction with the information provided below.
Myelosuppression.
Patients treated with bendamustine may experience bone marrow failure. In the event of treatment-related myelosuppression, leukocytes, platelets, haemoglobin, and neutrophils must be monitored at least weekly. Treatment-related myelosuppression may require dose adjustment and/or dose delays.
Treatment with bendamustine hydrochloride may cause prolonged lymphocytopenia (< 0.6 x 109/L) and low CD4-positive T-cell (T-helper cell) counts (< 0.2 x 109/L) for at least 7-9 months after the completion of treatment. Lymphocytopenia and CD4-positive T-cell depletion are more pronounced when bendamustine is combined with rituximab. Patients with lymphopenia and low CD4-positive T-cell count following treatment with bendamustine hydrochloride are more susceptible to (opportunistic) infections.
Prior to the initiation of the next cycle of therapy, the following parameters are recommended: Leukocyte and/or platelet values > 4 x 109/L or > 100 x 109/L, respectively. Treatment should not be started if leukocyte and/or platelet values drop to < 3 x 109/L or < 75 x 109/L, respectively.
Bendamustine should not be used during severe bone marrow suppression and severe blood count alterations (see Section 4.2 Dose and Method of Administration).
Infections.
Serious and fatal infections, including fatal sepsis, have occurred with bendamustine treatment. These infections included bacterial (pneumonia) and opportunistic infections such as Pneumocystis jirovecii Pneumonia (PJP), Varicella Zoster Virus (VZV), Cytomegalovirus (CMV), and progressive multifocal leukoencephalopathy (John Cunningham virus).
Cases of progressive multifocal leukoencephalopathy (PML) including fatal ones have been reported following the use of bendamustine mainly in combination with rituximab or obinutuzumab.
Patients with lymphopenia and low CD4-positive T-cell count following treatment with bendamustine hydrochloride are more susceptible to (opportunistic) infections. In case of low CD4-positive T-cell counts (< 0.2 x 109/L) Pneumocystis jirovecii pneumonia (PJP) prophylaxis should be considered. All patients should be monitored for respiratory signs and symptoms throughout treatment. Discontinuation of bendamustine hydrochloride should be considered if there are signs of (opportunistic) infections.
Consider PML in the differential diagnosis in patients with new or worsening neurological, cognitive or behavioural signs or symptoms. If PML is suspected then appropriate diagnostic evaluations should be undertaken and treatment suspended until PML is excluded.
Hepatitis B reactivation.
Reactivation of hepatitis B in patients who are chronic carriers of this virus has occurred after these patients received bendamustine hydrochloride. Some cases resulted in acute hepatic failure or a fatal outcome. Patients should be tested for HBV infection before initiating treatment with bendamustine hydrochloride. For patients with hepatitis B serology indicative of prior infection, a liver disease expert should be consulted before the start of treatment and the patient should be monitored and managed following local medical standards to prevent hepatitis B reactivation. Monitoring should continue for several months following termination of therapy.
Skin reactions.
A number of skin reactions have been reported. These events have included rash, severe cutaneous reactions and bullous exanthema. Cases of Stevens-Johnson syndrome (SJS) and Toxic Epidermal Necrolysis (TEN), some fatal, have also been reported. Some events of SJS and TEN occurred when bendamustine hydrochloride was administered concomitantly with allopurinol or when bendamustine hydrochloride was given in combination with other anticancer agents. Cases of drug reaction with eosinophilia and systemic symptoms (DRESS) have been reported with the use of bendamustine hydrochloride in combination with rituximab. Patients should be advised of the signs and symptoms of these reactions by their prescribers and should be told to seek medical attention immediately if they develop these symptoms. Where skin reactions occur, they may be progressive and increase in severity with further treatment. If skin reactions are progressive, Bendamustine Sandoz should be withheld or discontinued. For severe skin reactions where a relationship to bendamustine hydrochloride is suspected, treatment should be discontinued.
Non-melanoma skin cancer.
In clinical studies, an increased risk for non-melanoma skin cancers (basal cell carcinoma and squamous cell carcinoma) has been observed in patients treated with bendamustine containing therapies. Periodic skin examination is recommended for all patients, particularly those with risk factors for skin cancer.
Patients with cardiac disorders.
During treatment with Bendamustine Sandoz the concentration of potassium in the blood must be closely monitored and potassium supplement must be given when K+ < 3.5 mEq/L, and ECG measurement must be performed.
Fatal cases of myocardial infarction and cardiac failure have been reported with bendamustine treatment. Patients with concurrent or history of cardiac disease should be observed closely.
Nausea, vomiting.
An antiemetic may be given for the symptomatic treatment of nausea and vomiting.
Tumour lysis syndrome.
Tumour lysis syndrome associated with bendamustine treatment has been reported in patients in clinical trials. The onset tends to be within 48 hours of the first dose of bendamustine and, without intervention, may lead to acute renal failure and death. Preventive measures include adequate volume status, close monitoring of blood chemistry, particularly potassium and uric acid levels. The use of allopurinol during the first one to two weeks of bendamustine therapy can be considered but not necessarily as standard. However, there have been a few cases of Stevens-Johnson syndrome and Toxic necrolysis reported when bendamustine and allopurinol are concomitantly administered.
Infusion reactions and anaphylaxis.
