1 Name of Medicine
Bimekizumab.
2 Qualitative and Quantitative Composition
Bimzelx 160 mg solution for injection.
Each pre-filled syringe or pen contains 160 mg bimekizumab in 1 mL.
Bimzelx 320 mg solution for injection.
Each pre-filled syringe or pen contains 320 mg bimekizumab in 2 mL.
Bimekizumab is a recombinant humanized full-length monoclonal antibody of the IgG1 sub-class, expressed in a genetically engineered Chinese hamster ovary cell line.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Solution for injection.
The solution is clear to slightly opalescent and colourless to pale brownish-yellow.
4.1 Therapeutic Indications
Plaque psoriasis.
Bimzelx is indicated for the treatment of moderate to severe plaque psoriasis in adult patients who are candidates for systemic therapy or phototherapy.
Psoriatic arthritis.
Bimzelx is indicated for the treatment of adult patients with active psoriatic arthritis who have had an inadequate response to or who have been intolerant to previous disease modifying antirheumatic drug (DMARD) therapy.
Axial spondyloarthritis.
Non-radiographic axial spondyloarthritis (nr-axSpA).
Bimzelx is indicated for the treatment of adult patients with active non-radiographic axial spondyloarthritis with objective signs of inflammation as indicated by elevated C reactive protein (CRP) and/or magnetic resonance imaging (MRI) who have had an inadequate response to or are intolerant to nonsteroidal anti-inflammatory drugs (NSAIDs).
Ankylosing spondylitis (AS, radiographic axial spondyloarthritis).
Bimzelx is indicated for the treatment of adult patients with active ankylosing spondylitis who have responded inadequately or are intolerant to conventional therapy.
Hidradenitis suppurativa (HS).
Bimzelx is indicated for the treatment of adult patients with moderate to severe hidradenitis suppurativa with an inadequate response to conventional systemic HS therapy.4.2 Dose and Method of Administration
The Instructions for Use included as a pack insert must be followed carefully.
Plaque psoriasis.
The recommended dose of Bimzelx for adult patients with plaque psoriasis is 320 mg (given as 2 subcutaneous injections of 160 mg or 1 subcutaneous injection of 320 mg) at Week 0, 4, 8, 12, 16 and every 8 weeks thereafter.
Psoriatic arthritis.
The recommended dose of Bimzelx for adult patients with active psoriatic arthritis is 160 mg (given as one subcutaneous injection of 160 mg) every 4 weeks.
For psoriatic arthritis patients with coexistent moderate to severe plaque psoriasis, the use of the dose applicable to plaque psoriasis may be considered [i.e. 320 mg (given as 2 subcutaneous injections of 160 mg each or 1 subcutaneous injection of 320 mg) at Week 0, 4, 8, 12, 16 and every 8 weeks thereafter]. After 16 weeks, regular assessment of efficacy is recommended and if a sufficient clinical response in joints cannot be maintained, a switch to 160 mg every 4 weeks can be considered.
Axial spondyloarthritis (nr-axSpA and AS).
The recommended dose of Bimzelx for adult patients with active axial spondyloarthritis is 160 mg (given as one subcutaneous injection of 160 mg) every 4 weeks.
Hidradenitis suppurativa.
The recommended dose of Bimzelx for adult patients with hidradenitis suppurativa is 320 mg (given as 2 subcutaneous injections of 160 mg or 1 subcutaneous injection of 320 mg) every 4 weeks.
If more rapid disease control is desired and after consideration of the individual patient risks and benefits a Bimzelx dose for adult patients with hidradenitis suppurativa of 320 mg (given as 2 subcutaneous injections of 160 mg or 1 subcutaneous injection of 320 mg) every 2 weeks up to 16 weeks and every 4 weeks thereafter may be considered.
Method of administration.
Bimzelx is administered by subcutaneous injection. A 320 mg dose can be given as 2 subcutaneous injections of 160 mg or 1 subcutaneous injection of 320 mg. Suitable areas for injection include thigh, abdomen and upper arm. Injection sites should be rotated and injections should not be given into psoriasis plaques or areas where the skin is tender, bruised, erythematous, or indurated.
Bimzelx is for single use in one patient only. Discard any residue.
Special populations.
Overweight patients (plaque psoriasis).
For some patients with plaque psoriasis (including psoriatic arthritis with coexistent moderate to severe psoriasis) and a body weight ≥ 120 kg, 320 mg every 4 weeks after Week 16 may be considered (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
Elderly population.
No dose adjustment is required (see Section 5.2 Pharmacokinetic Properties).
Renal and hepatic impairment.
Bimzelx has not been studied in these patient populations. Dose adjustments are not considered necessary based on pharmacokinetics (see Section 5.2 Pharmacokinetic Properties). No specific PK studies using Bimzelx have been pursued in patients with underlying impaired hepatic function.
Paediatric population.
The safety and efficacy of Bimzelx in children and adolescents below the age of 18 years has not been established. No data are available.4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients (see Section 6.1 List of Excipients).
Live vaccines should not be given in patients treated with Bimzelx. No data are available on the response to live vaccines.
4.4 Special Warnings and Precautions for Use
Infections.
Bimzelx increases the risk of infections such as upper respiratory tract infections and mucocutaneous candidiasis (mostly oral, less frequent vulvovaginal, oesophageal) (see Section 4.8 Adverse Effects (Undesirable Effects)).
Caution should be exercised when considering the use of Bimzelx in patients with a chronic infection or a history of recurrent infection. Treatment with Bimzelx should not be initiated in patients with any clinically important active infection until the infection resolves or is adequately treated.
Patients treated with Bimzelx should be instructed to seek medical advice if signs or symptoms of clinically important chronic or acute infection and/or fungal infection occur. If a patient develops a clinically important infection or is not responding to standard therapy, the patient should be closely monitored and Bimzelx should not be administered until the infection resolves.
Pre-treatment evaluation for tuberculosis (TB).
No increased susceptibility to tuberculosis was reported from clinical studies. Prior to initiating treatment with Bimzelx, patients should be evaluated for TB infection. Bimzelx should not be given in patients with active TB. Patients receiving Bimzelx should be monitored for signs and symptoms of active TB. Anti-TB therapy should be considered prior to initiating Bimzelx in patients with a past history of latent or active TB in whom an adequate course of treatment cannot be confirmed.
Inflammatory bowel disease.
Cases of new onset or exacerbations of inflammatory bowel disease have been reported during bimekizumab treatment. Caution should be exercised when prescribing Bimzelx to patients with inflammatory bowel disease. Patients should be monitored closely.
Hypersensitivity.
If a serious hypersensitivity reaction occurs, administration of Bimzelx should be discontinued immediately and appropriate therapy initiated.
Vaccinations.
Prior to initiating therapy with Bimzelx, consider completion of all appropriate immunizations according to current immunization guidelines.
Patients treated with Bimzelx may receive inactivated or non-live vaccinations. Healthy individuals who received a single 320 mg dose of Bimzelx two weeks prior to vaccination with an inactivated seasonal influenza vaccine had similar antibody responses compared to individuals who did not receive Bimzelx prior to vaccination.
Use in hepatic impairment.
Bimzelx has not been studied in these patient populations. Dose adjustments are not considered necessary based on pharmacokinetics (see Section 5.2 Pharmacokinetic Properties).
Use in renal impairment.
Bimzelx has not been studied in these patient populations. Dose adjustments are not considered necessary based on pharmacokinetics (see Section 5.2 Pharmacokinetic Properties).
Use in the elderly.
Based on population pharmacokinetic analysis with a limited number of elderly patients (n = 110 for age ≥ 65 years and n = 14 for age ≥ 75 years), apparent clearance (CL/F) in elderly patients and patients less than 65 years of age was similar. No dose adjustment is required.
Paediatric use.
The safety and efficacy of Bimzelx in children and adolescents below the age of 18 years has not be established. No data are available.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
No CYP450 interaction studies have been performed in humans. There is no direct evidence for the role of IL-17A or IL-17F in the expression of CYP450 enzymes. Given that (1) bimekizumab, as an IgG1K mAb, is expected to be degraded into small peptides and amino acids via catabolic pathways in the same manner as endogenous IgG and (2) formation of some CYP450 enzymes which is suppressed by elevated levels of cytokines during inflammation (as in psoriasis), will be reversed by inflammatory suppressors, like IL-17A and IL-17F inhibitor bimekizumab, the resultant outcome will be a normalisation of CYP450 levels/activity. Extrapolation of the latter means that drugs metabolized by the CYP450 system may be co-administered with bimekizumab. However, monitoring of therapeutic plasma level and clinical effect of drugs with narrow therapeutic index (e.g. warfarin) metabolized via CYP450 system should be considered.
Population pharmacokinetic (PK) data analyses indicated that concomitant administration of conventional disease modifying antirheumatic drugs (cDMARDs) including methotrexate or prior exposure to biologic have no clinically relevant impact on the clearance of bimekizumab.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
The effect of bimekizumab on human fertility has not been evaluated. In animal studies, bimekizumab did not indicate harmful effects with respect to fertility as assessed by a lack of effects on reproductive organs, menstrual cycles or sperm in sexually mature cynomolgus monkeys that received bimekizumab for 26 weeks at a weekly SC dose of 200 mg/kg (dose resulting in 84 times the human exposure at 320 mg every 2 weeks based on AUC). The monkeys were not mated to evaluate functional fertility.
(Category C)
There are no adequate and well controlled studies in pregnant women to establish the safety of Bimzelx during pregnancy. Based on the mechanism of action of bimekizumab, the theoretical risk that use during pregnancy may affect neonatal immunity cannot be excluded. In an enhanced pre/postnatal development study in the cynomolgus monkey, bimekizumab showed no effects on gestation, parturition, infant survival, fetal or postnatal development when administered throughout organogenesis until parturition at a maternal dose of 50 mg/kg SC weekly resulting in 12 times the human exposure at 320 mg every 2 weeks based on AUC. At birth, serum bimekizumab concentrations in infant monkeys were comparable to those of mothers indicating placental transfer of bimekizumab. Bimzelx should be used in pregnancy only if the benefits clearly outweigh the potential risks.
It is not known whether bimekizumab is excreted in human milk or absorbed systemically after ingestion. As immunoglobulins can be excreted in human milk, caution should be exercised when Bimzelx is administered to a woman who is breastfeeding and a decision on whether to discontinue breastfeeding during treatment should be made.4.7 Effects on Ability to Drive and Use Machines
Bimekizumab is not anticipated to have any influence on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
A total of 5862 patients have been treated with bimekizumab in blinded and open-label clinical studies in plaque psoriasis, psoriatic arthritis, axial spondyloarthritis (nr-axSpA and AS) and hidradenitis suppurativa representing 11468.7 patient-years of exposure. Of these, over 4660 patients were exposed to bimekizumab for at least one year.
