1 Name of Medicine
Cefazolin sodium.
2 Qualitative and Quantitative Composition
Cefazolin-AFT is supplied in vials containing cefazolin sodium equivalent to 500 mg, 1 g and 2 g cefazolin.
The sodium content is 50 mg/g of cefazolin sodium.
Cefazolin sodium is a white to almost white crystalline powder with a solubility in water of greater than or equal to 100 mg/mL. Cefazolin-AFT powder for injection contains cefazolin sodium as a single ingredient.
For the full list of excipients, see Section 6.1.
3 Pharmaceutical Form
White to almost white powder for injection which reconstitutes with Sterile Water for Injection to give a colourless solution.
4.1 Therapeutic Indications
Treatment of the following serious infections due to susceptible organisms.
Respiratory tract infections due to Strep. pneumoniae, Klebsiella sp., H. influenzae, Staph. aureus (penicillin sensitive and penicillin resistant) and group A beta-haemolytic Streptococci. Injectable benzathine penicillin is considered to be the drug of choice in the treatment and prevention of streptococcal infections, including the prophylaxis of rheumatic fever. Cefazolin is effective in the eradication of Streptococci from the nasopharynx; however, data establishing the efficacy of cefazolin in the subsequent prevention of rheumatic fever are not available at present.
Genitourinary tract infections due to E. coli, P. mirabilis, Klebsiella sp. and some strains of Enterobacter and enterococci.
Skin and skin structure infections due to Staph. aureus (penicillin sensitive and penicillin resistant) and group A beta-haemolytic Streptococci and other strains of Streptococci.
Bone and joint infections due to Staph. aureus.
Septicaemia due to Strep. pneumoniae, Staph. aureus (penicillin sensitive and penicillin resistant), P. mirabilis, E. coli and Klebsiella sp.
Endocarditis due to Staph. aureus (penicillin sensitive and penicillin resistant) and group A beta-haemolytic Streptococci.
Appropriate culture and susceptibility studies should be performed to determine susceptibility of the causative organism to cefazolin.
4.2 Dose and Method of Administration
Dosage.
Adults. Usual dosage for mild Gram positive infections is cefazolin 250 to 500 mg every eight hours.
In mild to moderate infections of the respiratory tract caused by Strep. pneumoniae, or mild to moderate infections of the genitourinary tract caused by susceptible organisms, a dosage of 500 mg to 1 g every 12 hours may be used.
In moderate or severe infections, the usual adult dosage is cefazolin 500 mg to 1 g every six to eight hours. Cefazolin has been administered in dosages of 6 g/day in serious infections such as endocarditis.
Renal impairment.
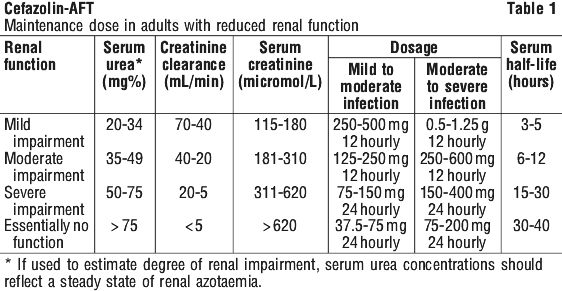
In patients with renal impairment, cefazolin is not readily excreted. After a loading dose of 500 mg, the recommendations in Table 1 for maintenance dosage may be used as a guide.
 Children. A total daily dosage of 25 to 50 mg/kg bodyweight, divided into three or four equal doses, is effective for most mild to moderately severe infections. Total daily dosage may be increased to 100 mg/kg bodyweight for severe infections.
Children. A total daily dosage of 25 to 50 mg/kg bodyweight, divided into three or four equal doses, is effective for most mild to moderately severe infections. Total daily dosage may be increased to 100 mg/kg bodyweight for severe infections.