Infusion reactions to bendamustine hydrochloride have occurred in clinical trials. Symptoms are generally mild and include fever, chills, pruritus and rash. In rare instances severe anaphylactic and anaphylactoid reactions have occurred. Patients must be asked about symptoms suggestive of infusion reactions after their first cycle of therapy. Measures to prevent severe reactions, including antihistamines, antipyretics and corticosteroids must be considered in subsequent cycles in patients who have previously experienced infusion reactions.
Patients who experienced Grade 3 or worse allergic-type reactions were typically not re-challenged.
Extravasation.
An extravasal injection should be stopped immediately. The needle should be removed after a short aspiration. Thereafter the affected area of tissue should be cooled. The arm should be elevated. Additional treatments like the use of corticosteroids are not of clear benefit.
Other malignancies.
The risk of myelodysplastic syndrome and acute myeloid leukaemias is increased in patients treated with alkylating agents (including bendamustine). The secondary malignancy may develop several years after chemotherapy has been discontinued.
Use in the elderly.
No data available.
Paediatric use.
There is no experience in children and adolescents with Bendamustine Sandoz.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
No in vivo interaction studies have been performed.
When Bendamustine Sandoz is combined with myelosuppresive agents, the effect of bendamustine and/or the co-administered medicinal products on the bone marrow may be potentiated. Any treatment reducing the patient's performance status or impairing bone marrow function can increase the toxicity of bendamustine.
Combination of Bendamustine Sandoz with ciclosporin or tacrolimus may result in excessive immunosuppression with risk of lymphoproliferation.
Cytostatics can reduce antibody formation following live-virus vaccination and increase the risk of infection which may lead to fatal outcome. This risk is increased in subjects who are already immunosuppressed by their underlying disease.
Bendamustine metabolism involves cytochrome P450 (CYP) 1A2 isoenzyme (see Section 5.2 Pharmacokinetic Properties). Therefore, potential for interaction with CYP1A2 inhibitors such as fluvoxamine, ciprofloxacin, aciclovir, cimetidine exists. Based on in vitro data, bendamustine is not likely to inhibit metabolism via CYP1A2, 2C9/10, 2D6, 2E1, or 3A4/5, or to induce metabolism of substrates of cytochrome P450 enzymes.
The role of active transport systems in bendamustine distribution has not been fully evaluated. In vitro data suggest that P-glycoprotein, breast cancer resistance protein (BCRP), and/or other efflux transporters may have a role in bendamustine transport. Inhibitors of these transporters may increase the plasma concentration of bendamustine.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No fertility studies have been conducted in animals. However, results from toxicity studies in rats and dogs indicate the potential of bendamustine to impair male reproductive function and fertility. Testicular atrophy was seen in dogs that received ≥ 1.65 mg/kg IV bendamustine, while at higher doses (6.6 mg/kg IV to dogs and ≥ 40 mg/kg/day PO to rats), reduced spermatogenesis was seen. Estimated exposures at the no-effect level were subclinical in dogs and similar to the clinical exposure in rats. Advice on conservation of sperm should be sought prior to treatment because of the possibility of irreversible infertility due to therapy with bendamustine.
Impaired spermatogenesis, azoospermia, and total germinal aplasia have been reported in male patients treated with alkylating agents, especially in combination with other drugs. In some instances spermatogenesis may return in patients in remission, but this may occur only several years after intensive chemotherapy has been discontinued. Patients should be warned of the potential risk to their reproductive capacities.
(Category D)
There are insufficient data from the use of Bendamustine Sandoz in pregnant women. Bendamustine was shown to be embryo/feto-lethal and teratogenic in animal studies. Single intra-peritoneal doses of bendamustine to pregnant mice (210 mg/m2) and rats (60 mg/m2) during the period of organogenesis, resulted in embryo-foetal lethality, decreased foetal body weights and an increase in foetal skeletal and visceral malformations (cleft palate, turricephaly, rib malformations and spinal deformities, and hepatic or intestinal ectopia).
During pregnancy Bendamustine Sandoz should not be used unless clearly necessary. The mother should be informed about the risk to the foetus. If treatment with Bendamustine Sandoz is absolutely necessary during pregnancy or if pregnancy occurs during treatment, the patient should be informed about the risks for the unborn child and be monitored carefully. The possibility of genetic counselling should be considered.
Women of childbearing potential must use effective methods of contraception both before and during bendamustine therapy.
Men being treated with Bendamustine Sandoz are advised not to father a child during and for up to 6 months following cessation of treatment. Advice on conservation of sperm should be sought prior to treatment because of the possibility of irreversible infertility due to therapy with bendamustine.
It is not known whether bendamustine passes into the breast milk, therefore, bendamustine is contraindicated during breastfeeding (see Section 4.3 Contraindications). Breastfeeding must be discontinued during treatment with bendamustine.4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed. However, somnolence has been reported during treatment with bendamustine (see Section 4.8 Adverse Effects (Undesirable Effects)). Patients should be instructed that if they experience these symptoms they should avoid potentially hazardous tasks such as driving and using machines.
4.8 Adverse Effects (Undesirable Effects)
Clinical trials data.
Chronic lymphocytic lymphoma.
The following tables describe the safety results reported in study 02CLLIII of 161 previously-untreated patients with Binet Stage B or C CLL requiring treatment. See Table 1.
