What is in this leaflet
This leaflet answers some common questions about CANESORAL® DUO.
It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have benefits and risks. Your doctor or pharmacist has weighed the risks of you using this medicine against the benefits expected for you.
If you have any concerns about using this medicine, talk to your doctor or pharmacist.
Keep this leaflet with your medicine. You may need to read it again.
The information in this leaflet relates only to CANESORAL® DUO. It is not to be used in relation to any other product, which may also contain the same active ingredients.
What CANESORAL DUO® is used for
CANESORAL® DUO is used to treat a type of fungal infection called vaginal thrush.
CANESORAL® DUO contains two components: CANESORAL® is an oral capsule which contains the active ingredient fluconazole; CANESTEN® CLOTRIMAZOLE CREAM contains the active ingredient clotrimazole. Both of these ingredients belong to a group of medicines called azole antifungals.
CANESORAL® is taken by mouth and works from the inside to prevent the growth of the fungi causing your infection; CANESTEN® CLOTRIMAZOLE CREAM is applied to the irritated area and provides soothing relief from external itching and irritation.
What is vaginal thrush
Vaginal thrush is a common name for vaginal candidiasis, an infection caused by a yeast-like fungus called Candida.
Candida is one of many organisms that live in the vagina. Your body’s natural balance (immune system) normally keeps Candida under control, but when this natural balance is upset, Candida can multiply and can cause thrush symptoms.
Common symptoms of vaginal thrush include:
- itching, burning or soreness around the vagina
- cottage-cheese like discharge
- swelling or irritation of the infected area.
Things that may help you to avoid thrush in the future:
- wear cotton briefs, stockings and loose-fitting clothing rather than tight synthetic clothing
- wash regularly, but do not wash and dry yourself harshly
- avoid perfumed soaps, bath additives and vaginal deodorants.
Your doctor or pharmacist may have more information on things you can do to avoid thrush in the future.
Ask your doctor or pharmacist if you have any questions about why this medicine has been recommended for you. Your doctor or pharmacist may have recommended it for another reason.
There is no evidence that either component in CANESORAL® DUO is addictive.
CANESORAL® DUO is a “Pharmacist Only Medicine”. It is available without a doctor’s prescription but your pharmacist’s advice is required.
CANESORAL® DUO is not recommended for children under 18 years of age except under doctor supervision.
Before you use CANESORAL® DUO
When you must not use it
Do not use CANESORAL® DUO if you have an allergy to:
- any medicine containing fluconazole
- any medicine containing clotrimazole
- any other azole antifungals related to fluconazole or clotrimazole such as miconazole (e.g. Daktarin), ketoconazole (e.g. Nizoral) or itraconazole (e.g. Sporanox)
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty in breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
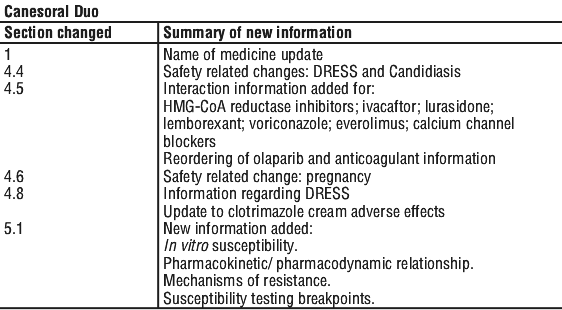
Do not use CANESORAL® DUO if you are taking:
- cisapride (Prepulsid), a medicine used to treat stomach problems
- astemizole
- pimozide
- erythromycin
- quinidine.
Combining CANESORAL® DUO with the above medicines may cause serious side effects such as an abnormal heart rhythm.
Do not use CANESORAL® DUO if you are pregnant, suspect you may be pregnant or if you may become pregnant during treatment. It may affect your developing baby if you use it during pregnancy.
Do not use CANESORAL® DUO if you are breastfeeding. The active ingredient fluconazole passes into breast milk and there is a possibility that your baby may be affected.
Do not use this medicine after the expiry date printed on the pack, or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start using this medicine, talk to your doctor or pharmacist.
Before you start to use it
Tell your doctor or pharmacist if you have allergies to any other medicines, foods, dyes or preservatives.
Tell your doctor or pharmacist if you are over 60 years of age.
Tell your doctor or pharmacist if you have or have had any of the following medical conditions:
- thrush more than twice in the last six months
- liver problems
- kidney problems
- heart problems
- HIV infection or AIDS
- diabetes.
Your doctor or pharmacist may want to take special care if you have any of these conditions.
Tell your doctor or pharmacist before using CANESORAL® DUO if you are taking warfarin (eg. Marevan, Coumadin) as bleeding or bruising may occur.