Overall, the safety profile of bimekizumab is consistent across all indications.
Plaque psoriasis.
The most frequently reported adverse drug reactions (ADRs) were upper respiratory tract infections (most frequently nasopharyngitis) and oral candidiasis. See Table 1.

Psoriatic arthritis.
See Table 2 and Table 3.


Axial spondyloarthritis (axSpA).
See Table 4 and Table 5.


Hidradenitis suppurativa.
See Table 6.
 Adverse drug reactions occurring in greater than 1% of subjects on Bimzelx and at a higher rate than in the placebo group through week 16 in the HS clinical trials that are not listed in the table above are constipation, asthenia, otitis externa, vulvovaginal mycotic infection, tinea pedis, myalgia, acne, seborrhoeic dermatitis, urticaria.
Adverse drug reactions occurring in greater than 1% of subjects on Bimzelx and at a higher rate than in the placebo group through week 16 in the HS clinical trials that are not listed in the table above are constipation, asthenia, otitis externa, vulvovaginal mycotic infection, tinea pedis, myalgia, acne, seborrhoeic dermatitis, urticaria.
Other adverse reactions (< 1%).
Adverse drug reactions that occurred in < 1% but > 0.1% of subjects in the Bimzelx group and at a higher rate than in the placebo group through Week 16 were, conjunctivitis, tinea infection, herpes simplex, dyshidrotic eczema, intertrigo, dermatitis, otitis media, cutaneous and other mucosal candidiasis (including oesophageal).
Description of selected adverse reactions.
Infections. In the placebo-controlled period of Phase III clinical studies in plaque psoriasis, infections were reported in 36.0% of patients treated with bimekizumab for up to 16 weeks compared with 22.5% of patients treated with placebo. The majority of infections consisted of non-serious mild to moderate upper respiratory tract infections such as nasopharyngitis. There were higher rates of oral and oropharyngeal candidiasis in patients treated with bimekizumab consistent with the mechanism of action (7.3% and 1.2% respectively compared to 0% for placebo-treated patients). In the PS0015 study over the 48-week comparator controlled period, a higher incidence rate of oral candidiasis was observed in BKZ (19.3%) compared to secukinumab (3.0%) in PSO patients. The vast majority of cases were non-serious, mild or moderate in severity, and did not require treatment discontinuation.
Serious infections occurred in 0.3% of patients treated with bimekizumab and 0% treated with placebo.
Over the entire treatment period of Phase III studies in plaque psoriasis, infections were reported in 63.2% of patients treated with bimekizumab (120.4 per 100 patient-years). Serious infections were reported in 1.5% of patients treated with bimekizumab (1.6 per 100 patient-years) (see Section 4.4).
Infection rates observed in PsA, axSpA (nr-axSpA and AS) and HS Phase III clinical studies were similar to those observed in plaque psoriasis.
In the placebo-controlled period, oral and oropharyngeal candidiasis rates in patients treated with bimekizumab were 2.3% and 0% respectively in PsA, 3.7% and 0.3% respectively in axSpA and 7.1% and 0% respectively in HS compared to 0% with placebo.
Neutropenia. Neutropenia was observed with bimekizumab in phase III clinical studies in plaque psoriasis. In the 16 weeks placebo-controlled period neutropenia grade 3/4 were observed at the same frequency of 0.6% in patients receiving bimekizumab or placebo. Over the entire treatment period of Phase III studies, neutropenia grade 3/4 were observed in 1% of patients treated with bimekizumab.
The frequency of neutropenia in PsA, axSpA (nr-axSpA and AS) and HS clinical studies was similar to that observed in plaque psoriasis studies.
Most cases of neutropenia were transient and did not require treatment discontinuation. No serious infections were associated with neutropenia.
Hepatic transaminases. Psoriatic arthritis.
During the placebo-controlled period, increased incidence of elevated hepatic transaminases was observed in psoriatic arthritis patients treated with bimekizumab compared to placebo.
Increase > 3 x upper limit of normal (ULN) and > 5 x ULN in hepatic transaminase levels were observed in the bimekizumab group (> 3-5 x ULN ALT: 0.9%, AST: 0.3%; > 5 x ULN ALT: 0.1%, AST: 0.4%), while no elevated ALT and AST was seen in the placebo group.
Elevations (mainly > 3 x ULN) in ALT and AST were also observed during the long-term treatment with bimekizumab.
The majority of ALT or AST elevations had confounding factors or alternative explanations, were transient and resolved.
Axial spondyloarthritis.
During the placebo-controlled period, hepatic transaminases elevations were observed in axial spondyloarthritis patients at a similar uncommon incidence in the bimekizumab and placebo group (> 3-5 x ULN ALT: 0.3% vs. 0.8%, AST: 0.9% vs. 0.4%; > 5 x ULN ALT: 0.6% vs. 0.4%, AST: 0.6% vs. 0.4%, respectively in the bimekizumab and placebo group).
Elevations (mainly > 3 x ULN) in ALT and AST were also observed during the long-term treatment with bimekizumab.
The majority of ALT or AST elevations had confounding factors or alternative explanations, were transient and resolved.
Hidradenitis suppurativa.
During the placebo-controlled period, increased incidence of elevated hepatic transaminases was observed in hidradenitis suppurativa patients treated with bimekizumab compared to placebo.
Increases > 3 x ULN and > 5 x ULN in hepatic transaminase levels were observed in the bimekizumab 320 mg Q2W group (> 3-5 x ULN ALT: 0.3%, AST: 0.9%; > 5 x ULN ALT: 0.2%, AST: 0.5%), and in the bimekizumab 320 mg Q4W group (> 3-5 x ULN ALT:0%, AST:0.7%; > 5 x ULN ALT or AST: 0%) while no > 3 x ULN elevations in ALT and AST were seen in the placebo group.
Elevations (mainly > 3 x ULN) in AST and ALT were also observed during long-term treatment with bimekizumab.
The majority of AST or ALT elevations had confounding factors or alternative explanations, were transient and resolved.
One study participant with history of hepatic enzyme increased of unknown etiology experienced a > 10 x ULN rise in ALT and AST 14 days after starting bimekizumab 320 mg Q2W. AST, ALT and GGT started to rise within 28 days prior to administration of the first dose. The event, reported as "potential DILI", led to study discontinuation as per protocol withdrawal criteria and resolved in < 1 month.
Immunogenicity. As with all therapeutic proteins, there is the potential for immunogenicity with bimekizumab. The detection of anti-drug antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of anti-drug antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors, including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to bimekizumab with the incidence of antibodies to other products may be misleading.
Plaque psoriasis.
Approximately 45% of plaque psoriasis patients treated with bimekizumab up to 56 weeks at the recommended dosing regimen (320 mg every 4 weeks up to Week 16 and 320 mg every 8 weeks thereafter) developed anti-drug antibodies. Of the patients who developed anti-drug antibodies, approximately 34% (16% of all patients treated with bimekizumab) had antibodies that were classified as neutralizing.
Psoriatic arthritis.
Approximately 31% of patients with psoriatic arthritis treated with bimekizumab at the recommended dosing regimen (160 mg every 4 weeks) up to 16 weeks had anti-drug antibodies. Of the patients with anti-drug antibodies, about 33% (10% of all patients treated with bimekizumab) had antibodies that were classified as neutralizing. By Week 52, approximately 47% of patients with PsA in the BE OPTIMAL study treated with bimekizumab at the recommended dosing regimen (160 mg every 4 weeks) had anti-drug antibodies. Of the patients in the BE OPTIMAL study with anti-drug antibodies, about 38% (18% of all patients treated with bimekizumab) had antibodies that were classified as neutralizing.
Axial spondyloarthritis.
nr-axSpA.
Approximately 57% of patients with nr-axSpA treated with bimekizumab up to 52 weeks at the recommended dosing regimen (160 mg every 4 weeks) had anti-drug antibodies. Of the patients with anti-drug antibodies, approximately 44% (25% of all patients treated with bimekizumab) had antibodies that were classified as neutralizing.
AS.
Approximately 44% of patients with AS treated with bimekizumab up to 52 weeks at the recommended dosing regimen (160 mg every 4 weeks) had anti-drug antibodies. Of the patients with anti-drug antibodies, approximately 44% (20% of all patients treated with bimekizumab) had antibodies that were classified as neutralizing.
Hidradenitis suppurativa.
Approximately 54% of HS patients treated with bimekizumab up to 48 weeks at the recommended dosing regimen (320 mg every 2 weeks up to Week 16 and 320 mg every 4 weeks thereafter) had or developed anti-drug antibodies. Of the patients who had or developed anti-drug antibodies, approximately 63% (37% of all patients treated with bimekizumab 320 mg every 2 weeks up to Week 16 and 320 mg every 4 weeks thereafter) had antibodies that were classified as neutralizing. Across all indications, no clinically meaningful impact on clinical response or safety profile was associated with anti-bimekizumab antibodies development.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at http://www.tga.gov.au/reporting-problems.4.9 Overdose
Single doses of 640 mg intravenously or 640 mg subcutaneously, followed by 320 mg subcutaneously every two weeks for five doses have been administered in clinical studies without dose-limiting toxicity. In the event of overdose, it is recommended that the patient be monitored for any signs and symptoms of adverse reactions and appropriate symptomatic treatment be instituted immediately.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Bimekizumab is a humanised IgG1/κ monoclonal antibody with two identical antigen binding regions that bind and neutralise IL-17A, IL-17F and IL-17AF cytokines. Levels of IL-17A and IL-17F are elevated in several immune mediated inflammatory diseases and drive chronic inflammation and damage across multiple tissues. IL-17A and IL-17F cooperate and/or synergise with other inflammatory cytokines to induce inflammation. IL-17F is produced in significant amounts by innate immune cells. This production can be independent of IL-23. In lesional HS tissue, both IL-17A and IL-17F are highly expressed compared to non-HS tissues. In human in vitro models, dual neutralisation of both IL-17A and IL-17F with bimekizumab suppresses the expression of inflammation related genes and proteins, inhibits the migration of inflammatory cells and pathological osteogenesis to a greater extent than inhibition of IL-17A alone.
Pharmacodynamic effects.
No formal pharmacodynamic (PD) studies have been conducted with bimekizumab.
Clinical trials.