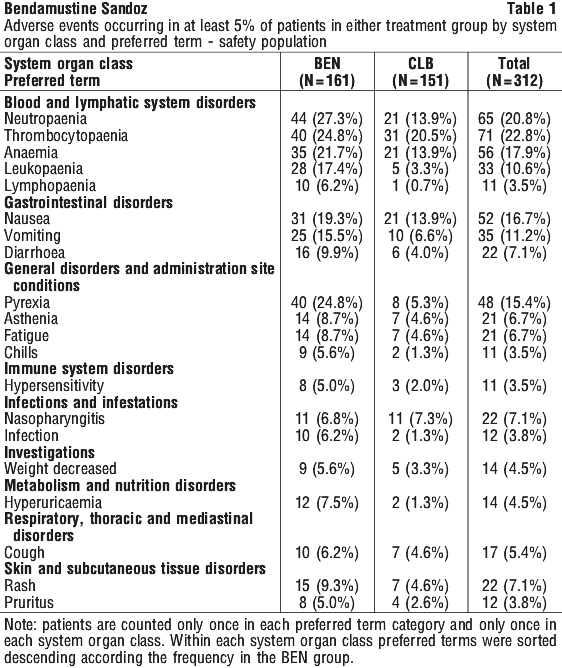 A total of 50 patients had 60 serious adverse events. Most frequently occurring serious adverse events in the bendamustine group were hypersensitivity and pneumonia (each with 3 patients) and anaemia, vomiting, pyrexia and tumour lysis syndrome (each with 2 patients). Most frequent documented serious adverse event in the chlorambucil group was herpes zoster (with 2 patients). All other events were documented only once by patient. See Table 2.
A total of 50 patients had 60 serious adverse events. Most frequently occurring serious adverse events in the bendamustine group were hypersensitivity and pneumonia (each with 3 patients) and anaemia, vomiting, pyrexia and tumour lysis syndrome (each with 2 patients). Most frequent documented serious adverse event in the chlorambucil group was herpes zoster (with 2 patients). All other events were documented only once by patient. See Table 2.
 Number of adverse events possible, probable or definite related to the study medication (including missing relationship) was higher in the bendamustine arm than in the chlorambucil arm. Especially blood and lymphatic system disorders, gastrointestinal disorders and pyrexia occurred more frequently under bendamustine than under chlorambucil. See Table 3.
Number of adverse events possible, probable or definite related to the study medication (including missing relationship) was higher in the bendamustine arm than in the chlorambucil arm. Especially blood and lymphatic system disorders, gastrointestinal disorders and pyrexia occurred more frequently under bendamustine than under chlorambucil. See Table 3.
 The most frequent adverse reactions leading to study withdrawal for patients receiving bendamustine were hypersensitivity (2%) and pyrexia (1%).
The most frequent adverse reactions leading to study withdrawal for patients receiving bendamustine were hypersensitivity (2%) and pyrexia (1%).
Results from the NHL1-2003 clinical trial in patients with previously untreated advanced indolent non-Hodgkin's lymphoma and mantle cell lymphoma.
Table 4 and Table 5 describe safety data from the NHL1-2003 study with previously untreated advanced indolent NHL who received bendamustine IV (90 mg/m2) in combination with rituximab (375 mg/m2).
Adverse event data provided below is based on published data and is therefore limited in nature.


Relapsed/refractory non-Hodgkin's lymphoma.
Table 6 lists adverse events occurring in at least 5% of patients in study SDX-105-03.
 The data described below reflect exposure to bendamustine in 176 patients with indolent B-cell NHL treated in two single-arm studies (SDX-105-03 and SDX-105-01).
The data described below reflect exposure to bendamustine in 176 patients with indolent B-cell NHL treated in two single-arm studies (SDX-105-03 and SDX-105-01).
The adverse reactions occurring in at least 5% of the NHL patients, regardless of severity, are shown in Table 7.
 Haematologic toxicities, based on laboratory values and CTC grade, in NHL patients treated in both single arm studies combined are described in Table 8. Clinically important chemistry laboratory values that were new or worsened from baseline and occurred in > 1% of patients at Grade 3 or 4, in NHL patients treated in both single arm studies combined were hyperglycaemia (3%), elevated creatinine (2%), hyponatraemia (2%), and hypocalcaemia (2%).
Haematologic toxicities, based on laboratory values and CTC grade, in NHL patients treated in both single arm studies combined are described in Table 8. Clinically important chemistry laboratory values that were new or worsened from baseline and occurred in > 1% of patients at Grade 3 or 4, in NHL patients treated in both single arm studies combined were hyperglycaemia (3%), elevated creatinine (2%), hyponatraemia (2%), and hypocalcaemia (2%).
 In both studies, serious adverse reactions, regardless of causality, were reported in 37% of patients receiving bendamustine. The most common serious adverse reactions occurring in 5% of patients were febrile neutropaenia and pneumonia. Other important serious adverse reactions reported in clinical trials and/or postmarketing experience were acute renal failure, cardiac failure, hypersensitivity, skin reactions, pulmonary fibrosis, pneumonitis, pulmonary alveolar haemorrhage, and myelodysplastic syndrome.