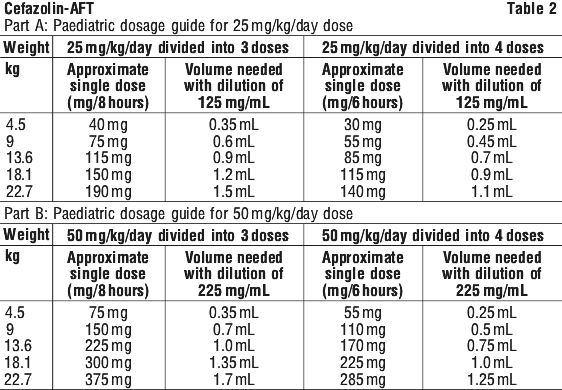
Infants. Since safety for use in premature infants and in infants aged under one month has not been established, the use of cefazolin in these patients is not recommended (see Table 2).
Renal impairment.
In children with mild to moderate impairment of renal function (creatinine clearance of 70 to 40 mL/minute), 60% of the normal daily dose given in divided doses every 12 hours should be sufficient. In patients with moderate impairment (creatinine clearance of 40 to 20 mL/minute), 25% of the normal daily dose given in divided doses every 12 hours should be sufficient. In children with marked impairment (creatinine clearance of 20 to 5 mL/minute), 10% of the normal daily dose given every 24 hours should be adequate. All dosage recommendations apply after an initial loading dose.
Method of administration.
Cefazolin may be administered intramuscularly or intravenously after reconstitution. The intrathecal administration of cefazolin is not an approved route of administration for this antibiotic; in fact, there have been reports of severe CNS toxicity including seizures when cefazolin was administered in this manner.
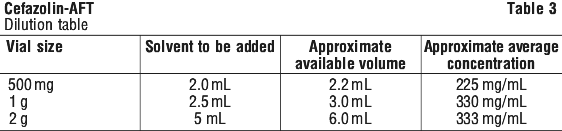
Intramuscular administration. Reconstitute with water for injections or sodium chloride 0.9% injection according to the dilution table (see Table 3). Shake well until dissolved. To facilitate putting the product into solution, the vial should be warmed in the hands while shaking. Do not use the reconstituted injection solution if there is any sign of turbidity. Cefazolin should be injected into a large muscle mass.
 Intravenous administration. Cefazolin may be administered by direct intravenous injection or by intermittent or continuous infusion. Total daily dosages are the same as with intramuscular injection.
Intravenous administration. Cefazolin may be administered by direct intravenous injection or by intermittent or continuous infusion. Total daily dosages are the same as with intramuscular injection.
Direct intravenous injection.
Dilute the reconstituted 500 mg, 1 g or 2 g Cefazolin-AFT powder for injection in a minimum of 10 mL of water for injections. Inject solution slowly over three to five minutes. It may be administered directly into a vein or via the giving line for a patient receiving a compatible IV solution. Cefazolin sodium has been reported to be compatible with the following IV fluids:
0.9% sodium chloride injection;
5% or 10% dextrose injection;
5% Dextrose in Lactated Ringer's injection;
5% dextrose and 0.9% sodium chloride injection (also may be used with 5% dextrose and 0.2% sodium chloride injection);
Lactated Ringer's injection;
Plasma-lyte with 5% dextrose;
Ringer's injection;
Sterile water for injection.
Intermittent intravenous infusion.
Cefazolin sodium can be administered along with primary IV fluid management programmes in a volume control set or in a separate, secondary intravenous infusion bottle. Reconstituted cefazolin 500 mg, 1 g or 2 g may be diluted in 50 to 100 mL of water for injections or one of the previously listed parenteral fluids, and infused over a period of three to five minutes. If a Y-type administration set is used, it is desirable to discontinue the other solution during the infusion of the solution containing cefazolin sodium.
Continuous intravenous infusion.
The total daily dose of cefazolin, diluted and well mixed with at least 50 mL of water for injections, may be added to an intravenous bottle containing one of the previously listed parenteral fluids. The choice of saline or glucose solution and the volume to be employed are dictated by fluid and electrolyte management.
Stability.