Psoriasis - phase 3 pivotal studies. The safety and efficacy of bimekizumab was evaluated in 1480 patients with moderate to severe plaque psoriasis in three Phase III multicenter, randomized, placebo and/or active comparator controlled studies. Patients were at least 18 years of age, had a Psoriasis Area and Severity Index (PASI) score ≥ 12 and Body Surface Area (BSA) affected by PSO ≥ 10%, an Investigators Global Assessment (IGA) score ≥ 3 on a 5-point scale and were candidates for systemic psoriasis therapy and/or phototherapy. The efficacy and safety of bimekizumab were evaluated versus placebo and ustekinumab (BE VIVID - PS0009), versus placebo (BE READY - PS0013) and versus adalimumab (BE SURE - PS0008).
The BE VIVID study evaluated 567 patients for 52 weeks where patients were randomized to receive either bimekizumab 320 mg every 4 weeks, ustekinumab (45 mg or 90 mg, depending on patient weight, at baseline and Week 4 and then every 12 weeks), or placebo for an initial 16 weeks followed by bimekizumab 320 mg every 4 weeks.
The BE READY study evaluated 435 patients for 56 weeks. Patients were randomized to receive bimekizumab 320 mg every 4 weeks or placebo. At Week 16, patients who achieved a PASI 90 response entered the 40-week randomized withdrawal period. Patients initially randomized to bimekizumab 320 mg every 4 weeks were re-randomized to either bimekizumab 320 mg every 4 weeks or bimekizumab 320 mg every 8 weeks or placebo (i.e. withdrawal of bimekizumab). Patients initially randomized to placebo continued to receive placebo provided they were PASI 90 responders.
Patients who did not achieve a PASI 90 response at Week 16 entered an open-label escape arm and received bimekizumab 320 mg every 4 weeks for 12 weeks. Patients who relapsed (did not achieve PASI 75 response) during the randomized withdrawal period also entered the 12-week escape arm.
The BE SURE study evaluated 478 patients for 56 weeks. Patients were randomized to receive either bimekizumab 320 mg every 4 weeks through Week 56, bimekizumab 320 mg every 4 weeks through Week 16 followed by bimekizumab 320 mg every 8 weeks through Week 56 or adalimumab as per labeling recommendation through Week 24 followed by bimekizumab 320 mg every 4 weeks through Week 56.
Baseline characteristics were consistent across all 3 studies. Among those, the median baseline BSA was 20%, the median baseline PASI score was 18 and the baseline IGA score was severe in 33% of patients. The median baseline scores for Patient Symptoms Diary (PSD) pain, itch and scaling items ranged between 6 and 7 on a 0-10 points scale and the median baseline Dermatology Life Quality Index (DLQI) total score was 9.
Across all 3 studies, 38% of patients had received a prior biologic therapy; 23% had received at least one anti-IL17 agent and 13% had received at least one TNF-antagonist. Twenty-two percent were naïve to any systemic therapy (including non-biologic and biologic) and 39% of patients had received prior phototherapy or chemotherapy.
The efficacy of bimekizumab was evaluated with respect to impact on skin disease overall, specific body locations (scalp, nails and hand and foot), patient reported symptoms and impact on quality of life. The two co-primary end-points in all 3 studies were the proportion of patients who achieved 1) a PASI 90 response and 2) an IGA "clear or almost clear" (IGA 0/1 with at least two points improvement from baseline) response at Week 16. PASI 100, IGA 0 response at Week 16 and PASI 75 response at Week 4 were key secondary endpoints in all 3 studies.
Skin disease overall.
Treatment with bimekizumab resulted in significant improvement in the measures of disease activity compared to placebo, ustekinumab or adalimumab at Week 16. The key efficacy results are shown in Table 7.
 Bimekizumab was associated with a rapid onset of efficacy. In BE VIVID, at Week 2 and Week 4, PASI 90 response rates were significantly higher for bimekizumab-treated patients (12.1% and 43.6% respectively) compared to placebo (1.2% and 2.4% respectively) and ustekinumab (1.2% and 3.1% respectively). See Figure 1.
Bimekizumab was associated with a rapid onset of efficacy. In BE VIVID, at Week 2 and Week 4, PASI 90 response rates were significantly higher for bimekizumab-treated patients (12.1% and 43.6% respectively) compared to placebo (1.2% and 2.4% respectively) and ustekinumab (1.2% and 3.1% respectively). See Figure 1.
 In the BE VIVID study, at Week 52, bimekizumab-treated patients achieved significantly higher response rates than the ustekinumab-treated patients on the endpoints of PASI 90 (81.6% bimekizumab vs 55.8% ustekinumab, p < 0.001), IGA 0/1 (77.9% bimekizumab vs 60.7% ustekinumab, p < 0.001) and PASI 100 (64.2% bimekizumab vs 38.0% ustekinumab).
In the BE VIVID study, at Week 52, bimekizumab-treated patients achieved significantly higher response rates than the ustekinumab-treated patients on the endpoints of PASI 90 (81.6% bimekizumab vs 55.8% ustekinumab, p < 0.001), IGA 0/1 (77.9% bimekizumab vs 60.7% ustekinumab, p < 0.001) and PASI 100 (64.2% bimekizumab vs 38.0% ustekinumab).
In the BE SURE study at Week 24, a significantly higher percentage of patients treated with bimekizumab achieved a PASI 90 and an IGA 0/1 responses as compared with adalimumab (85.6% and 86.5% respectively vs 51.6% and 57.9% respectively, p < 0.001). Among the 65 adalimumab nonresponders at Week 24 (< PASI 90), 78.5% achieved a PASI 90 response after 16 weeks of treatment with bimekizumab. No new safety findings were observed in patients who switched from adalimumab to bimekizumab. At Week 56, 70.2% of bimekizumab-treated patients achieved a PASI 100 response. See Figure 2.
 The efficacy of bimekizumab was demonstrated regardless of age, gender, race, disease duration, body weight, PASI baseline severity and previous treatment with a biologic. Bimekizumab was efficacious in prior biologic exposed patients, including anti-TNF/ anti IL-17 and in systemic treatment-naïve patients.
The efficacy of bimekizumab was demonstrated regardless of age, gender, race, disease duration, body weight, PASI baseline severity and previous treatment with a biologic. Bimekizumab was efficacious in prior biologic exposed patients, including anti-TNF/ anti IL-17 and in systemic treatment-naïve patients.
Based on population PK/ PD analysis in patients with moderate to severe plaque psoriasis, some patients with higher body weight (≥ 120 kg) benefit from Bimzelx 320 mg every four weeks after the initial 16 weeks of treatment.
Across the entire bimekizumab Phase 3 psoriasis program, including 3 pivotal Phase 3 studies (BE SURE, BE VIVID, and BE READY) and the Phase 3b study (BE RADIANT - PS0015), a total of 1362 study participants were randomized to treatment with bimekizumab. Of these, 116 (8.5%) presented with a body weight ≥ 120 kg at Baseline.
A post-hoc analyses of efficacy for the subgroups of study participants weighing < 120 kg or ≥ 120 kg in the Initial Treatment Period (with bimekizumab 320 mg Q4W dosing from Week 0 to Week 16), pooled across all psoriasis Phase 3/3b studies is shown in Table 8.
 Although the number of subjects in the ≥ 120 kg group is low, the data demonstrate that response rates were lower in patients weighing more than 120 kg compared with patients who weighed less and, represents a clinical correlation with the reduction in exposure as predicted by the PK/PD modelling.
Although the number of subjects in the ≥ 120 kg group is low, the data demonstrate that response rates were lower in patients weighing more than 120 kg compared with patients who weighed less and, represents a clinical correlation with the reduction in exposure as predicted by the PK/PD modelling.
The difference with bimekizumab 320 mg Q4W dosing between patients < 120 kg and patients ≥ 120 kg weights, however, is only numerical [9.3% for both PASI 90 and IGA 0/1]. There was no statistical analysis.
Based on the pooled analysis, greater increases in PASI 100 and IGA 0 were seen in the patients ≥ 120 kg beyond Week 16 with Q4W maintenance dosing compared to Q8W maintenance dosing. No statistical comparison was performed. Thus, it can be stated that increasing the dosing frequency in heavier patients has not been shown to result in greater statistically significant efficacy but may be used to increase plasma concentrations of bimekizumab in heavier patients.
Maintenance of response.
See Table 9.

Durability of PASI 90 response (after bimekizumab discontinuation).
See Figure 3.
 In BE READY, for PASI 90 responders at Week 16 who were re-randomized to placebo and withdrawn from bimekizumab, the median time to relapse, defined as loss of PASI 75, was approximately 28 weeks (32 weeks after the last bimekizumab dose). Among these patients, 88.1% regained a PASI 90 response within 12 weeks of restarting treatment with bimekizumab 320 mg every 4 weeks.
In BE READY, for PASI 90 responders at Week 16 who were re-randomized to placebo and withdrawn from bimekizumab, the median time to relapse, defined as loss of PASI 75, was approximately 28 weeks (32 weeks after the last bimekizumab dose). Among these patients, 88.1% regained a PASI 90 response within 12 weeks of restarting treatment with bimekizumab 320 mg every 4 weeks.
Specific body locations.
Significant improvements were observed in psoriasis involving the scalp, nails and hands and feet in patients treated with bimekizumab at Week 16 (see Table 10).
 Scalp IGA and palmoplantar IGA responses were maintained through Week 52/56. Nail psoriasis continued to improve beyond Week 16. In BE VIVID, at Week 52, 60.3% of patients treated with bimekizumab 320 mg every 4 weeks achieved complete nail clearance (mNAPSI 100). In BE READY, at Week 56, 67.7% and 69.8% of Week 16 PASI 90 responders achieved complete nail clearance with bimekizumab 320 mg every 8 weeks and bimekizumab 320 mg every 4 weeks respectively.
Scalp IGA and palmoplantar IGA responses were maintained through Week 52/56. Nail psoriasis continued to improve beyond Week 16. In BE VIVID, at Week 52, 60.3% of patients treated with bimekizumab 320 mg every 4 weeks achieved complete nail clearance (mNAPSI 100). In BE READY, at Week 56, 67.7% and 69.8% of Week 16 PASI 90 responders achieved complete nail clearance with bimekizumab 320 mg every 8 weeks and bimekizumab 320 mg every 4 weeks respectively.
Health-related quality of life/patient reported outcomes.
Across all 3 studies, a greater proportion of patients treated with bimekizumab experienced no impact of psoriasis on their quality of life as measured by the Dermatology Life Quality Index (DLQI) compared to placebo and active comparator-treated patients at Week 16 (Table 11).