In both studies, serious adverse reactions, regardless of causality, were reported in 37% of patients receiving bendamustine. The most common serious adverse reactions occurring in 5% of patients were febrile neutropaenia and pneumonia. Other important serious adverse reactions reported in clinical trials and/or postmarketing experience were acute renal failure, cardiac failure, hypersensitivity, skin reactions, pulmonary fibrosis, pneumonitis, pulmonary alveolar haemorrhage, and myelodysplastic syndrome.
Serious drug-related adverse reactions reported in clinical trials included myelosuppression, infection, pneumonia, tumour lysis syndrome and infusion reactions (see Section 4.4 Special Warnings and Precautions for Use). Adverse reactions occurring less frequently but possibly related to bendamustine treatment were haemolysis, dysgeusia/taste disorder, atypical pneumonia, sepsis, Herpes zoster, erythema, dermatitis, and skin necrosis.
Post marketing experience.
The following adverse reactions have been identified during post-approval use of bendamustine (Table 9).
The frequencies are provided according to the following convention:
Very common ≥ 1/10; Common ≥ 1/100 and < 1/10; Uncommon ≥ 1/ 1000 and < 1/100; Rare ≥ 1/10,000 and < 1/1000; Very rare < 1/10,000, including isolated reports.
 Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: anaphylaxis; and injection or infusion site reactions including phlebitis, pruritus, irritation, pain, and swelling. Increases in alanine aminotransferase; aspartate aminotransferase; blood bilirubin and blood urea levels have been reported. Somnolence, atrial fibrillations and palpitations have also been reported.
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: anaphylaxis; and injection or infusion site reactions including phlebitis, pruritus, irritation, pain, and swelling. Increases in alanine aminotransferase; aspartate aminotransferase; blood bilirubin and blood urea levels have been reported. Somnolence, atrial fibrillations and palpitations have also been reported.
Skin reactions including Stevens-Johnson Syndrome (SJS), Toxic Epidermal Necrolysis (TEN), and Drug Reaction with Eosinophilia and Systemic Symptoms (combination therapy with rituximab), some fatal, have been reported with the use of bendamustine hydrochloride (see Section 4.4 Special Warnings and Precautions for Use, Skin reactions, Tumour lysis syndrome). In addition, a few cases of hepatitis B reactivation resulting in hepatic failure have been reported in patients treated with bendamustine. Pancytopenia, headache, dizziness, opportunistic infection (e.g. herpes zoster, cytomegalovirus, Pneumocystis jirovecii pneumonia), bone marrow failure, hepatic failure, renal failure and nephrogenic diabetes insipidus have also been reported in patients treated with bendamustine.
The CD4/CD8 ratio may be reduced. A reduction of the lymphocyte count was seen. In immunosuppressed patients, the risk of infection (e.g. with herpes zoster) may be increased.
There have been isolated reports of necrosis after accidental extra-vascular administration, tumour lysis syndrome, and anaphylaxis.
The risk of myelodysplastic syndrome and acute myeloid leukaemias is increased in patients treated with alkylating agents (including bendamustine). The secondary malignancy may develop several years after chemotherapy has been discontinued.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
The maximum single dose of bendamustine received in clinical studies was 280 mg/m2 body surface area.
Three out of the four patients treated with 280 mg/m2 experienced ECG changes which were regarded as dose-limiting toxicities on days 7 and 21. These changes included prolonged QT, sinus tachycardia (1 patient), displaced ST and T waves (2 patients) and left interior fascicle block (1 patient).
There is no specific antidote for bendamustine overdosage. Supportive therapy should be given when needed.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Bendamustine hydrochloride is an alkylating antitumour agent with unique activity. The antineoplastic and cytocidal effect of bendamustine hydrochloride is based essentially on a cross-linking of DNA single and double strands by alkylation. As a result, DNA matrix functions and DNA synthesis and repair are impaired. Bendamustine is active against both quiescent and dividing cells.
The active substance revealed no or very low cross-resistance in human tumour cell lines with different resistance mechanisms.
The exact mechanism of action of bendamustine remains unknown.
Clinical trials.
Chronic lymphocytic lymphoma.
The safety and efficacy of bendamustine were evaluated in an open-label, randomised, controlled multicentre trial (02CLLIII) comparing bendamustine to chlorambucil. The trial was conducted in 319 previously-untreated patients with Binet Stage B or C CLL requiring treatment. Need-to-treat criteria included hematopoietic insufficiency, B-symptoms, rapidly progressive disease or risk of complications from bulky lymphadenopathy. Patients with autoimmune haemolytic anaemia or autoimmune thrombocytopenia, Richter's syndrome, or transformation to prolymphocytic leukaemia were excluded from the study.
The patient populations in the bendamustine and chlorambucil treatment groups were balanced with regard to the following baseline characteristics: age (median 63 vs. 66 years), gender (63% vs. 61% male), Binet stage (72% vs. 71% Binet B), lymphadenopathy (79% vs. 82%), enlarged spleen (77% vs. 78%), enlarged liver (49% vs. 45%), hypercellular bone marrow (80% vs. 73%), lymphocyte count (mean 68.9 x 109/L vs. 62.4 x 109/L). Ninety percent of patients in both treatment groups had immuno-phenotypic confirmation of CLL (CD5, CD23 and either CD19 or CD20 or both). Most of the patients had WHO-performance status (PS) 0 (70% vs. 65%). PS 1 was documented in 27% vs 29% patients.