Cefazolin-AFT injections reconstituted in the recommended IV fluids are stable for 24 hours if stored under refrigeration (2-8°C). To minimise the risk of microbial contamination and growth, the preferred practice is to use the reconstituted product as soon as practical after reconstitution and to discard any remaining residue. If storage of the reconstituted solution is necessary, hold at 2 to 8°C for not more than 24 hours.4.3 Contraindications
Known allergy to the cephalosporin group of antibiotics or previous experience of a major allergy to penicillin (see Section 4.4 Special Warnings and Precautions for Use).
Lidocaine (lignocaine) should not be used as a diluent for intramuscular injection in patients who are hypersensitive to lidocaine (lignocaine).
4.4 Special Warnings and Precautions for Use
Before cefazolin therapy is instituted, careful inquiry should be made concerning previous hypersensitivity reactions to cephalosporins and penicillin. Cephalosporin C derivatives should be given cautiously in penicillin sensitive patients. Serious acute hypersensitivity reactions may require adrenaline and other emergency measures. There is some clinical and laboratory evidence of partial cross allergenicity of the penicillins and the cephalosporins. Patients have been reported to have had severe reactions (including anaphylaxis) to both drugs. Antibiotics, including cefazolin, should be administered cautiously to any patient who has demonstrated some form of allergy, particularly to drugs. If an allergic reaction to cefazolin occurs, the drug should be discontinued and the patient treated with the usual agents (e.g. adrenaline or other pressor amines, antihistamines or corticosteroids).
Prolonged use of cefazolin may result in the overgrowth of nonsusceptible organisms. Careful clinical observation of the patient is essential. If superinfection occurs during therapy, appropriate measures should be taken.
Antibiotic associated pseudomembranous colitis has been reported with many antibiotics including cefazolin. A toxin produced by Clostridium difficile appears to be the primary cause. The severity of the colitis may range from mild to life threatening. It is important to consider this diagnosis in patients who develop diarrhoea or colitis in association with antibiotic use (this may occur up to several weeks after cessation of antibiotic therapy). Mild cases usually respond to drug discontinuation alone. However, in moderate to severe cases, appropriate therapy with a suitable oral antibacterial agent effective against Clostridium difficile should be considered. Fluids, electrolytes and protein replacement should be provided when indicated.
Drugs that delay peristalsis, e.g. opiates and diphenoxylate with atropine (e.g. Lomotil), may prolong and/or worsen the condition and should not be used.
Prolonged use of cefazolin may result in the overgrowth of non-susceptible organisms. Careful clinical observation of the patient is essential. If superinfection occurs during therapy, appropriate measures should be taken.
The intrathecal administration of cefazolin is not an approved route of administration for this antibiotic; in fact, there have been reports of severe CNS toxicity including seizures when cefazolin was administered in this manner.
The intrathecal administration of cefazolin is not an approved route of administration for this antibiotic; in fact there have been reports of tremulousness, headache, agitation, lightheadedness and sensations of seeing flashing lights when cefazolin was administered in this manner for the treatment of infected ventricular shunts.
Severe cutaneous adverse reactions.
Severe cutaneous adverse reactions (SCAR), such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalised exanthematous pustulosis (AGEP) have been reported in patients taking beta-lactam antibiotics. When SCAR is suspected, Cefazolin-AFT should be discontinued immediately and an alternative treatment should be considered.
History of gastrointestinal disease.
Cefazolin, as with all cephalosporins, should be prescribed with caution in individuals with a history of gastrointestinal disease.
Use in renal impairment.
As with other beta-lactam antibiotics, seizures may occur if inappropriately high doses are administered to patients with impaired renal function. When cefazolin is administered to patients with low urinary output because of impaired renal function, lower daily dosage is required (see Section 4.2 Dose and Method of Administration).
Encephalopathy has been reported with the use of cefazolin in patients with renal failure (see Section 4.8 Adverse Effects (Undesirable Effects)). The symptoms have included tonic-clonic seizures, lethargy, disorientation, memory loss, asterixis and multifocal myoclonus. Toxicity has been attributed to increased cefazolin serum levels and increased permeability of the blood brain barrier caused by uraemia. The dose of cefazolin should be reduced or the dosing interval increased in patients with renal failure.