Tell your doctor or pharmacist if you are experiencing any of the following:
- abnormal or irregular vaginal bleeding or blood stained discharge
- foul smelling or unusual coloured discharge
- vulval or vaginal sores, ulcers or blisters
- lower abdominal pain or burning when passing urine
- fever or chills.
If you have not told your doctor or pharmacist about any of the above, tell him/her before you start using CANESORAL® DUO.
Using the CANESTEN® CLOTRIMAZOLE CREAM component of CANESORAL® DUO may reduce the effectiveness and safety of latex products, such as condoms and diaphragms. This effect is temporary and occurs only during treatment.
Taking other medicines
Do not use CANESORAL® DUO if you are taking
- cisapride (Prepulsid), a medicine used to treat stomach problems
- astemizole
- pimozide
- erythromycin
- quinidine.
Tell your doctor or pharmacist if you are taking any other medicines, including any that you buy without a prescription from a pharmacy, supermarket or health food shop.
Some medicines and CANESORAL® DUO may interfere with each other. These include:
- warfarin (eg. Marevan, Coumadin), a medicine used to prevent blood clots
- phenytoin (eg. Dilantin) or carbamazepine (eg. Tegretol), medicines used to treat epilepsy
- cyclosporin (eg. Neoral), sirolimus (eg. Rapamune) or tacrolimus (eg. Prograf), medicines used to prevent organ transplant rejection or to treat certain problems with the immune system
- certain medicines use to treat diabetes such as:
- glibenclamide (eg. Daonil, Glimel), glipizide (eg. Minidiab, Melizide), glimepiride (eg. Amaryl), gliclazide (eg. Diamicron, Glyade)
- pioglitazone (eg. Actos), rosiglitazone (eg. Avandia) - rifampicin (eg. Rifadin, Rimycin) or rifabutin (eg. Mycobutin), antibiotics used to treat infections
- theophylline (eg. Nuelin), a medicine used to treat asthma
- midazolam (eg. Hypnovel) and triazolam (eg. Halcion), medicines used as sedatives or to treat anxiety
- zidovudine (eg. Retrovir), saquinavir (eg. Invirase), medicines used to treat AIDS patients
- hydrochlorothiazide (eg. Dithiazide), a medicine used for treating fluid problems and high blood pressure
- the contraceptive pill (birth control pill)
- amphotericin B (eg. Fungilin) and voriconazole, medicines used to treat fungal infection
- azithromycin (eg. Zithromax), an antibiotic used to treat certain types of bacterial infections
- anticancer drugs such as cyclophophamide, vincristine and vinblastine, medicines used to treat certain types of cancers
- carbamazepine (eg. Tegretol), a medicine used in the treatment of epilepsy and bipolar disorder
- NSAIDS such as naproxen, diclofenac and celecoxib (eg. Celebrex)
- opioid pain killers such as alfentanil, fentanyl and methadone
- losartan, a medicine used to treat high blood pressure
- calcium channel blockers, such as nifedipine, amlodipine and felodipine used in relieving high blood pressure and certain heart conditions
- statins such as atorvastatin, simvastatin and fluvastatin, used to control high cholesterol levels
- antidepressants such as amitriptyline (eg. Endep) and nortriptyline.
Talk to your doctor about the need for an additional method of contraception while using CANESORAL® DUO. It may decrease the effectiveness of some birth control pills.
Your doctor can tell you what to do if you are taking any of these medicines.
These medicines may be affected by CANESORAL® DUO or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while using this medicine.
How to use CANESORAL® DUO
Follow all directions given to you by your doctor and pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the pack, ask your doctor or pharmacist for help.
How to use it
For vaginal thrush in adults, only a single dose (1 capsule) is needed.
Swallow the capsule whole with a glass of water.
The capsule can be taken with or without food.
Use the cream thinly on the external irritated area 2-3 times a day to relieve the symptoms for as long as they last, usually not more than 3 days.
Do not give CANESORAL® DUO to children under 18 years of age except under doctor supervision.
If you use too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone: Australia 13 11 26 or New Zealand 0800 764 766 [0800 POISON]) for advice, or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have used too much CANESORAL® DUO.
Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are using CANESORAL® DUO
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are using CANESORAL® DUO.
Tell any other doctors, dentists and pharmacists who treat you that you are using this medicine.
Use effective contraception to prevent pregnancy while using CANESORAL® DUO.
If you become pregnant while using this medicine, tell your doctor immediately.
If your symptoms do not improve after 3 days, tell your doctor or pharmacist.
Things you must not do
Do not use CANESORAL® DUO to treat any other complaints unless your doctor or pharmacist tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Things to be careful of
Tell your doctor immediately if you develop a rash while using CANESORAL® DUO.