Patients were randomly assigned to receive either bendamustine at 100 mg/m2, administered intravenously over a period of 30 minutes on Days 1 and 2 or chlorambucil at 0.8 mg/kg (Broca's normal weight) administered orally on Days 1 and 15 of each 28-day cycle.
The primary efficacy endpoints of overall response rate and progression-free survival were calculated using a pre-specified algorithm based on NCI working group criteria for CLL.
The results of this open-label randomised study demonstrated a higher rate of overall response and a longer progression-free survival for bendamustine compared to chlorambucil. Survival data are not mature.
An ICRA (Independent Committee for Response Assessment) review was completed 12 months after the last patient completed treatment in the study. The results from this follow up ITT analysis are presented in Table 10.
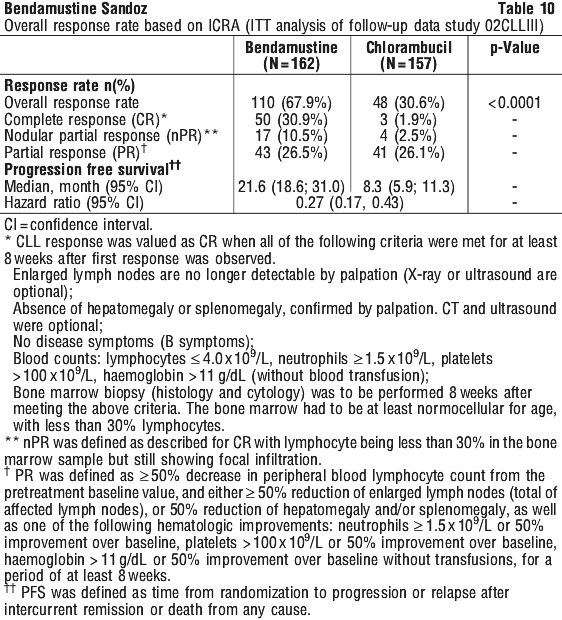 Complete response is lower than noted in Table 10 - for both bendamustine and chlorambucil - if there is a requirement for imaging to confirm lymph nodes ≤ 1.5 cm.
Complete response is lower than noted in Table 10 - for both bendamustine and chlorambucil - if there is a requirement for imaging to confirm lymph nodes ≤ 1.5 cm.
Progression free survival based upon the Independent Committee Response Assessment (ICRA) criteria by treatment group in the follow-up report (ITT analysis) in Study 02CLLIII is shown in Figure 1.

Previously untreated advanced indolent non-Hodgkin's lymphoma (NHL) and mantle cell lymphoma (MCL).
The safety and efficacy of bendamustine in previously untreated advanced indolent NHL and MCL have been assessed in a Phase III trial.
The NHL1-2003 study is a prospective phase III, multicentre, randomised (1:1), non-inferiority, open-label clinical study of 549 patients, conducted to determine that bendamustine (90 mg/m2) in combination with rituximab 375 mg/m2 is non-inferior to CHOP (cycles every 3 weeks of cyclophosphamide 750 mg/m2, doxorubicin 50 mg/m2, and vincristine 1.4 mg/m2 on day 1, and prednisone 100 mg/day for 5 days) plus rituximab 375 mg/m2. Rituximab was administered in both treatment arms on day 1 of each cycle. Treatment was administered for a maximum of 6 cycles. Baseline demographics and patient characteristics are summarized in Table 11.
Patients were stratified by histological lymphoma subtype, then randomly assigned according to a pre-specified randomisation list to receive either intravenous bendamustine (90 mg/m2 on days 1 and 2 of a 4-week cycle) or CHOP (cycles every 3 weeks of cyclophosphamide 750 mg/m2, doxorubicin 50 mg/m2, and vincristine 1.4 mg/m2 on day 1, and prednisone 100 mg/day for 5 days) for a maximum of six cycles. Patients in both groups received rituximab 375 mg/m2 on day 1 of each cycle. Patients aged 18 years or older with a WHO performance status of 2 or less were eligible if they had newly diagnosed stage III or IV indolent or mantle-cell lymphoma. Patients and treating physicians were not masked to treatment allocation. The primary endpoint was progression-free survival, with a non-inferiority margin of 10%.
 At median follow-up of 45 months (IQR 25-57), median progression-free survival was significantly longer in the bendamustine plus rituximab group than in the R-CHOP group (69.5 months [26.1 to not yet reached] vs. 31.2 months [15.2-65.7]; hazard ratio 0.58, 95% CI 0.44-0.74; p < 0.0001).
At median follow-up of 45 months (IQR 25-57), median progression-free survival was significantly longer in the bendamustine plus rituximab group than in the R-CHOP group (69.5 months [26.1 to not yet reached] vs. 31.2 months [15.2-65.7]; hazard ratio 0.58, 95% CI 0.44-0.74; p < 0.0001).
A significant benefit for progression-free survival was shown with B-R vs. R-CHOP for all histological subtypes except for marginal-zone lymphoma (see Figure 2).
 The improvement in progression-free survival with B-R was independent of age, concentration of lactate dehydrogenase (LDH), and FLIPI score (see Table 12). Overall survival did not differ between the two treatment groups.