 DLQI 0/1 responses continued to increase beyond Week 16 and then were maintained through Week 52/ 56. In BE VIVID, DLQI 0/1 response rate at Week 52 was higher in bimekizumab-treated patients (74.5%) compared with ustekinumab-treated patients (63.2%).
DLQI 0/1 responses continued to increase beyond Week 16 and then were maintained through Week 52/ 56. In BE VIVID, DLQI 0/1 response rate at Week 52 was higher in bimekizumab-treated patients (74.5%) compared with ustekinumab-treated patients (63.2%).
Psoriasis - phase 3 open label extension study. Patients who completed one of the pivotal phase 3 studies ('feeder studies') could enter a 144-week open-label extension study (OLE) (PS0014) to assess the long-term safety and efficacy of bimekizumab.
From Week 1 to Week 144 of the OLE, patients who were treated with bimekizumab 320 mg every 8 weeks (BKZ 320 mg Q8W) during the feeder study and who achieved PASI90 at the end of the feeder study continued to receive bimekizumab 320 mg Q8W.
Improvements achieved with bimekizumab across the spectrum of efficacy endpoints including PASI90, PASI100, IGA 0/I, PASI < 2, and DLQI 0/1 in the feeder studies were maintained through an additional 144 weeks of open-label treatment (in total, 4 years of treatment) (Table 12).
 Psoriasis - phase 3b head to head study. The efficacy and safety of bimekizumab were also evaluated in a double-blind study compared with secukinumab, an IL-17A inhibitor, (BE RADIANT - PS0015). Patients were randomized to receive bimekizumab (N=373, 320 mg at Week 0, 4, 8, 12 and 16 (Q4W) followed by 320 mg every 4 weeks (Q4W/Q4W) or 320 mg every 8 weeks (Q4W/Q8W)) or secukinumab (N=370, 300 mg at Weeks 0,1, 2, 3, 4 followed by 300 mg every 4 weeks). Baseline characteristics were consistent with a population of moderate to severe plaque psoriasis patients with a median BSA of 19% and a median PASI score of 18.
Psoriasis - phase 3b head to head study. The efficacy and safety of bimekizumab were also evaluated in a double-blind study compared with secukinumab, an IL-17A inhibitor, (BE RADIANT - PS0015). Patients were randomized to receive bimekizumab (N=373, 320 mg at Week 0, 4, 8, 12 and 16 (Q4W) followed by 320 mg every 4 weeks (Q4W/Q4W) or 320 mg every 8 weeks (Q4W/Q8W)) or secukinumab (N=370, 300 mg at Weeks 0,1, 2, 3, 4 followed by 300 mg every 4 weeks). Baseline characteristics were consistent with a population of moderate to severe plaque psoriasis patients with a median BSA of 19% and a median PASI score of 18.
Bimekizumab-treated patients achieved significantly higher response rates compared to secukinumab for the primary endpoint of PASI100 (complete skin clearance) at Week 16. Significantly higher response rates were also achieved with bimekizumab for the secondary endpoints of PASI 100 at Week 48 (for both Q4W/Q4W and Q4W/Q8W regimens) and PASI75 at Week 4. Comparative PASI response rates are presented in Table 13. Bimekizumab was associated with a rapid onset of efficacy. Differences in response rates between bimekizumab and secukinumab-treated patients were noted as early as Week 1 for PASI 75 (7.2% and 1.4% respectively) and as early as Week 2 for PASI 90 (7.5% and 2.4% respectively).
 Bimekizumab and secukinumab PASI 100 response rates through Week 48 are presented in Figure 4.
Bimekizumab and secukinumab PASI 100 response rates through Week 48 are presented in Figure 4.
 Psoriatic arthritis. The safety and efficacy of bimekizumab were evaluated in 1112 adult patients (at least 18 years of age) with active psoriatic arthritis (PsA) in two multicenter, randomized, double-blind, placebo-controlled studies (PA0010 - BE OPTIMAL and PA0011- BE COMPLETE). The BE OPTIMAL study included an active reference treatment arm (adalimumab, N=140). For both studies, patients had a diagnosis of active psoriatic arthritis for at least 6 months based on the Classification Criteria for Psoriatic Arthritis (CASPAR) and had active disease with tender joint count (TJC) ≥ 3 and swollen joint count (SJC) ≥ 3. Patients had a diagnosis of PsA for a median of 4.6 years. Patients with each subtype of PsA were enrolled in these studies, including polyarticular symmetric arthritis, oligoarticular asymmetric arthritis, distal interphalangeal joint predominant, spondylitis predominant and arthritis mutilans. At baseline, 55.9% of patients had ≥ 3% Body Surface Area (BSA) with active plaque psoriasis with 10.4% of patients having moderate to severe plaque psoriasis. 31.9% and 12.3% had enthesitis and dactylitis at baseline respectively. The primary efficacy endpoint in both studies was the American College of Rheumatology (ACR) 50 response at Week 16.
Psoriatic arthritis. The safety and efficacy of bimekizumab were evaluated in 1112 adult patients (at least 18 years of age) with active psoriatic arthritis (PsA) in two multicenter, randomized, double-blind, placebo-controlled studies (PA0010 - BE OPTIMAL and PA0011- BE COMPLETE). The BE OPTIMAL study included an active reference treatment arm (adalimumab, N=140). For both studies, patients had a diagnosis of active psoriatic arthritis for at least 6 months based on the Classification Criteria for Psoriatic Arthritis (CASPAR) and had active disease with tender joint count (TJC) ≥ 3 and swollen joint count (SJC) ≥ 3. Patients had a diagnosis of PsA for a median of 4.6 years. Patients with each subtype of PsA were enrolled in these studies, including polyarticular symmetric arthritis, oligoarticular asymmetric arthritis, distal interphalangeal joint predominant, spondylitis predominant and arthritis mutilans. At baseline, 55.9% of patients had ≥ 3% Body Surface Area (BSA) with active plaque psoriasis with 10.4% of patients having moderate to severe plaque psoriasis. 31.9% and 12.3% had enthesitis and dactylitis at baseline respectively. The primary efficacy endpoint in both studies was the American College of Rheumatology (ACR) 50 response at Week 16.
The BE OPTIMAL study evaluated 852 patients not previously exposed to any biologic disease-modifying anti-rheumatic drug (bDMARD) for the treatment of psoriatic arthritis or psoriasis. Patients were randomized (3:2:1) to receive bimekizumab 160 mg every 4 weeks up to Week 52 or placebo up to Week 16 followed by bimekizumab 160 mg every 4 weeks up to Week 52 or an active reference treatment (adalimumab 40 mg every 2 weeks) up to Week 52. In this study, 78.3% of patients had received prior treatment with ≥ 1 conventional DMARDs (cDMARDs) and 21.7% of patients had no prior treatment with cDMARDs. At baseline, 58.2% of patients were receiving concomitant methotrexate (MTX), 11.3% were receiving concomitant cDMARDs other than MTX, and 30.5% were receiving no cDMARDs.
The BE COMPLETE study evaluated 400 patients with an inadequate response (lack of efficacy) or intolerance to treatment with 1 or 2 tumor necrosis factor alpha inhibitors (anti-TNFα - IR) for either psoriatic arthritis or psoriasis. Patients were randomized (2:1) to receive bimekizumab 160 mg every 4 weeks or placebo up to Week 16. At baseline, 42.5% of patients were receiving concomitant MTX, 8.0% were receiving concomitant cDMARDs other than MTX, and 49.5% were receiving no cDMARDs.
Clinical response.
In bDMARD-naïve patients (BE OPTIMAL) and anti-TNFα IR patients (BE COMPLETE) treatment with bimekizumab resulted in significant improvement in signs and symptoms and measures of disease activity compared to placebo at Week 16, with similar response rates seen in both patient populations (see Table 14). Clinical responses were sustained up to Week 52 in BE OPTIMAL as assessed by ACR 20, ACR 50, ACR 70, MDA, PASI 90, PASI 100 and ACR 50 / PASI 100.
 In BE OPTIMAL, at Week 16, in patients treated with adalimumab (active reference treatment without any statistical comparison to bimekizumab or placebo performed), 45.7%, 41.2% and 45.0% achieved ACR50, PASI 90 and MDA respectively.
In BE OPTIMAL, at Week 16, in patients treated with adalimumab (active reference treatment without any statistical comparison to bimekizumab or placebo performed), 45.7%, 41.2% and 45.0% achieved ACR50, PASI 90 and MDA respectively.
Improvements from baseline were shown in all individual ACR components with bimekizumab at Week 16 (see Table 15) and were sustained up to Week 52 in BE OPTIMAL.
 Bimekizumab was associated with a rapid onset of efficacy in both bDMARD-naïve (BE OPTIMAL) and anti-TNFα-IR (BE COMPLETE) patients. In BE OPTIMAL, as early as Week 2 (first post-baseline visit), the ACR 20 response rate was higher in the bimekizumab-treated group (27.1%) compared with the placebo group (7.8%). By Week 4, after one dose, 17.6% of patients achieved an ACR 50 with bimekizumab compared to 3.2% with placebo. In BE COMPLETE, after one dose, as early as Week 4 (first post-baseline visit) the ACR 20 and ACR 50 response rates were higher for the bimekizumab-treated group (42.7% and 16.1%, respectively) compared with the placebo group (6.8% and 1.5%, respectively). See Figure 5 and Figure 6.
Bimekizumab was associated with a rapid onset of efficacy in both bDMARD-naïve (BE OPTIMAL) and anti-TNFα-IR (BE COMPLETE) patients. In BE OPTIMAL, as early as Week 2 (first post-baseline visit), the ACR 20 response rate was higher in the bimekizumab-treated group (27.1%) compared with the placebo group (7.8%). By Week 4, after one dose, 17.6% of patients achieved an ACR 50 with bimekizumab compared to 3.2% with placebo. In BE COMPLETE, after one dose, as early as Week 4 (first post-baseline visit) the ACR 20 and ACR 50 response rates were higher for the bimekizumab-treated group (42.7% and 16.1%, respectively) compared with the placebo group (6.8% and 1.5%, respectively). See Figure 5 and Figure 6.

 In BE OPTIMAL, of bimekizumab-treated patients who achieved an ACR 50 response at Week 16, 87.2% maintained this response at Week 52.
In BE OPTIMAL, of bimekizumab-treated patients who achieved an ACR 50 response at Week 16, 87.2% maintained this response at Week 52.
In BE OPTIMAL at Week 52, 65.5% of bimekizumab-treated patients achieved complete nail clearance (mNAPSI resolution in patients with mNAPSI > 0 at baseline).