Neurotoxicity.
There have been reports of neurotoxicity associated with cephalosporin treatment. Symptoms of neurotoxicity include seizures and/or myoclonus. Risk factors for developing neurotoxicity with cephalosporin treatment include being elderly, renal impairment, central nervous system disorders and intravenous administration. Withdrawal of the medicine should be considered if there are signs of neurotoxicity.
Use in the elderly.
No data available.
Paediatric use.
Safety for use in premature infants and infants under 1 month of age has not been established.
Effects on laboratory tests.
A false positive reaction for glucose in the urine may occur with Benedict's solution, Fehling's solution or with Clinitest tablets, but not with enzyme based tests, such as Clinistix and Tes-Tape. Positive direct and indirect antiglobulin (Coombs') tests have occurred; these may also occur in neonates whose mothers received cephalosporins before delivery.4.5 Interactions with Other Medicines and Other Forms of Interactions
Probenecid.
Probenecid may decrease renal tubular secretion of cephalosporins when used concurrently, resulting in increased and more prolonged cephalosporin blood levels.
Aminoglycoside antibiotics.
Coadministration of aminoglycoside antibiotics with cephalosporins could produce additive nephrotoxic effects. Use of these agents should be avoided in patients with prior renal insufficiency. If coadministration of these two antibiotic classes is necessary, patients should be monitored for evidence of nephrotoxicity.
Live typhoid vaccine.
Antibiotics which possess bacterial activity against Salmonella typhi organisms may interfere with the immunological response to the live typhoid vaccine. Allow 24 hours or more to elapse between the administration of the last dose of the antibiotic and the live typhoid vaccine.
Warfarin.
Patients receiving oral anticoagulant therapy with warfarin should be closely monitored using the prothrombin time ratio or international normalised ratio (INR) during concurrent therapy with cefazolin. Adjustment of the warfarin dosage to maintain the desired anticoagulant effect may be necessary. An alternative would be to use a cephalosporin which does not possess hypoprothrombinemic properties.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No data available.
(Category B1)
Safety of this product for use during pregnancy has not been established in human clinical trials. Studies in animals are inadequate or lacking, but available data show no evidence of an increased occurrence of fetal damage. Studies of cord blood show prompt transfer of cefazolin across the placenta. Drug levels in cord blood were approximately one-quarter to one-third maternal drug levels.
Cefazolin is present in very low concentrations in the milk of breastfeeding mothers. Caution should be exercised when cefazolin is administered to a breastfeeding mother.4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
The following reactions have been reported:
Hypersensitivity.
Drug fever, skin rash, vulvar pruritus, eosinophilia, itching and Stevens-Johnson syndrome have occurred.
Haematological.
The most common blood disorder associated with cefazolin has been eosinophilia. Neutropenia, leucopenia, thrombocythaemia, thrombocytopenia and positive direct and indirect Coombs' tests have occurred.
Hepatic and renal.
Isolated transient rise in AST, ALT, serum urea, and alkaline phosphatase levels has been observed without evidence of renal or hepatic impairment.
Gastrointestinal.
Nausea, anorexia, vomiting, diarrhoea and oral candidiasis (oral thrush) have been reported. As with other broad spectrum antibiotics, colitis, including rare instances of pseudomembranous colitis, has been reported in conjunction with therapy with cefazolin (see Section 4.4 Special Warnings and Precautions for Use).
Skin and other subcutaneous tissue disorders.
Severe cutaneous adverse reactions, including Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalised exanthematous pustulosis (AGEP) have been reported in beta-lactam antibiotics.
Other.
Pain at the site of injection after intramuscular administration has occurred, some with induration. Phlebitis at the site of injection has been noted. Other reactions have included genital and anal pruritus, genital candidiasis and vaginitis.
Post-marketing experience.
Nervous system disorders.
Seizures, encephalopathy, myoclonus.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
Signs and symptoms.