The improvement in progression-free survival with B-R was independent of age, concentration of lactate dehydrogenase (LDH), and FLIPI score (see Table 12). Overall survival did not differ between the two treatment groups.
The rate of overall response did not differ between the treatment groups (93% for B-R vs. 91% for R-CHOP); however the rate of complete response was significantly increased in patients in the B-R group (104 [40%] vs. 76 [30%]; p=0.021).

Relapsed/refractory NHL.
The efficacy of bendamustine was evaluated in a single arm study (SDX-105-03) of 100 patients with indolent B-cell NHL that had progressed during or within six months of treatment with rituximab or a rituximab-containing regimen. Patients were included if they relapsed within 6 months of either the first dose (monotherapy) or last dose (maintenance regimen or combination therapy) of rituximab. All patients received bendamustine intravenously at a dose of 120 mg/m2, on Days 1 and 2 of a 21-day treatment cycle. Patients were treated for up to 8 cycles.
The median age was 60 years, 65% were male, and 95% had a baseline WHO performance status of 0 or 1. Major tumour subtypes were follicular lymphoma (62%), diffuse small lymphocytic lymphoma (21%), and marginal zone lymphoma (16%). Ninety-nine percent of patients had received previous chemotherapy, 91% of patients had received previous alkylator therapy, and 97% of patients had relapsed within 6 months of either the first dose (monotherapy) or last dose (maintenance regimen or combination therapy) of rituximab.
Efficacy was based on the assessments by a blinded independent review committee (IRC) and included overall response rate (complete response + complete response unconfirmed + partial response) and duration of response (DR) as summarized in Table 13.
 Progression-free survival (PFS), a secondary endpoint in this study, was comparable across all patient groups defined by baseline characteristics (see Table 14). The median PFS was 72 weeks in patients without previous alkylator therapy, and 51 weeks in patients who were sensitive to the previous alkylator therapy or chemotherapy. In the patients who had received previous radio-immunotherapy, the PFS was 53 weeks. Disease characteristics at baseline (FLIPI risk category, number of lymph nodal sites, or bulky disease) did not markedly affect duration of PFS.
Progression-free survival (PFS), a secondary endpoint in this study, was comparable across all patient groups defined by baseline characteristics (see Table 14). The median PFS was 72 weeks in patients without previous alkylator therapy, and 51 weeks in patients who were sensitive to the previous alkylator therapy or chemotherapy. In the patients who had received previous radio-immunotherapy, the PFS was 53 weeks. Disease characteristics at baseline (FLIPI risk category, number of lymph nodal sites, or bulky disease) did not markedly affect duration of PFS.

5.2 Pharmacokinetic Properties
Absorption.
Peak plasma concentrations of bendamustine occurred typically at the completion of 30 or 60 minute infusions, after which the drug was rapidly cleared from the plasma. After a single‐dose of bendamustine 120 mg/m2 infused over 60 minutes in a clinical study in patients with indolent NHL refractory to rituximab, the drug was eliminated from the plasma in a generally tri‐phasic manner characterized by an initial rapid distribution phase, a slower secondary phase, and a longer terminal phase. In this study, the mean ± SD Cmax was 5.6 ± 2.4 microgram/mL and the mean ± SD AUCinf was 7.2 ± 3.8 microgram.hr/mL. There were no formal dose proportionality studies in humans with bendamustine administered by I.V. infusion.
Distribution.
Following 30 min I.V. infusion the central volume of distribution was 19.3 L. Under steady-state conditions following I.V. bolus injection the volume of distribution was 15.8-20.5 L.
More than 95% of the substance is bound to plasma proteins (primarily albumin).
Metabolism.
A major route of clearance of bendamustine is the hydrolysis to monohydroxy- and dihydroxy-bendamustine. Formation of N-desmethyl-bendamustine and gamma-hydroxy-bendamustine by hepatic metabolism involves cytochrome P450 (CYP) 1A2 isoenzyme. Another major route of bendamustine metabolism involves conjugation with glutathione.
In vitro bendamustine does not inhibit CYP 1A2, CYP 2C9/10, CYP 2D6, CYP 2E1 and CYP 3A4.
Excretion.
The elimination half-life t1/2β after 30 min I.V. infusion of 120 mg/m2 to 12 subjects was 28.2 minutes. The mean total clearance after 30 min I.V. infusion of 120 mg/m2 body surface area to 12 subjects was 639.4 mL/minute. About 20% of the administered dose was recovered in urine within 24 hours. Amounts excreted in urine were in the order monohydroxy-bendamustine > bendamustine > dihydroxy-bendamustine > oxidised metabolite > N-desmethyl bendamustine. In the bile, primarily polar metabolites are eliminated. Mean recovery of total radioactivity in cancer patients following intravenous infusion of [14C]-bendamustine hydrochloride was approximately 76% of the radiochemical dose when collected up to day 8 (168 hrs post-dose). Approximately half (45.5%) of the dose was recovered in the urine and approximately a quarter (25.2%) of the dose was recovered in the faeces.
Urinary excretion was confirmed as a relatively minor pathway of elimination of unmodified bendamustine, with only approximately 3.3% of the dose recovered in the urine as the parent compound. Less than 1% of the dose was recovered in the urine as M3 and M4, and less than 5% of the dose was recovered in the urine as HP2.