The efficacy and safety of bimekizumab was demonstrated regardless of age, gender, race, baseline body weight, baseline psoriasis involvement, baseline CRP, disease duration, and prior cDMARDs use.
In both studies, similar responses were observed with bimekizumab regardless of whether patients were on concomitant cDMARDs, including MTX, or not.
The modified Psoriatic Arthritis Response Criteria (PsARC) is a specific composite responder index comprising of tender joint count, swollen joints count, patient and physician global assessment. The proportion of patients achieving modified PsARC at Week 16 was higher in the bimekizumab-treated patients compared to placebo (80.3% versus 40.2% in BE OPTIMAL and 85.4% versus 30.8% in BE COMPLETE). In addition, the proportion of patients achieving a low disease activity or remission as measured by the Disease Activity Index for Psoriatic Arthritis (DAPSA score less or equal to 14) was higher in the bimekizumab-treated patients compared to placebo in both studies. PsARC response and DAPSA response were sustained up to Week 52 in BE OPTIMAL.
Patients with axial involvement at baseline (74.1% of patients in BE OPTIMAL and 75% in BE COMPLETE), (defined as a Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) score ≥ 4) showed greater improvement from baseline in BASDAI compared with placebo at Week 16. Improvement achieved at Week 16 was sustained up to Week 52 in BE OPTIMAL.
Radiographic response.
In BE OPTIMAL, inhibition of progression of structural damage was assessed radiographically and expressed as the change from baseline in the Van der Heijde modified total Sharp Score (vdHmTSS) (see Table 16) and its components, the Erosion Score (ES) and the Joint Space Narrowing score (JSN) at Week 16 and Week 52.
 Bimekizumab significantly inhibited the rate of progression of joint damage at Week 16 in both the population with elevated hs-CRP and/or at least 1 bone erosion at baseline and the overall population compared to placebo. The percentage of patients with no radiographic joint damage progression (defined as a change from baseline in mTSS of ≤ 0.5) from randomization to Week 16 was 83.9% for bimekizumab and 77.5% for placebo in the population with elevated hs-CRP and/or at least 1 bone erosion. Similar responses were achieved in the overall population (85.7% for bimekizumab and 78.8% for placebo). The percentage of patients with no radiographic joint damage progression was sustained up to Week 52 with bimekizumab in both patient populations.
Bimekizumab significantly inhibited the rate of progression of joint damage at Week 16 in both the population with elevated hs-CRP and/or at least 1 bone erosion at baseline and the overall population compared to placebo. The percentage of patients with no radiographic joint damage progression (defined as a change from baseline in mTSS of ≤ 0.5) from randomization to Week 16 was 83.9% for bimekizumab and 77.5% for placebo in the population with elevated hs-CRP and/or at least 1 bone erosion. Similar responses were achieved in the overall population (85.7% for bimekizumab and 78.8% for placebo). The percentage of patients with no radiographic joint damage progression was sustained up to Week 52 with bimekizumab in both patient populations.
Physical function and other health-related outcomes.
Both bDMARD-naïve (BE OPTIMAL) and anti-TNFα-IR (BE COMPLETE) patients receiving bimekizumab showed significant improvement from baseline in physical function compared to placebo patients at Week 16 (p < 0.001) as assessed by the HAQ-DI (see Table 15). In both studies, a greater proportion of patients achieved a clinically meaningful reduction of at least 0.35 in HAQ-DI score from baseline in the bimekizumab group compared with placebo at Week 16. Response was sustained up to Week 52 in BE OPTIMAL.
Bimekizumab-treated patients reported significant improvement from baseline in the Short Form-36 item Health Survey Physical Component Summary (SF-36 PCS) score at Week 16 compared to placebo (LS Mean change from baseline: 6.3 versus 1.9, p < 0.001 in BE OPTIMAL and 6.2 versus 0.1, p < 0.001 in BE COMPLETE).
In both studies, bimekizumab-treated patients reported meaningful reduction from baseline in fatigue as measured by the Functional Assessment of Chronic Illness Therapy (FACIT)-Fatigue score at Week 16 compared to placebo. Meaningful improvement from baseline was also observed in PsA specific health-related quality of life as measured by the Psoriatic Arthritis Impact of Disease-12 (PsAID-12) score in the bimekizumab-treated group compared to the placebo group at Week 16.
Bimekizumab-treated patients reported greater improvement in work productivity and daily activity at Week 16 as reported by the Work Productivity and Activity Impairment Questionnaire-specific health problem (WPAI-SHP) compared to placebo.
Improvements in HAQ-DI score, SF-36 PCS score, FACIT-Fatigue score, PsAID-12 score and WPAI-SHP achieved at Week 16 were sustained up to Week 52 in BE OPTIMAL.
Axial spondyloarthritis. The efficacy and safety of bimekizumab were evaluated in 586 adult patients (at least 18 years of age) with active axial spondyloarthritis (axSpA) in two multicenter, randomized, double-blind, placebo-controlled studies, one in non-radiographic axial spondyloarthritis (nr-axSpA) and one in ankylosing spondylitis (AS), also known as radiographic axSpA. The primary endpoint in both studies was the percentage of patients achieving an Assessment of SpondyloArthritis International Society (ASAS) 40 response at Week 16. Consistent results were seen across both patient populations (nr-axSpA and AS).
The BE MOBILE 1 study (AS0010) evaluated 254 patients with active nr-axSpA. Patients had axSpA (age of symptoms onset < 45 years) meeting the ASAS classification criteria and had active disease as defined by a Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) ≥ 4 and spinal pain ≥ 4 on a 0 to 10 numeric rating scale (NRS) (from BASDAI Item 2) and no evidence of radiographic changes in the sacroiliac joints that would meet the modified New York criteria for AS. Patients also had objective signs of inflammation as indicated by elevated C-reactive protein (CRP) level and/or evidence of sacroiliitis on Magnetic Resonance Imaging (MRI) as well as a history of inadequate response to 2 different non-steroidal anti-inflammatory drugs (NSAIDs) or intolerance or contraindication to NSAIDs. Patients were randomized (1:1) to receive bimekizumab 160 mg every 4 weeks up to Week 52 or placebo up to Week 16 followed by bimekizumab 160 mg every 4 weeks up to Week 52. At baseline, patients had symptoms of nr-axSpA for a mean of 9 years (median of 5.5 years). 10.6% of patients were previously treated with an anti-TNFα agent.
The BE MOBILE 2 study (AS0011) evaluated 332 patients with active AS determined by documented radiologic evidence (X-ray) fulfilling the Modified New York criteria for AS. Patients had active disease as defined by a BASDAI ≥ 4 and spinal pain ≥ 4 on a 0 to 10 numeric rating scale (NRS) (from BASDAI Item 2). Patients had to have a history of inadequate response to 2 different NSAIDs or intolerance or contraindication to NSAIDs. Patients were randomized (2:1) to receive bimekizumab 160 mg every 4 weeks up to Week 52 or placebo up to Week 16 followed by bimekizumab 160 mg every 4 weeks up to Week 52. At baseline, patients had symptoms of AS for a mean of 13.5 years (median of 11 years). 16.3% of patients were previously treated with an anti-TNFα agent.
Clinical response.
Treatment with bimekizumab resulted in significant improvement in signs and symptoms and measures of disease activity compared to placebo at Week 16 in both nr-axSpA and AS patient populations (see Table 17). Clinical responses were sustained up to Week 52 in both patient populations as assessed by all the endpoints presented in Table 17.
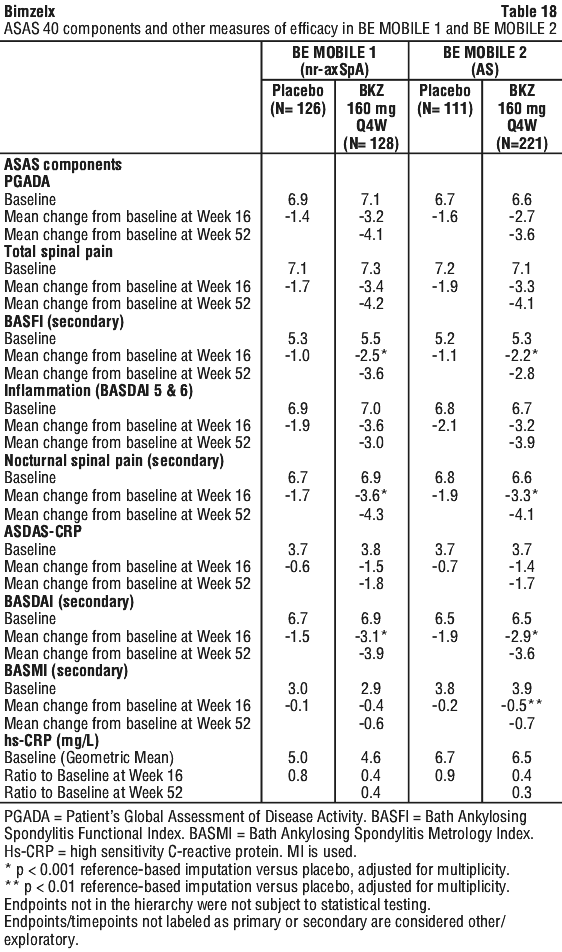
 Improvements in the components of the ASAS 40 response criteria and other measures of efficacy are shown in Table 18. Improvements achieved at Week 16 were sustained up to Week 52 in both patient populations.
Improvements in the components of the ASAS 40 response criteria and other measures of efficacy are shown in Table 18. Improvements achieved at Week 16 were sustained up to Week 52 in both patient populations.
 Bimekizumab was associated with a rapid onset of efficacy in both nr-axSpA and AS patient population.
Bimekizumab was associated with a rapid onset of efficacy in both nr-axSpA and AS patient population.
In BE MOBILE 1, as early as Week 1, the ASAS 20 and ASAS 40 response rates in bimekizumab-treated patients were greater than in placebo-treated patients (31.3% versus 11.9% and 16.4% versus 1.6%, respectively). Bimekizumab was also associated with a rapid decrease in systemic inflammation as measured by the hs-CRP levels. As early as Week 2 (first measurement of hs-CRP), the geometric mean hs-CRP ratio to baseline was lower in the bimekizumab-treated patients (0.4) compared to placebo (0.9).
In BE MOBILE 2, as early as Week 2, the ASAS 20 and ASAS 40 response rates in bimekizumab-treated patients were greater than in placebo-treated patients (43.4% versus 25.2% and 16.7% versus 7.2% respectively). Bimekizumab was also associated with a rapid decrease in systemic inflammation as measured by hs-CRP levels. As early as Week 2 (first measurement of hs-CRP), the geometric mean hs-CRP ratio to baseline was lower in the bimekizumab-treated patients (0.4) compared to placebo (0.9). See Figure 7 and Figure 8.