Toxic signs and symptoms following an overdose of cefazolin may include pain, inflammation and phlebitis at the injection site.
The administration of inappropriately large doses of parenteral cephalosporins may cause dizziness, paraesthesias and headaches. Seizures may occur following overdosage with some cephalosporins, particularly in patients with renal impairment, in whom accumulation is likely to occur.
Laboratory abnormalities may include elevations in creatinine, serum urea, liver enzymes and bilirubin, a positive Coombs' test, thrombocytosis, thrombocytopenia, eosinophilia, leucopenia and prolongation of the prothrombin time.
Treatment.
In managing overdosage, the possibility of multiple drug overdoses, interaction among drugs and unusual drug kinetics should be considered.
If seizures occur, the drug should be discontinued promptly; anticonvulsant therapy may be administered if clinically indicated. Protect the patient's airway and support ventilation and perfusion. Meticulously monitor and maintain, within acceptable limits, the patient's vital signs, blood gases, serum electrolytes, etc.
In cases of severe overdosage, especially in a patient with renal failure, combined haemodialysis and haemoperfusion may be considered if response to more conservative therapy fails. However, no data supporting such therapy are available.5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Semisynthetic cephalosporin for parenteral administration.
Microbiology.
In vitro tests demonstrate that the bactericidal action of cephalosporins results from inhibition of cell wall synthesis. Cefazolin is active against the following organisms in vitro: Staphylococcus aureus (penicillin sensitive and penicillin resistant); group A beta-haemolytic Streptococci and other strains of Streptococci (many strains of enterococci are resistant); Streptococcus pneumoniae, Escherichia coli, Proteus mirabilis, Klebsiella sp., Enterobacter aerogenes, Haemophilus influenzae. Most strains of Enterobacter cloacae and indole positive proteus (P. vulgaris, P. morganii, P. rettgeri) are resistant. Methicillin resistant Staphylococci, Serratia, Pseudomonas, Acinetobacter calcoaceticus (formerly mima and herellea sp.) are almost uniformly resistant to cefazolin.
Susceptibility tests.
Dilution or diffusion techniques - either quantitative (MIC) or breakpoint, should be used following a regularly updated, recognised and standardised method (e.g. NCCLS). Standardised susceptibility test procedures require the use of laboratory control microorganisms to control the technical aspects of the laboratory procedures.
A report of "Susceptible" indicates that the pathogen is likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable. A report of "Intermediate" indicates that the result should be considered equivocal, and if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where high dosage of drug can be used. This category also provides a buffer zone, which prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of "Resistant" indicates that the pathogen is not likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable, other therapy should be selected.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
Absorption.
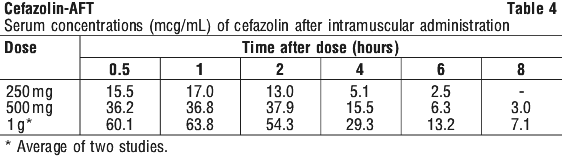
Table 4 demonstrates the blood levels and duration of cefazolin following intramuscular administration.
 Clinical pharmacology studies in patients hospitalised with infections indicate that cefazolin produces mean peak serum levels approximately equivalent to those seen in normal volunteers.
Clinical pharmacology studies in patients hospitalised with infections indicate that cefazolin produces mean peak serum levels approximately equivalent to those seen in normal volunteers.
In a study (using normal volunteers) of constant intravenous infusion with dosages of 3.5 mg/kg for one hour (approximately 250 mg) and 1.5 mg/kg for the next two hours (approximately 100 mg) cefazolin produced a steady serum level at the third hour of approximately 28 microgram/mL. Table 5 shows the average serum concentration after intravenous injection of a single 1 g dose; average half-life was 1.4 hours.

Distribution.
Cefazolin readily crosses an inflamed synovial membrane and the concentration of the antibiotic achieved in the joint space is comparable to levels measured in serum. Cefazolin readily crosses the placental barrier into the cord blood and amniotic fluid. Cefazolin is present in very low concentrations in the milk of breastfeeding mothers.