Renal impairment.
In patients with creatinine clearance > 10 mL/min including dialysis dependent patients, no significant difference to patients with normal liver and kidney function was observed with respect to Cmax, tmax, AUC, t1/2β, volume of distribution and clearance.
Hepatic impairment.
In patients with 30 - 70% tumour involvement of the liver and mild hepatic impairment (serum bilirubin < 1.2 mg/dL) the pharmacokinetic behaviour was not changed. There was no significant difference to patients with normal liver and kidney function with respect to Cmax, tmax, AUC, t1/2β, volume of distribution and clearance. AUC and total body clearance of bendamustine correlate inversely with serum bilirubin. Patients with moderate to severe hepatic impairment, whether measured by tumour involvement and serum bilirubin or Child-Pugh classification have not been assessed.
Elderly subjects.
Subjects up to 84 years of age [< 65 yrs (n=30); 65-75yrs (n=14) and > 75 yrs (n=5)] were included in pharmacokinetic studies. Higher age does not influence the pharmacokinetics of bendamustine.
5.3 Preclinical Safety Data
Genotoxicity.
Bendamustine was mutagenic in the bacterial mutation assay and clastogenic in in vitro (human lymphocytes) and in vivo (rat micronucleus test) studies.
Carcinogenicity.
After 4 daily intra-peritoneal or oral doses of bendamustine to female mice, drug-related tumours appeared during the life-time follow-up period-peritoneal sarcoma in mice that received intra-peritoneal doses (≥ 50 mg/kg/day or 150 mg/m2), and pulmonary adenomas and mammary carcinomas in mice that received oral doses (250 mg/kg/day).6 Pharmaceutical Particulars
6.1 List of Excipients
Mannitol.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
For information about interactions with other medicines and other forms of interaction, see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Unopened vials.
Store below 25°C. Keep the container in the outer carton in order to protect from light.
The powder should be reconstituted immediately after opening of the vial. The reconstituted concentrate should be diluted immediately with 0.9% sodium chloride solution.
Reconstituted solution.
To reduce microbiological hazard, use as soon as possible after reconstitution/preparation. If storage is necessary, hold at not more than 3.5 hours at 25°C or at 2-8°C for not more than 24 hours. From a microbiological point of view, the solution should be used immediately.
6.5 Nature and Contents of Container
Bendamustine Sandoz is supplied in Type I amber glass vials of 25 mL or 50 mL with rubber stopper and an aluminium cap with flip-top.
25 mL vials contain 25 mg bendamustine hydrochloride; supplied in cartons containing 1 vial.
50 mL vials contain 100 mg bendamustine hydrochloride; supplied in cartons containing 1 vial.
The vials are for single use only.
Not all presentations may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
For information about special precautions for disposal and handling, see Section 4.2 Dose and Method of Administration, Special precautions for disposal and handling.
6.7 Physicochemical Properties
Bendamustine Sandoz contains bendamustine hydrochloride (as monohydrate), an alkylating drug, as the active ingredient. Bendamustine hydrochloride contains a mechlorethamine group and a benzimidazole heterocyclic ring with a butyric acid substituent.
Chemical structure.
 Chemical formula: 1H-benzimidazole-2-butanoic acid, 5-[bis(2-chloroethyl)amino]-1-methyl-, monohydrochloride, monohydrate.
Chemical formula: 1H-benzimidazole-2-butanoic acid, 5-[bis(2-chloroethyl)amino]-1-methyl-, monohydrochloride, monohydrate.
Molecular formula: C16H21Cl2N3O2.HCl.H2O.
Molecular weight: 412.74.
CAS number.
3543-75-7.7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Summary Table of Changes


 A total of 50 patients had 60 serious adverse events. Most frequently occurring serious adverse events in the bendamustine group were hypersensitivity and pneumonia (each with 3 patients) and anaemia, vomiting, pyrexia and tumour lysis syndrome (each with 2 patients). Most frequent documented serious adverse event in the chlorambucil group was herpes zoster (with 2 patients). All other events were documented only once by patient. See Table 2.
A total of 50 patients had 60 serious adverse events. Most frequently occurring serious adverse events in the bendamustine group were hypersensitivity and pneumonia (each with 3 patients) and anaemia, vomiting, pyrexia and tumour lysis syndrome (each with 2 patients). Most frequent documented serious adverse event in the chlorambucil group was herpes zoster (with 2 patients). All other events were documented only once by patient. See Table 2. Number of adverse events possible, probable or definite related to the study medication (including missing relationship) was higher in the bendamustine arm than in the chlorambucil arm. Especially blood and lymphatic system disorders, gastrointestinal disorders and pyrexia occurred more frequently under bendamustine than under chlorambucil. See Table 3.
Number of adverse events possible, probable or definite related to the study medication (including missing relationship) was higher in the bendamustine arm than in the chlorambucil arm. Especially blood and lymphatic system disorders, gastrointestinal disorders and pyrexia occurred more frequently under bendamustine than under chlorambucil. See Table 3. The most frequent adverse reactions leading to study withdrawal for patients receiving bendamustine were hypersensitivity (2%) and pyrexia (1%).
The most frequent adverse reactions leading to study withdrawal for patients receiving bendamustine were hypersensitivity (2%) and pyrexia (1%).