 In an integrated analysis of BE MOBILE 1 and BE MOBILE 2, of bimekizumab-treated patients who achieved an ASAS 40 response at Week 16, 82.1% maintained this response at Week 52.
In an integrated analysis of BE MOBILE 1 and BE MOBILE 2, of bimekizumab-treated patients who achieved an ASAS 40 response at Week 16, 82.1% maintained this response at Week 52.
The efficacy of bimekizumab was demonstrated regardless of age, gender, race, disease duration, baseline inflammation status, baseline ASDAS and concomitant cDMARDs. Patients with a BMI ≥ 30 kg/m2 may take longer time to achieve clinical response.
Similar response in ASAS 40 was seen in patients regardless of prior anti-TNFα exposure.
At Week 16, among patients with enthesitis at baseline, the proportion of patients (NRI) with enthesitis resolution as assessed by the Maastricht Ankylosing Spondylitis Enthesitis (MASES) index was greater with bimekizumab compared to placebo (BE MOBILE 1: 51.1% versus 23.9% and BE MOBILE 2: 51.5% versus 32.8%). Response with bimekizumab was sustained up to Week 52 in both studies (BE MOBILE 1: 54.3% and BE MOBILE 2: 50.8%).
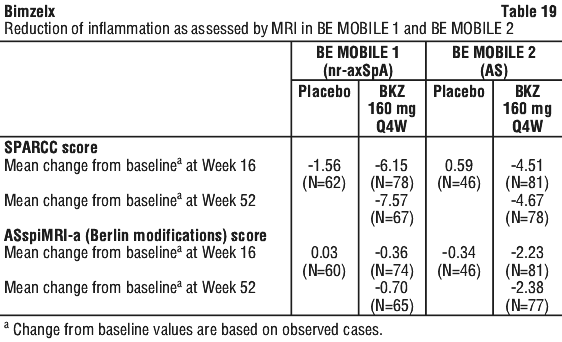
Reduction of inflammation.
Bimekizumab reduced inflammation as measured by hs-CRP (see Table 18) and as assessed by MRI in an imaging sub-study. Signs of inflammation were assessed by MRI at baseline and Week 16 and expressed as change from baseline in Spondyloarthritis Research Consortium of Canada (SPARCC) score for sacroiliac joints and Ankylosing Spondylitis spine Magnetic Resonance Imagine-activity (ASspiMRI-a score in the Berlin modification) for the spine. Reduction of inflammatory signs in both sacroiliac joints and the spine was observed in patients treated with bimekizumab as compared with placebo (see Table 19). Reduction of inflammation as measured by hs-CRP and as assessed by MRI was sustained to Week 52.
Physical function and other health-related outcomes.
Patients treated with bimekizumab showed significant improvement from baseline in physical function as assessed by the BASFI (see Table 18). Patients treated with bimekizumab reported significant improvement from baseline compared to placebo-treated patients in SF-36 PCS score (LS Mean change from baseline at Week 16 in BE MOBILE 1: 9.3 versus 5.4, p < 0.001 and in BE MOBILE 2: 8.5 versus 5.2, p < 0.001).
Patients treated with bimekizumab reported significant improvement from baseline in health-related quality of life as measured by the AS Quality of Life Questionnaire (ASQoL) compared to placebo (LS Mean change from baseline at Week 16 in BE MOBILE 1: -4.9 versus -2.3, p < 0.001 and in BE MOBILE 2: -4.6 versus -3.0, p < 0.001) as well as meaningful reduction in fatigue as assessed by the FACIT-Fatigue score (Mean change from baseline at Week 16 in BE MOBILE 1: 8.5 for bimekizumab versus 3.9 for placebo and in BE MOBILE 2: 8.4 for bimekizumab versus 5.0 for placebo).
Bimekizumab-treated patients reported greater improvement in work productivity and daily activities at Week 16 as reported by the WPAI-SHP compared to placebo.
Improvements in BASFI, SF-36 PCS score, ASQoL, FACIT-Fatigue score and WPAI-SHP achieved at Week 16 were sustained up to Week 52 in both studies.
Extra-articular manifestation.
In pooled data from BE MOBILE 1 (nr-axSpA) and BE MOBILE 2 (AS), at Week 16, the proportion of patients developing a uveitis event was lower with bimekizumab (0.6%) compared to placebo (4.6%). The incidence of uveitis remains low in bimekizumab-treated patients (1.2/100 patient-years in the pooled phase 2/3 studies).
Hidradenitis suppurativa. The safety and efficacy of bimekizumab was evaluated in 1014 adult patients (at least 18 years of age) with moderate to severe Hidradenitis Suppurativa (HS) in two Phase 3 multicenter, randomized, double-blind, placebo-controlled studies (HS0003 - BE HEARD I and HS0004 - BE HEARD II).
Patients had a diagnosis of HS for at least 6 months with Hurley Stage II or Hurley Stage III disease, and with ≥ 5 inflammatory lesions (i.e. number of abscesses plus number of inflammatory nodules) and had a history of inadequate response to a course of systemic antibiotics for the treatment of HS.
In both studies patients were randomized (2:2:2:1) to receive bimekizumab 320 mg every 2 weeks for 48 weeks (320 mg Q2W/Q2W) or bimekizumab 320 mg every 4 weeks for 48 weeks (320 mg Q4W/Q4W) or bimekizumab 320 mg every 2 weeks to Week 16 followed by 320 mg every 4 weeks up to Week 48 (320 mg Q2W/Q4W) or placebo to Week 16 followed by bimekizumab 320 mg every 2 weeks up to Week 48. Concomitant oral antibiotic use was allowed if the patient was on a stable dose regimen of doxycycline, minocycline, or an equivalent systemic tetracycline for 28 days prior to baseline.
The primary efficacy endpoint in both studies was the Hidradenitis Suppurativa Clinical Response 50 (HiSCR50) at Week 16, i.e. at least a 50% reduction in the total abscess and inflammatory nodule count with no increase in abscess or draining tunnel count relative to baseline.
Baseline characteristics were consistent across both studies and reflective of a population with moderate to severe HS. Patients had a median disease duration of 5.3 years (mean 8.0 years). The proportions of Hurley Stage II and Stage III patients were 55.7% and 44.3% respectively, and 8.5% were receiving concomitant antibiotic therapy for HS. 56.8% of patients were female and the mean age of all patients was 36.6 years. 79.7% of patients were White, and 10.8% were Black or African American. 45.6% of patients were current smokers.
The mean International Hidradenitis Suppurativa Severity Score System (IHS4) score was 34.2 indicating severe disease. The mean baseline Abscess and Inflammatory Nodule (AN) Count was 16.3, the mean baseline worst skin pain score was 5.5 and the mean baseline total Hidradenitis suppurativa Quality of Life (HiSQOL) and total Dermatology Life Quality Index (DLQI) scores were 25.2 and 11.4, respectively.
Clinical response.
Treatment with bimekizumab resulted in improvement in disease activity compared to placebo at Week 16. Key efficacy results are shown in Table 20. The results in Table 20 reflect the predefined primary analysis in which any systemic antibiotic use prior to Week 16 resulted in imputation of nonresponse.
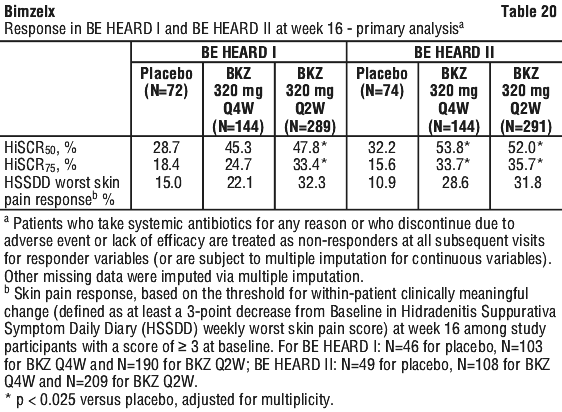 At Week 4, HiSCR50 response rates were higher for bimekizumab-treated patients (36.5% and 35.1%) compared to placebo (18.1% and 22.2%) in BE HEARD I and BE HEARD II, respectively.
At Week 4, HiSCR50 response rates were higher for bimekizumab-treated patients (36.5% and 35.1%) compared to placebo (18.1% and 22.2%) in BE HEARD I and BE HEARD II, respectively.
Improvement in patients reporting worst skin pain response as measured by the HSSDD started at Week 2. See Figure 9
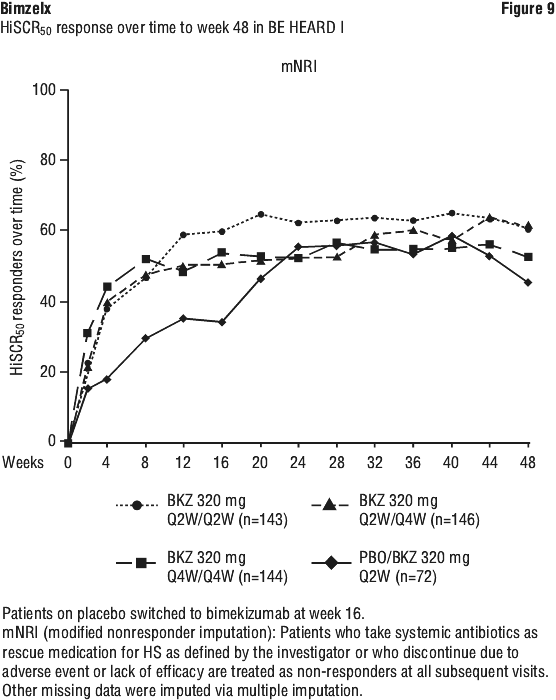 See Figure 10.
See Figure 10.
