What is in this leaflet
This leaflet answers some common questions about Canesten® Clotrimazole Thrush 1 Day Pessary & Cream.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your pharmacist has weighed the risks of you taking Canesten® Clotrimazole Thrush 1 Day Pessary & Cream against the benefits they expect it will have for you.
If you have any concerns about using this treatment, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Canesten® Clotrimazole Thrush 1 Day Pessary & Cream is used for
The pessary is used to treat a fungal infection in the vagina and vulval area (thrush). The cream works to relieve the symptoms of external irritation.
This medicine belongs to a group of medicines called azole antifungals.
What is vaginal thrush?
Candida albicans is a yeast fungus that can multiply rapidly and cause symptoms in the vagina when other micro-organisms aren’t present in a normal balance.
Some situations which can increase susceptibility to thrush are:
- Diabetes
- Obesity
- Illness
- Fatigue
- Hormonal fluctuations (pregnancy, menstrual cycle, oral contraceptive pill, HRT)
- Prolonged use of antibiotics or steroids
Thrush may cause the following symptoms:
- Vaginal itching
- Vaginal soreness
- A white odourless discharge from the vagina (like cottage cheese)
- Pain during intercourse
The symptoms below are not usually signs of thrush:
- Abnormal or irregular vaginal bleeding
- Blood stained discharge
- Vulva or vaginal sores, ulcers or blisters
- Lower abdominal pain
- Pain when passing urine
- Fever or chills
- Foul smelling and/or unusual coloured discharge
See your doctor if you experience any of the above symptoms.
Ask your doctor or pharmacist if you have any questions about whether this medicine is suitable for you.
This medicine is available without a doctor’s prescription, only through your pharmacist.
Before you use Canesten® Clotrimazole Thrush 1 Day Pessary & Cream
When you must not use it
Do not use Canesten® Clotrimazole Thrush 1 Day Pessary & Cream if you have an allergy to:
- any medicine containing clotrimazole
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include swelling to the area to which treatment has been applied; rash, itching or hives on the skin.
Do not use during your period as it may be less effective.
Do not use this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start using this medicine, talk to your doctor or pharmacist.
Before you start to use it
See your doctor if:
- you are under the age of 18 years or over the age of 60 years
- you are unsure whether you have thrush or this is the first time you have had thrush (see What is vaginal thrush?)
- you have had 3 or more infections in the last 6 months
- your symptoms include pain, fever or a foul smelling, unusual coloured or blood stained discharge
- you have allergies to any other topical treatments
Tell your doctor if you are pregnant, or think you might be. You should see your doctor if you suspect you have thrush during your pregnancy. Your doctor can discuss with you the risks and benefits involved with treatment. To treat internal thrush, your doctor may recommend that you use the pessary without the help of an applicator.
Tell your doctor if you are breastfeeding and want to use this treatment. It is not known whether the active ingredient, clotrimazole, is excreted in breast milk. Your doctor will be able to discuss with you the risks and benefits involved with treatment.
Tell your doctor if you are diabetic. Thrush in diabetics can mean that your blood sugar levels are not well controlled. Your doctor will be able to check this for you and advise treatment.
If you have not told your doctor about any of the above, tell him/her before you start using Canesten® Clotrimazole Thrush 1 Day Pessary & Cream.
Taking other medicines
Tell your doctor or pharmacist if you are taking or using any other medicines, including any that you buy without a prescription from a pharmacy, supermarket or health food shop.
Tell your doctor or pharmacist if you are taking tacrolimus (e.g. Prograf®), a medicine used to prevent organ transplant rejection or to treat certain problems with the immune system.
How to use Canesten® Clotrimazole Thrush 1 Day Pessary & Cream
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions below, or on the box, ask your doctor or pharmacist for help.
How to use it
Wash your hands thoroughly before handling the foil blister pack, cream or the applicator, and again afterwards when you have finished using the applicator
To avoid the possibility of re-infection, strict personal hygiene must be observed.
Pessary
The pessary should preferably be inserted in the evening, just before bed time, with the use of the applicator. Using it before you go to sleep will have the best effect.