 The data described below reflect exposure to bendamustine in 176 patients with indolent B-cell NHL treated in two single-arm studies (SDX-105-03 and SDX-105-01).
The data described below reflect exposure to bendamustine in 176 patients with indolent B-cell NHL treated in two single-arm studies (SDX-105-03 and SDX-105-01). Haematologic toxicities, based on laboratory values and CTC grade, in NHL patients treated in both single arm studies combined are described in Table 8. Clinically important chemistry laboratory values that were new or worsened from baseline and occurred in > 1% of patients at Grade 3 or 4, in NHL patients treated in both single arm studies combined were hyperglycaemia (3%), elevated creatinine (2%), hyponatraemia (2%), and hypocalcaemia (2%).
Haematologic toxicities, based on laboratory values and CTC grade, in NHL patients treated in both single arm studies combined are described in Table 8. Clinically important chemistry laboratory values that were new or worsened from baseline and occurred in > 1% of patients at Grade 3 or 4, in NHL patients treated in both single arm studies combined were hyperglycaemia (3%), elevated creatinine (2%), hyponatraemia (2%), and hypocalcaemia (2%). In both studies, serious adverse reactions, regardless of causality, were reported in 37% of patients receiving bendamustine. The most common serious adverse reactions occurring in 5% of patients were febrile neutropaenia and pneumonia. Other important serious adverse reactions reported in clinical trials and/or postmarketing experience were acute renal failure, cardiac failure, hypersensitivity, skin reactions, pulmonary fibrosis, pneumonitis, pulmonary alveolar haemorrhage, and myelodysplastic syndrome.
In both studies, serious adverse reactions, regardless of causality, were reported in 37% of patients receiving bendamustine. The most common serious adverse reactions occurring in 5% of patients were febrile neutropaenia and pneumonia. Other important serious adverse reactions reported in clinical trials and/or postmarketing experience were acute renal failure, cardiac failure, hypersensitivity, skin reactions, pulmonary fibrosis, pneumonitis, pulmonary alveolar haemorrhage, and myelodysplastic syndrome. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: anaphylaxis; and injection or infusion site reactions including phlebitis, pruritus, irritation, pain, and swelling. Increases in alanine aminotransferase; aspartate aminotransferase; blood bilirubin and blood urea levels have been reported. Somnolence, atrial fibrillations and palpitations have also been reported.
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: anaphylaxis; and injection or infusion site reactions including phlebitis, pruritus, irritation, pain, and swelling. Increases in alanine aminotransferase; aspartate aminotransferase; blood bilirubin and blood urea levels have been reported. Somnolence, atrial fibrillations and palpitations have also been reported. Complete response is lower than noted in Table 10 - for both bendamustine and chlorambucil - if there is a requirement for imaging to confirm lymph nodes ≤ 1.5 cm.
Complete response is lower than noted in Table 10 - for both bendamustine and chlorambucil - if there is a requirement for imaging to confirm lymph nodes ≤ 1.5 cm.
 At median follow-up of 45 months (IQR 25-57), median progression-free survival was significantly longer in the bendamustine plus rituximab group than in the R-CHOP group (69.5 months [26.1 to not yet reached] vs. 31.2 months [15.2-65.7]; hazard ratio 0.58, 95% CI 0.44-0.74; p < 0.0001).
At median follow-up of 45 months (IQR 25-57), median progression-free survival was significantly longer in the bendamustine plus rituximab group than in the R-CHOP group (69.5 months [26.1 to not yet reached] vs. 31.2 months [15.2-65.7]; hazard ratio 0.58, 95% CI 0.44-0.74; p < 0.0001). The improvement in progression-free survival with B-R was independent of age, concentration of lactate dehydrogenase (LDH), and FLIPI score (see Table 12). Overall survival did not differ between the two treatment groups.
The improvement in progression-free survival with B-R was independent of age, concentration of lactate dehydrogenase (LDH), and FLIPI score (see Table 12). Overall survival did not differ between the two treatment groups.
 Progression-free survival (PFS), a secondary endpoint in this study, was comparable across all patient groups defined by baseline characteristics (see Table 14). The median PFS was 72 weeks in patients without previous alkylator therapy, and 51 weeks in patients who were sensitive to the previous alkylator therapy or chemotherapy. In the patients who had received previous radio-immunotherapy, the PFS was 53 weeks. Disease characteristics at baseline (FLIPI risk category, number of lymph nodal sites, or bulky disease) did not markedly affect duration of PFS.
Progression-free survival (PFS), a secondary endpoint in this study, was comparable across all patient groups defined by baseline characteristics (see Table 14). The median PFS was 72 weeks in patients without previous alkylator therapy, and 51 weeks in patients who were sensitive to the previous alkylator therapy or chemotherapy. In the patients who had received previous radio-immunotherapy, the PFS was 53 weeks. Disease characteristics at baseline (FLIPI risk category, number of lymph nodal sites, or bulky disease) did not markedly affect duration of PFS.
 Chemical formula: 1H-benzimidazole-2-butanoic acid, 5-[bis(2-chloroethyl)amino]-1-methyl-, monohydrochloride, monohydrate.
Chemical formula: 1H-benzimidazole-2-butanoic acid, 5-[bis(2-chloroethyl)amino]-1-methyl-, monohydrochloride, monohydrate.