Metabolism.
Controlled studies on adult normal volunteers receiving 1 g four times daily for ten days, monitoring complete blood count, AST, ALT, bilirubin, alkaline phosphatase, serum urea, creatinine and urinalysis, indicated no clinically significant changes attributed to cefazolin.
Excretion.
Cefazolin is excreted unchanged in the urine. Following intramuscular injection of 500 mg, 56 to 89% of the administered dose was recovered within six hours and 80 to nearly 100% was recovered in 24 hours. Cefazolin achieves peak urine concentrations greater than 1,000 microgram/mL and 4,000 microgram/mL, respectively, following 500 mg and 1 g intramuscular doses. When cefazolin is administered to patients with unobstructed biliary tracts, high concentrations, well over serum levels, occur in the gall bladder tissue and bile. In the presence of obstruction, however, concentration of the antibiotic in bile is considerably lower than the serum level.
5.3 Preclinical Safety Data
Genotoxicity.
No data available.
Carcinogenicity.
Long-term studies in animals to determine the carcinogenic potential of cefazolin have not been performed.6 Pharmaceutical Particulars
6.1 List of Excipients
Cefazolin-AFT powder for injection contains cefazolin sodium as a single ingredient.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
Cefazolin-AFT must not be reconstituted with other products except those mentioned in Section 4.2 Dose and Method of Administration.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Cefazolin-AFT 500 mg and 1 g: Store below 25°C.
Cefazolin-AFT 2 g: Store below 30°C.
Protect from light.
Reconstituted solution: Store at 2°C to 8°C. Refrigerate. Do not freeze. (Use within 24 hours after initial reconstitution.) To reduce microbiological hazard, use as soon as practicable after dilution. If storage is necessary, hold at 2-8°C for not more than 24 hours. Product is for single use in one patient only. Discard any residue.
6.5 Nature and Contents of Container
This powder for injection is supplied in vials containing cefazolin sodium equivalent to 500 mg,1 g and 2 g cefazolin.
Cefazolin-AFT is supplied in type I (500 mg and 1 g) or type II glass vials (500 mg, 1 g and 2 g) sealed with film-coated butyl rubber stopper.
Available in packs of 1, 5 and 10 vials.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
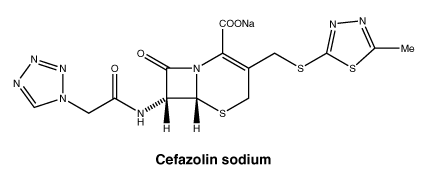 Chemical name: sodium (6R, 7R)-3-[[5-methyl-1,3,4- thiadiazol-2-yl)sulphanyl]methyl]-8- oxo-7-[(1H-tetrazol-1-ylacetyl)amino]-5- thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate.
Chemical name: sodium (6R, 7R)-3-[[5-methyl-1,3,4- thiadiazol-2-yl)sulphanyl]methyl]-8- oxo-7-[(1H-tetrazol-1-ylacetyl)amino]-5- thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate.
Molecular formula: C14H13N8NaO4S3.
Molecular weight: 476.5.
CAS number.
27164-46-1.7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Summary Table of Changes





 Clinical pharmacology studies in patients hospitalised with infections indicate that cefazolin produces mean peak serum levels approximately equivalent to those seen in normal volunteers.
Clinical pharmacology studies in patients hospitalised with infections indicate that cefazolin produces mean peak serum levels approximately equivalent to those seen in normal volunteers.
 Chemical name: sodium (6R, 7R)-3-[[5-methyl-1,3,4- thiadiazol-2-yl)sulphanyl]methyl]-8- oxo-7-[(1H-tetrazol-1-ylacetyl)amino]-5- thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate.
Chemical name: sodium (6R, 7R)-3-[[5-methyl-1,3,4- thiadiazol-2-yl)sulphanyl]methyl]-8- oxo-7-[(1H-tetrazol-1-ylacetyl)amino]-5- thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate.