What is in this leaflet
This leaflet answers some common questions about Carmustine powder for injection.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
The information in this leaflet was last updated on the date listed on the final page. More recent information on the medicine may be available.
You should ensure that you speak to your pharmacist or doctor to obtain the most up-to-date information on the medicine. Those updates may contain important information about the medicine and its use which you should be aware.
All medicines have risks and benefits. Your doctor has weighed the risks of you being given this medicine against the benefits they expect it will have for you.
If you have any concerns about being given this medicine, talk to doctor or pharmacist.
Keep this leaflet. You may need to read it again.
What Carmustine powder for injection is given for
Carmustine is used to treat malignant glioma, a type of brain cancer, and multiple myeloma, a cancer of the blood. It may also be used to treat other types of cancers called Hodgkin's Disease and Non- Hodgkin's lymphomas.
Carmustine belongs to a group of medicines called cytotoxic medicines. You may also hear of these being called chemotherapy medicines. Carmustine may be used in combination with other medicines to treat your cancer.
Your doctor may have prescribed Carmustine for another use. Ask your doctor if you have any questions about why Carmustine was prescribed.
This medicine is only available with a doctor's prescription.
How Carmustine powder for injection works
Carmustine works by killing cancer cells.
The use of Carmustine to treat your condition can lead to side-effects, which are discussed below.
Before you are given Carmustine powder for injection
When you must not have this medicine
You should not have Carmustine powder for injection if you have a history of severe allergic reactions to carmustine or any of the ingredients listed at the end of this leaflet.
You should not be given Carmustine powder for injection if you have, or have had any of the following conditions unless you have discussed it with your doctor:
- liver problems
- kidney problems
- lung disease
- blood disorder with a reduced number of white blood cells
- blood disorder with a low blood platelet count
- blood disorder with a decreased number of red blood cells
- lowered immunity due to treatment with medicines such as corticosteroids, cyclosporin or other medicines used to treat cancer.
Tell your doctor if you have an infection or high temperature. Your doctor may decide to delay your treatment until the infection has gone. A mild illness, such as a cold, is not usually a reason to delay treatment.
If you are not sure whether you should be given this medicine, talk to your doctor or pharmacist.
Do not use this medicine after the expiry date (EXP) printed on the pack. If you are given it after the expiry date has passed, it may not work very well.
Do not use this medicine if the packaging is torn or shows signs of tampering or if the injection shows any visible signs of deterioration.
Before you start to have this medicine
Tell your doctor if you have allergies to
- any other medicines
- any other substances, such as foods, preservatives or dyes.
Females: Tell your doctor or pharmacist if you are pregnant or intend to become pregnant.
Like most cytotoxic medicines, Carmustine is not recommended for use during pregnancy. If there is any need to consider Carmustine during your pregnancy, your doctor will discuss with you the benefits and risks of using it. Carmustine may affect your developing baby if you take it during pregnancy.
Males: Tell your doctor if your partner intends to become pregnant while you are using Carmustine or shortly after you have stopped using Carmustine.
You should use some kind of birth control while you are using Carmustine and for at least 12 weeks after you stop using it. Carmustine may cause birth defects if either the male or female is using it at the time of conception.
Tell your doctor if you are currently breastfeeding or you plan to breast-feed. Do not breastfeed while using Carmustine. It is not known whether Carmustine passes into breast milk. Therefore, there is a possibility that the breast-fed baby may be affected.
Tell your doctor or pharmacist if you have or have had any medical conditions, especially the following:
- liver problems
- kidney problems
- heart problems, including a heart attack
- lung problems, including asthma
- blood disorder with a reduced number of red or white blood cells
- If you are pregnant or intend to become pregnant.
- If you are breast-feeding or intend to breast-feed.
If you have not told your doctor about any of the above, tell them before you have Carmustine powder for injection.
Taking other medicines
Tell your doctor if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Carmustine powder for injection may interfere with each other.
These medicines may be affected by carmustine,or may affect how well this medicine works or may increase side effects. You may need to use different amounts of the medicines or take different medicines.
Carmustine when used in combination with other medicines or radiation therapy may further depress your immune system.
Your doctor or pharmacist has more information on medicines to be careful with or avoid while using this medicine.
If you have not told your doctor about any of the above, tell them before you start having Carmustine.