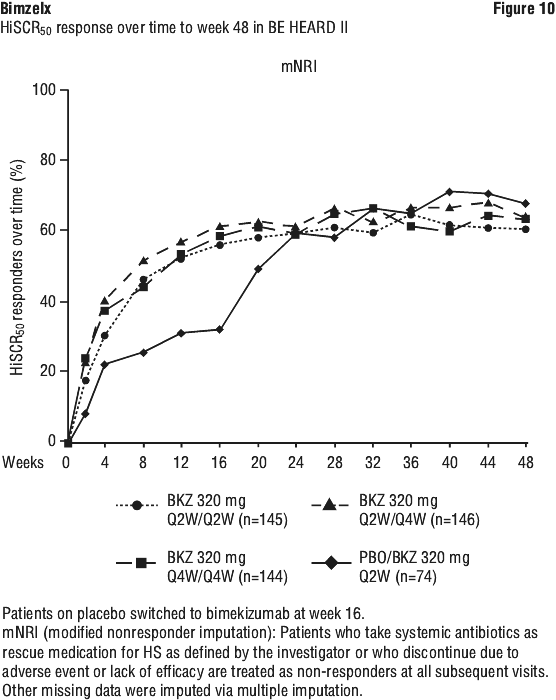 Consistent with the HiSCR50 response rates, the ≥ 50% reduction in the total abscess and inflammatory nodule (AN50) count responder rates at Week 16 were higher in the bimekizumab treated patients (320 mg every 2 weeks) compared to placebo (51.8% versus 34.7% in BE HEARD I and 58.1% versus 39.0% in BE HEARD II. Responses were sustained up to Week 48 with bimekizumab 320 mg every 4 weeks.
Consistent with the HiSCR50 response rates, the ≥ 50% reduction in the total abscess and inflammatory nodule (AN50) count responder rates at Week 16 were higher in the bimekizumab treated patients (320 mg every 2 weeks) compared to placebo (51.8% versus 34.7% in BE HEARD I and 58.1% versus 39.0% in BE HEARD II. Responses were sustained up to Week 48 with bimekizumab 320 mg every 4 weeks.
The efficacy of bimekizumab was demonstrated regardless of age, gender, race, disease duration, weight, prior biologics therapy and systemic antibiotic use at baseline.
Health-related quality of life.
Across both studies, patients treated with bimekizumab experienced greater improvement in their health-related quality of life as measured by DLQI questionnaire compared to placebo (Table 21).
 Improvement achieved at Week 16 in health-related quality of life measurements with bimekizumab were sustained through Week 48.
Improvement achieved at Week 16 in health-related quality of life measurements with bimekizumab were sustained through Week 48.
5.2 Pharmacokinetic Properties
The pharmacokinetic (PK) properties of bimekizumab were similar in patients with plaque psoriasis, psoriatic arthritis and axial spondyloarthritis (nr-axSpA and AS). Based on population PK analyses and using a reference bodyweight of 90 kg, the bimekizumab apparent clearance and volume of distribution, respectively, in patients with HS were estimated to be approximately 31 and 18% higher than for the aforementioned indications, with an estimated half-life in HS of 20 days. Consequently, the median steady state trough concentration at a dose of 320 mg every 4 weeks was approximately 40% lower in HS compared to other indications.
Bimekizumab exhibited dose-proportional pharmacokinetics in patients with plaque psoriasis over a dose range from 64 mg to 480 mg following multiple subcutaneous administrations, with apparent clearance being independent of dose.
Absorption.
Based on population PK analysis, following a single subcutaneous dose of 320 mg in plaque psoriasis patients, bimekizumab reached a median (2.5th and 97.5th percentile) peak plasma concentration of 25 (12-50) microgram/mL, between 3 and 4 days post dose.
Population pharmacokinetic analysis showed that bimekizumab was absorbed with an average absolute bioavailability of 70.1% in healthy volunteers.
Distribution.
Based on population PK analyses, the median (coefficient of variation %) volume of distribution (V/F) at steady state was 11.2 (30.5%) L in plaque psoriasis patients.
Metabolism.
Bimekizumab is a monoclonal antibody and is expected to be degraded into small peptides and amino acids via catabolic pathways in the same manner as endogenous immunoglobulins.
Excretion.
Based on population PK analyses, the median (coefficient of variation %) apparent clearance (CL/F) of bimekizumab was 0.337 L/day (32.7%) and the mean terminal elimination half-life of bimekizumab was 23 days in clinical studies in patients with plaque psoriasis.
Based on simulated data, the median (2.5th and 97.5th percentile) peak and trough concentration at steady-state following subcutaneous administration of 320 mg every 4 weeks are 43 (20-91) microgram/mL and 20 (7-50) microgram/mL respectively and steady-state is reached after approximately 16 weeks with every 4 weeks dosing regimen. Compared with exposure after a single dose, the population PK analysis showed that patients exhibited a 1.74-fold increase in peak plasma concentrations and area under the curve (AUC) following repeated four weekly dosing.
After switching from the 320 mg every 4 weeks dosing regimen to 320 mg every 8 weeks dosing regimen at Week 16, steady-state is achieved approximately 16 weeks after the switch. Median (2.5th and 97.5th percentile) peak and trough plasma concentrations are 30 (14 -60) microgram/mL and 5 (1-16) microgram/mL respectively.
Pharmacokinetic/pharmacodynamic relationship.
A population PK/PD model was developed using all available data in moderate to severe plaque psoriasis patients. The analysis showed that higher bimekizumab concentrations are related to better Psoriasis Area and Severity Index (PASI) and Investigators Global Assessment (IGA) response and a dose of 320 mg at Week 0, 4, 8, 12, 16 and every 8 weeks thereafter provides maximum benefit to the majority of moderate to severe plaque psoriasis patients (see Special populations, Body weight).
Special populations.
Elderly.
Based on population PK analysis with a limited number of elderly patients (n = 355 for age ≥ 65 years and n = 47 for age ≥ 75 years), apparent clearance (CL/F) in elderly patients and patients less than 65 years of age was similar. No dose adjustment is required.
Renal or hepatic impairment.
No specific studies have been conducted to determine the effect of renal or hepatic impairment on the pharmacokinetics of bimekizumab. Caution should be taken in people with hepatic and renal dysfunction, due to the absence of data from these populations in the studies. The renal elimination of intact bimekizumab, an IgG monoclonal antibody, is expected to be low and of minor importance. Similarly, IgGs are mainly eliminated via intracellular catabolism and hepatic impairment is not expected to influence clearance of bimekizumab. Based on population PK analyses, hepatic function markers (ALT/ bilirubin) did not have any impact on bimekizumab clearance in patients with plaque psoriasis.
Body weight.
Population PK modelling indicated that exposure decreased as body weight increased. The average plasma concentration in adult patients weighing ≥ 120 kg following a 320 mg subcutaneous injection was predicted to be at least 30% lower than in adult patients weighing 90 kg. Dose adjustment may be appropriate in some patients (see Section 4.2 Dose and Method of Administration).
Race/gender.
No clinically meaningful differences in bimekizumab exposure were observed in Japanese or Chinese subjects compared to Caucasian subjects in a clinical PK study. No dose adjustment is required.
Population PK modelling indicated females may have 10% faster apparent clearance (CL/F) compared to males and it is not clinically meaningful. No dose adjustment is required.
5.3 Preclinical Safety Data
Genotoxicity.
No genotoxicity studies were conducted with bimekizumab. Monoclonal antibodies are not expected to damage DNA or chromosomes.
Carcinogenicity.
No carcinogenicity studies were conducted with bimekizumab.6 Pharmaceutical Particulars
6.1 List of Excipients
The inactive ingredients are glycine, sodium acetate trihydrate, acetic acid, polysorbate 80, water for injections.
6.2 Incompatibilities
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal products.
6.4 Special Precautions for Storage
Store in a refrigerator (2°C - 8°C).
Do not freeze.
Keep the pre-filled syringe or pen in the outer carton in order to protect from light.
The Bimzelx pre-filled syringe and pre-filled pen may be stored at room temperature (up to 25°C) for a single period of maximum 30 days with protection from light. Once removed from the refrigerator and stored under these conditions, discard after 30 days or by the expiry date printed on the container, whichever occurs first. A field for the date is provided on the carton to record the date removed from the refrigerator.
6.5 Nature and Contents of Container
Bimzelx 160 mg solution for injection in pre-filled syringe.
One mL pre-filled syringe (type I glass) with a fluoropolymer-laminated bromobutyl rubber stopper, staked 27 G, ½" thin wall needle, and a rigid needle shield (consisting of a thermoplastic elastomer needle cover and a polypropylene rigid shield) assembled in a passive safety device.
Pack size of 2 pre-filled syringes.
Bimzelx 320 mg solution for injection in pre-filled syringe.
Two mL pre-filled syringe (type I glass) with a fluoropolymer-laminated bromobutyl rubber stopper, staked 27 G, ½" thin wall needle, and a rigid needle shield (consisting of a thermoplastic elastomer needle cover and a polypropylene rigid shield) assembled in a passive safety device.
Pack size of 1 pre-filled syringe.
Bimzelx 160 mg solution for injections in pre-filled pen.
One mL pre-filled pen containing a pre-filled syringe (type I glass) with a fluoropolymer-laminated bromobutyl rubber stopper, staked 27 G, ½" thin wall needle, and a rigid needle shield consisting of a thermoplastic elastomer needle cover and a polypropylene rigid shield.
Pack size of 2 pre-filled pens.
Bimzelx 320 mg solution for injection in pre-filled pen.
Two mL pre-filled pen containing a pre-filled syringe (type I glass) with a fluoropolymer-laminated bromobutyl rubber stopper, staked 27 G, ½" thin wall needle, and a rigid needle shield consisting of a thermoplastic elastomer needle cover and a polypropylene rigid shield.
Pack size of 1 pre-filled pen.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical name: Immunoglobulin G1, anti-IL17A and anti-IL17F.
Nominal theoretical molecular mass: approximately 149,886 Daa.
a Theoretical mass based on presence of G0F glycans and clipped heavy chain C-terminal modifications.
Chemical structure.

CAS number.
1418205-77-2.7 Medicine Schedule (Poisons Standard)
S4.
Summary Table of Changes







 Adverse drug reactions occurring in greater than 1% of subjects on Bimzelx and at a higher rate than in the placebo group through week 16 in the HS clinical trials that are not listed in the table above are constipation, asthenia, otitis externa, vulvovaginal mycotic infection, tinea pedis, myalgia, acne, seborrhoeic dermatitis, urticaria.
Adverse drug reactions occurring in greater than 1% of subjects on Bimzelx and at a higher rate than in the placebo group through week 16 in the HS clinical trials that are not listed in the table above are constipation, asthenia, otitis externa, vulvovaginal mycotic infection, tinea pedis, myalgia, acne, seborrhoeic dermatitis, urticaria. Bimekizumab was associated with a rapid onset of efficacy. In BE VIVID, at Week 2 and Week 4, PASI 90 response rates were significantly higher for bimekizumab-treated patients (12.1% and 43.6% respectively) compared to placebo (1.2% and 2.4% respectively) and ustekinumab (1.2% and 3.1% respectively). See Figure 1.