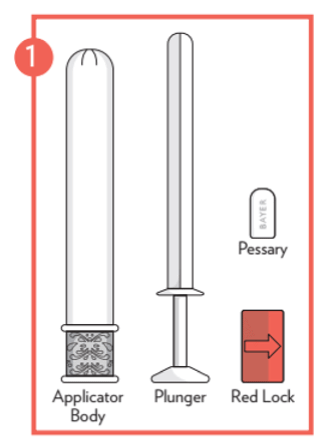
- Remove the applicator from the packaging and pull out the plunger and red lock from the body of the applicator.
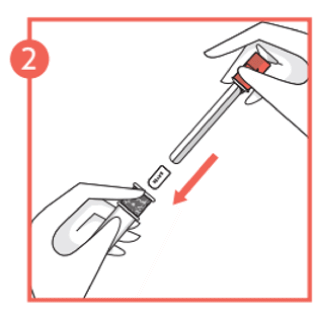
- Remove the pessary from the foil blister pack and place into the open end of the applicator body with the curved edge of the pessary facing down. Push the plunger and lock into the applicator body until you feel a click.
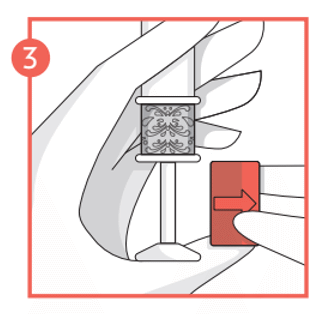
- Once you have felt the click, remove the lock from the plunger and then carefully insert the applicator as deep as is comfortable into the vagina up to the patterned grip zone. This is easiest when lying on your back with your knees bent up.
- Hold the applicator body at the patterned grip zone and slowly push the plunger until it stops. This places the pessary in the vagina.
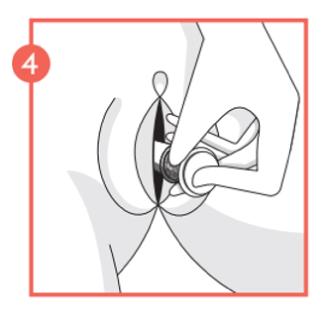
- Remove the applicator from the vagina. Discard the applicator. The applicator cannot be flushed down the toilet.
The pessary needs moisture in the vagina to dissolve completely, otherwise undissolved pieces of the pessary might crumble out of the vagina. To prevent this it is important to insert the medication as deeply as possible into the vagina at bedtime. Should the pessary not dissolve completely within one night the use of a vaginal cream should be considered.
Anti-fungal cream
Before use, pierce the tube seal by inverting the cap over the end of the tube and press.
Apply the cream thinly to the external irritated area 2-3 times a day to relieve the symptoms of itching.
How long to use it
Insert one pessary, once.
Continue using the cream for as long as you experience itching or your doctor or pharmacist tells you to.
This treatment helps to relieve your condition. It is important to keep using it until the fungal infection is gone.
If you are not sure what to do, ask your doctor or pharmacist.
DO NOT TAKE THE PESSARY OR CREAM BY MOUTH.
Seek medical advice if you or anyone else has taken Canesten® Clotrimazole Thrush 1 Day Pessary & Cream by mouth. Do this even if there are no signs of discomfort or poisoning. You may need medical attention.
While you are using Canesten® Clotrimazole Thrush 1 Day Pessary & Cream
Things you must do
Discontinue treatment and tell your doctor or pharmacist if you develop a local reaction to this product.
See your doctor if the symptoms do not improve within 4 days or if the problem returns within 2 months.
Things you must not do
Do not take Canesten® Clotrimazole Thrush 1 Day Pessary & Cream to treat any other complaints unless your doctor tells you to.
Do not give your treatment to anyone else, even if you suspect they have the same condition as you.
Things to be careful of
Canesten® Clotrimazole Thrush 1 Day Pessary & Cream may reduce the effectiveness and safety of latex products, such as condoms and diaphragms, if it comes into contact with them. Therefore you should prevent Canesten® Clotrimazole Thrush 1 Day Pessary & Cream from coming into contact with these products. If Canesten® Clotrimazole Thrush 1 Day Pessary & Cream does accidently come into contact with a condom or diaphragm, replace the condom or diaphragm before using it as contraception.