People with HIV, AIDS or a weak immune system may be more prone to serious side effects of the skin.
Be careful when driving vehicles or operating machinery as occasional dizziness or seizures may occur.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using CANESORAL® DUO. This medicine helps most people and is generally well tolerated. However it may have unwanted side effects in some people.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Do not be alarmed by the following list of possible side effects.
You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- nausea (feeling sick), vomiting
- stomach pain, indigestion
- diarrhoea
- acne
- headache
- rash
- itching.
The above list includes the more common side effects of your medicine. They are usually mild and short-lived.
Tell your doctor as soon as possible if you notice any of the following:
- unusual muscle stiffness causing poor control of movement
- signs of frequent or worrying infections such as fever, severe chills, sore throat or mouth ulcers
- swelling of the face, lips or tongue which may cause difficulty in swallowing or breathing
- asthma, wheezing, shortness of breath
- sudden or severe itching, skin rash, hives
- fainting, seizures or fits
- flaking of the skin
- bleeding or bruising more easily than normal, reddish or purplish blotches under the skin
- passing more urine than normal, kidney pain (pain on the sides of the body)
- symptoms of liver disease such as yellowing of the skin or eyes; dark urine, pale stools; loss of appetite; unusual tiredness
- irregular heart beat or palpitations
- increased sweating.
The above list includes serious side effects that may require medical attention. Serious side effects are rare.
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
After using CANESORAL® DUO
Storage
Keep your capsule and cream in the pack until it is time to use them. If you take the capsule or cream out of the pack it may not keep well.
Keep CANESORAL® DUO in a cool dry place where the temperature stays below 25°C.
Do not store CANESORAL® DUO or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor or pharmacist tells you to stop using this medicine or its expiry date has passed, ask your pharmacist what to do with it.
Product description
What CANESORAL® DUO looks like
CANESORAL® DUO contains two components:
- CANESORAL® is a hard white gelatin capsule, marked with “CAN 150” in black ink.
Each pack contains 1 capsule. - CANESTEN® CLOTRIMAZOLE CREAM is an aluminium tube with a screw-cap containing 10 g of a white opaque cream.
Ingredients
The active ingredient in the CANESORAL® capsule is fluconazole 150 mg.
Each capsule also contains the following inactive ingredients:
- gelatin
- lactose monohydrate
- maize starch
- colloidal anhydrous silica
- magnesium stearate
- sodium lauryl sulfate
- titanium dioxide (E171)
- Opacode S-1-17823 black printing ink.
The active ingredient in CANESTEN® CLOTRIMAZOLE CREAM is 10 mg clotrimazole per gram of cream as the active ingredient.
The cream also contains:
- sorbitan monostearate
- polysorbate 60
- cetyl palmitate
- cetostearyl alcohol
- octyldodecanol
- benzyl alcohol
- purified water.
This medicine does not contain sucrose, gluten, tartrazine or any other azo dyes.
Supplier/Distributor
CANESORAL® DUO is distributed in Australia by:
Bayer Australia Limited
875 Pacific Highway
Pymble NSW 2073
Toll Free: 1800 008 757
www.canesten.com.au
CANESORAL® DUO is distributed in New Zealand by:
Bayer New Zealand Limited
Auckland, New Zealand
Toll Free: 0800 229 376
www.canesten.co.nz
Code: GO/DRUGS/361
® = Registered trademark of Bayer AG
AUST R 203513
This leaflet was prepared in August 2019.
Published by MIMS July 2022


 There have been no reported drug interactions with Canesten clotrimazole cream.
There have been no reported drug interactions with Canesten clotrimazole cream. For the most recent susceptibility testing interpretation according to the European committee on antimicrobial susceptibility testing (EUCAST), antifungal agents, please see (https://www.eucast.org/).
For the most recent susceptibility testing interpretation according to the European committee on antimicrobial susceptibility testing (EUCAST), antifungal agents, please see (https://www.eucast.org/).
 Fluconazole is a white to off-white crystalline powder, which is sparingly soluble in water and saline.
Fluconazole is a white to off-white crystalline powder, which is sparingly soluble in water and saline. Clotrimazole is a colourless, crystalline, weakly alkaline substance, melting point 141-145°C, soluble in acetone, chloroform and ethanol and practically insoluble in water. It forms stable salts with both organic and inorganic acids. It is not photosensitive but is slightly hygroscopic and may be hydrolysed in acid media.
Clotrimazole is a colourless, crystalline, weakly alkaline substance, melting point 141-145°C, soluble in acetone, chloroform and ethanol and practically insoluble in water. It forms stable salts with both organic and inorganic acids. It is not photosensitive but is slightly hygroscopic and may be hydrolysed in acid media.