Bimekizumab was associated with a rapid onset of efficacy. In BE VIVID, at Week 2 and Week 4, PASI 90 response rates were significantly higher for bimekizumab-treated patients (12.1% and 43.6% respectively) compared to placebo (1.2% and 2.4% respectively) and ustekinumab (1.2% and 3.1% respectively). See Figure 1. In the BE VIVID study, at Week 52, bimekizumab-treated patients achieved significantly higher response rates than the ustekinumab-treated patients on the endpoints of PASI 90 (81.6% bimekizumab vs 55.8% ustekinumab, p < 0.001), IGA 0/1 (77.9% bimekizumab vs 60.7% ustekinumab, p < 0.001) and PASI 100 (64.2% bimekizumab vs 38.0% ustekinumab).
In the BE VIVID study, at Week 52, bimekizumab-treated patients achieved significantly higher response rates than the ustekinumab-treated patients on the endpoints of PASI 90 (81.6% bimekizumab vs 55.8% ustekinumab, p < 0.001), IGA 0/1 (77.9% bimekizumab vs 60.7% ustekinumab, p < 0.001) and PASI 100 (64.2% bimekizumab vs 38.0% ustekinumab). The efficacy of bimekizumab was demonstrated regardless of age, gender, race, disease duration, body weight, PASI baseline severity and previous treatment with a biologic. Bimekizumab was efficacious in prior biologic exposed patients, including anti-TNF/ anti IL-17 and in systemic treatment-naïve patients.
The efficacy of bimekizumab was demonstrated regardless of age, gender, race, disease duration, body weight, PASI baseline severity and previous treatment with a biologic. Bimekizumab was efficacious in prior biologic exposed patients, including anti-TNF/ anti IL-17 and in systemic treatment-naïve patients. Although the number of subjects in the ≥ 120 kg group is low, the data demonstrate that response rates were lower in patients weighing more than 120 kg compared with patients who weighed less and, represents a clinical correlation with the reduction in exposure as predicted by the PK/PD modelling.
Although the number of subjects in the ≥ 120 kg group is low, the data demonstrate that response rates were lower in patients weighing more than 120 kg compared with patients who weighed less and, represents a clinical correlation with the reduction in exposure as predicted by the PK/PD modelling.
 In BE READY, for PASI 90 responders at Week 16 who were re-randomized to placebo and withdrawn from bimekizumab, the median time to relapse, defined as loss of PASI 75, was approximately 28 weeks (32 weeks after the last bimekizumab dose). Among these patients, 88.1% regained a PASI 90 response within 12 weeks of restarting treatment with bimekizumab 320 mg every 4 weeks.
In BE READY, for PASI 90 responders at Week 16 who were re-randomized to placebo and withdrawn from bimekizumab, the median time to relapse, defined as loss of PASI 75, was approximately 28 weeks (32 weeks after the last bimekizumab dose). Among these patients, 88.1% regained a PASI 90 response within 12 weeks of restarting treatment with bimekizumab 320 mg every 4 weeks. Scalp IGA and palmoplantar IGA responses were maintained through Week 52/56. Nail psoriasis continued to improve beyond Week 16. In BE VIVID, at Week 52, 60.3% of patients treated with bimekizumab 320 mg every 4 weeks achieved complete nail clearance (mNAPSI 100). In BE READY, at Week 56, 67.7% and 69.8% of Week 16 PASI 90 responders achieved complete nail clearance with bimekizumab 320 mg every 8 weeks and bimekizumab 320 mg every 4 weeks respectively.
Scalp IGA and palmoplantar IGA responses were maintained through Week 52/56. Nail psoriasis continued to improve beyond Week 16. In BE VIVID, at Week 52, 60.3% of patients treated with bimekizumab 320 mg every 4 weeks achieved complete nail clearance (mNAPSI 100). In BE READY, at Week 56, 67.7% and 69.8% of Week 16 PASI 90 responders achieved complete nail clearance with bimekizumab 320 mg every 8 weeks and bimekizumab 320 mg every 4 weeks respectively. DLQI 0/1 responses continued to increase beyond Week 16 and then were maintained through Week 52/ 56. In BE VIVID, DLQI 0/1 response rate at Week 52 was higher in bimekizumab-treated patients (74.5%) compared with ustekinumab-treated patients (63.2%).
DLQI 0/1 responses continued to increase beyond Week 16 and then were maintained through Week 52/ 56. In BE VIVID, DLQI 0/1 response rate at Week 52 was higher in bimekizumab-treated patients (74.5%) compared with ustekinumab-treated patients (63.2%).
 Bimekizumab and secukinumab PASI 100 response rates through Week 48 are presented in Figure 4.
Bimekizumab and secukinumab PASI 100 response rates through Week 48 are presented in Figure 4.
 In BE OPTIMAL, at Week 16, in patients treated with adalimumab (active reference treatment without any statistical comparison to bimekizumab or placebo performed), 45.7%, 41.2% and 45.0% achieved ACR50, PASI 90 and MDA respectively.
In BE OPTIMAL, at Week 16, in patients treated with adalimumab (active reference treatment without any statistical comparison to bimekizumab or placebo performed), 45.7%, 41.2% and 45.0% achieved ACR50, PASI 90 and MDA respectively. Bimekizumab was associated with a rapid onset of efficacy in both bDMARD-naïve (BE OPTIMAL) and anti-TNFα-IR (BE COMPLETE) patients. In BE OPTIMAL, as early as Week 2 (first post-baseline visit), the ACR 20 response rate was higher in the bimekizumab-treated group (27.1%) compared with the placebo group (7.8%). By Week 4, after one dose, 17.6% of patients achieved an ACR 50 with bimekizumab compared to 3.2% with placebo. In BE COMPLETE, after one dose, as early as Week 4 (first post-baseline visit) the ACR 20 and ACR 50 response rates were higher for the bimekizumab-treated group (42.7% and 16.1%, respectively) compared with the placebo group (6.8% and 1.5%, respectively). See Figure 5 and Figure 6.
Bimekizumab was associated with a rapid onset of efficacy in both bDMARD-naïve (BE OPTIMAL) and anti-TNFα-IR (BE COMPLETE) patients. In BE OPTIMAL, as early as Week 2 (first post-baseline visit), the ACR 20 response rate was higher in the bimekizumab-treated group (27.1%) compared with the placebo group (7.8%). By Week 4, after one dose, 17.6% of patients achieved an ACR 50 with bimekizumab compared to 3.2% with placebo. In BE COMPLETE, after one dose, as early as Week 4 (first post-baseline visit) the ACR 20 and ACR 50 response rates were higher for the bimekizumab-treated group (42.7% and 16.1%, respectively) compared with the placebo group (6.8% and 1.5%, respectively). See Figure 5 and Figure 6.
 In BE OPTIMAL, of bimekizumab-treated patients who achieved an ACR 50 response at Week 16, 87.2% maintained this response at Week 52.
In BE OPTIMAL, of bimekizumab-treated patients who achieved an ACR 50 response at Week 16, 87.2% maintained this response at Week 52. Bimekizumab significantly inhibited the rate of progression of joint damage at Week 16 in both the population with elevated hs-CRP and/or at least 1 bone erosion at baseline and the overall population compared to placebo. The percentage of patients with no radiographic joint damage progression (defined as a change from baseline in mTSS of ≤ 0.5) from randomization to Week 16 was 83.9% for bimekizumab and 77.5% for placebo in the population with elevated hs-CRP and/or at least 1 bone erosion. Similar responses were achieved in the overall population (85.7% for bimekizumab and 78.8% for placebo). The percentage of patients with no radiographic joint damage progression was sustained up to Week 52 with bimekizumab in both patient populations.
Bimekizumab significantly inhibited the rate of progression of joint damage at Week 16 in both the population with elevated hs-CRP and/or at least 1 bone erosion at baseline and the overall population compared to placebo. The percentage of patients with no radiographic joint damage progression (defined as a change from baseline in mTSS of ≤ 0.5) from randomization to Week 16 was 83.9% for bimekizumab and 77.5% for placebo in the population with elevated hs-CRP and/or at least 1 bone erosion. Similar responses were achieved in the overall population (85.7% for bimekizumab and 78.8% for placebo). The percentage of patients with no radiographic joint damage progression was sustained up to Week 52 with bimekizumab in both patient populations. Improvements in the components of the ASAS 40 response criteria and other measures of efficacy are shown in Table 18. Improvements achieved at Week 16 were sustained up to Week 52 in both patient populations.
Improvements in the components of the ASAS 40 response criteria and other measures of efficacy are shown in Table 18. Improvements achieved at Week 16 were sustained up to Week 52 in both patient populations. Bimekizumab was associated with a rapid onset of efficacy in both nr-axSpA and AS patient population.
Bimekizumab was associated with a rapid onset of efficacy in both nr-axSpA and AS patient population.
 In an integrated analysis of BE MOBILE 1 and BE MOBILE 2, of bimekizumab-treated patients who achieved an ASAS 40 response at Week 16, 82.1% maintained this response at Week 52.
In an integrated analysis of BE MOBILE 1 and BE MOBILE 2, of bimekizumab-treated patients who achieved an ASAS 40 response at Week 16, 82.1% maintained this response at Week 52.
 At Week 4, HiSCR50 response rates were higher for bimekizumab-treated patients (36.5% and 35.1%) compared to placebo (18.1% and 22.2%) in BE HEARD I and BE HEARD II, respectively.
At Week 4, HiSCR50 response rates were higher for bimekizumab-treated patients (36.5% and 35.1%) compared to placebo (18.1% and 22.2%) in BE HEARD I and BE HEARD II, respectively. See Figure 10.
See Figure 10. Consistent with the HiSCR50 response rates, the ≥ 50% reduction in the total abscess and inflammatory nodule (AN50) count responder rates at Week 16 were higher in the bimekizumab treated patients (320 mg every 2 weeks) compared to placebo (51.8% versus 34.7% in BE HEARD I and 58.1% versus 39.0% in BE HEARD II. Responses were sustained up to Week 48 with bimekizumab 320 mg every 4 weeks.
Consistent with the HiSCR50 response rates, the ≥ 50% reduction in the total abscess and inflammatory nodule (AN50) count responder rates at Week 16 were higher in the bimekizumab treated patients (320 mg every 2 weeks) compared to placebo (51.8% versus 34.7% in BE HEARD I and 58.1% versus 39.0% in BE HEARD II. Responses were sustained up to Week 48 with bimekizumab 320 mg every 4 weeks. Improvement achieved at Week 16 in health-related quality of life measurements with bimekizumab were sustained through Week 48.
Improvement achieved at Week 16 in health-related quality of life measurements with bimekizumab were sustained through Week 48.