Do not use tampons, intravaginal douches, spermicides or other vaginal products while using this product.
Avoid vaginal intercourse while you have thrush and during use of this product because your partner could become infected.
Things that may help to resolve Thrush
Some self help measures suggested below may help your condition. Your doctor or pharmacist can give you more information about these measures.
- Blot with toilet paper after urinating (instead of wiping back to front).
- Change underwear, night attire, towels and linen daily.
Avoid where possible:
- Perfumed toilet paper
- Fragrant soaps and bath oils
- Wearing tight or synthetic clothing
- Douches or vaginal deodorants
- Soaking in baths
Side effects
Tell your doctor or pharmacist if you experience discomfort while you are using Canesten® Clotrimazole Thrush 1 Day Pessary & Cream.
This medicine helps most people with thrush but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following list of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
After you use the pessary you might experience:
- Itching, rash, swelling, redness, discomfort, burning, vaginal peeling, discharge or bleeding
- Pain (e.g. abdominal pain, vulvovaginal pain)
- Nausea
After you apply the cream you might experience:
- Itching, rash, blisters, burning, discomfort, swelling, redness, or peeling of skin.
If you experience any of the above effects, tell your doctor or pharmacist immediately.
Tell your doctor immediately or go to Accident and Emergency at your nearest hospital if you experience any of the following:
- Rash
- Swallowing or breathing problems
- Swelling of your lips, face, throat or tongue
- Nausea, weakness, feeling dizzy or faint
Any of the above may be signs of an allergic reaction. Your doctor or pharmacist may recommend you discontinue treatment if you experience these effects.
After using Canesten® Clotrimazole Thrush 1 Day Pessary & Cream
Storage
Keep the pessary in the protective blister pack until it is time to use it.
Keep the cream in the tube until it is time to use it. If you take the cream out of the tube it may not keep well.
Keep your pessary and cream in a cool dry place where the temperature stays below 25°C.
Keep the treatment where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Expiry
Pessary: The expiry date is marked on the end panel of the carton and is also printed on the back of the blister. The date refers to the month and year of expiration e.g. 0422 refers to the fourth month of 2022. The product can be used until the end of this month.
Cream: The expiry date is marked on the end panel of the carton and is also printed on the crimp at the end of the tube. The date refers to the month and year of expiration e.g. 0422 refers to the fourth month of 2022. The product can be used until the end of this month.
Disposal
If your doctor tells you to stop using this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Discard the medicine out of the reach of children.
Product description
What it looks like
The pessary is a white pellet-shaped pessary packaged in an aluminium blister.
The package also contains 1 plastic applicator for the insertion of the pessary. The plastic applicator is for use with the Canesten pessary only. Discard applicator after use.
The cream is a white opaque cream presented in a screw –cap tube containing 10 g of cream.
Ingredients
Canesten® Clotrimazole Thrush 1 Day Pessary & Cream
The pessary contains 500 mg clotrimazole as the active ingredient. It also contains:
- Lactose monohydrate
- Microcrystalline cellulose
- Lactic acid
- Maize starch
- Crospovidone
- Calcium lactate pentahydrate
- Magnesium stearate
- Colloidal anhydrous silica
- Hypromellose
The cream contains 10 mg clotrimazole per gram of cream as the active ingredient. It also contains:
- Sorbitan monostearate
- Polysorbate 60
- Cetyl palmitate
- Cetostearyl alcohol
- Octyldodecanol
- Benzyl alcohol
- Purified water
How to Recycle

Supplier/Distributor
Canesten® Clotrimazole Thrush 1 Day Pessary & Cream is distributed in Australia by:
Bayer Australia Ltd
875 Pacific Highway
Pymble NSW 2073
Freecall: 1800 008 757
www.canesten.com.au
Canesten® Clotrimazole Thrush 1 Day Pessary & Cream is distributed in New Zealand by:
Bayer New Zealand Limited
Auckland, New Zealand
Freecall: 0800 229 376
www.canesten.co.nz
® = Registered Trademark
AUST R number 92426
This leaflet was prepared in April 2022.

Published by MIMS April 2026

 Molecular formula: C22H17ClN2.
Molecular formula: C22H17ClN2.