How Carmustine powder for injection is given
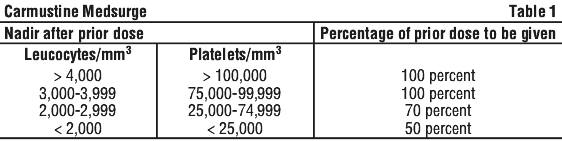
Carmustine is usually given every six weeks. This is called one cycle of chemotherapy. Your doctor will decide how many of these cycles you will need.
Several courses of Carmustine therapy may be needed depending on your response to treatment. Additional treatment may not be repeated until your blood cell numbers return to acceptable levels and any uncontrolled effects have been controlled.
Ask your doctor if you want to know more about the dose of Carmustine you receive.
How much medicine is given
Your doctor will decide what dose of Carmustine you will receive based in the characteristics of your cancer. The dose also depends on your condition and other factors, such as your weight, kidney function and other chemotherapy medicines you are being given. Carmustine may be given alone or in combination with other drugs.
How this medicine is given
Carmustine is administered as a slow infusion into your vein, over 1 to 2 hours. The infusion will be prepared and given in hospital by your doctor or nurse.
If you are given too much (overdose)
As Carmustine is given to you under the supervision of your doctor, it is very unlikely that you will receive too much. However, if you experience any side effects after being given Carmustine, tell your doctor or nurse immediately or go to Accident and Emergency at your nearest hospital. You may need urgent medical attention.
Symptoms of a Carmustine overdose include the side effects listed below in the "Side Effects" section but are usually of a more severe nature.
Ask your doctor or pharmacist if you have any concerns.
Immediately telephone your doctor, or the Poisons Information Centre (Australia: telephone 13 11 26) for advice or go to Accident and Emergency at your nearest hospital, if you think you or anyone else may have been given too much Carmustine powder for injection.
Do this even if there are no signs of discomfort or poisoning.
You may need urgent medical attention.
Keep telephone numbers for these places handy.
Side effects
Tell your doctor as soon as possible if you do not feel well while you are having Carmustine powder for injection.
Like all medicines, it is possible that Carmustine may have unwanted side effects in some people. During treatment with Carmustine you will require close medical supervision.
The more common side effects of Carmustine are:
- Suppression of your immune system; reduced white blood cells, reduced platelets, reduced red blood cells
- Nausea, vomiting
- Lung toxicity
- Respiratory disorders (lung related disorders) with breathing problems.
Tell your doctor or nurse immediately or if you are not currently in hospital, go to accident and emergency at your nearest hospital if you notice any of the following:
- Symptoms of an allergic reaction, such as:
- shortness of breath, wheezing, difficulty breathing or a tight feeling in your chest
- swelling of the face, lips, tongue or other parts of the body
- rash, itching, hives or flushed, red skin
- dizziness or light headedness. - Frequent infections, fever, severe chills, sore throat or mouth ulcers
- Bleeding or bruising more easily than normal
- Passing little or no urine, drowsiness, nausea, vomiting and breathlessness
- Weakness, tiredness, loss of appetite, weight loss and stomach pain
- Changes to vision or loss of sight
- Shortness of breath, particularly with exertion, chronic dry, hacking coughing
- Chest discomfort, irregular heartbeat
- Loss of appetite and rapid weight loss
- Pain, swelling or burning at the injection site.
These are serious side effects. You may need urgent medical attention or hospitalisation.
Other side effects not listed above may also occur in some patients. Tell your doctor if you notice anything else that is making you feel unwell.
After having it
Storage
Carmustine powder for injection will be stored in the pharmacy or on the ward. The unopened vials of the dry powder will be stored under refrigeration (2°C - 8°C) (Refrigerate. Do not freeze).
Product description
What it looks like
Carmustine is presented in composite packs in.
Pack of 1s:
Each carton pack consists of a vial containing 100 mg carmustine and a vial containing a 3 mL ethanol absolute sterile diluent.
Ingredients
Active ingredient:
- carmustine 100 mg/vial.
Other ingredient:
- ethanol absolute 3 mL/vial (required for preparation of the infusion).
Manufacturer / Sponsor
Australian Sponsor
Medicianz Healthcare Pty Ltd
Unit 2, 6-7 Gilda Court
MULGRAVE
VICTORIA 3170
Marketed and distributed by Medsurge Healthcare.
Tel: 1300 788 261
www.medsurge.com.au
Registration Number:
Carmustine Medsurge 100mg (powder for injection with diluent):
AUST R 341128
This leaflet was updated in August 2021.
Published by MIMS November 2021