What is in this leaflet
This leaflet answers some common questions about Celebrex. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking Celebrex against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Celebrex is used for
Celebrex is used to relieve the symptoms of joint pain, tenderness, swelling and stiffness in:
- osteoarthritis
- rheumatoid arthritis
- ankylosing spondylitis, a chronic inflammatory rheumatic disorder that primarily affects, but is not limited to, the spine.
Celebrex also provides short-term pain relief in conditions such as:
- menstrual cramps or period pain
- after surgery
- muscle and joint injuries.
Celebrex contains celecoxib and belongs to a group of medicines called coxibs which are used to relieve pain and inflammation in a number of conditions.
Although Celebrex can relieve the symptoms of pain and inflammation, it will not cure your condition.
Your doctor, however, may have prescribed Celebrex for another purpose.
Ask your doctor if you have any questions about why Celebrex has been prescribed for you.
Celebrex is not recommended for use in children or adolescents under 18 years of age.
This medicine is only available with a doctor's prescription.
Before you take Celebrex
When you must not take it
Do not take Celebrex if you
- suffer from chest pains or angina and they occur even when you are resting and are becoming more frequent, severe, or lasting longer than usual
- have or have had problems with your blood circulation
- have or if your doctor has told you that severe heart or blood vessel disease affecting the circulation in your brain or limbs.
- have severe liver problems
Your doctor will decide if your condition is too severe to take this medicine. - have problems with your kidney function
- are undergoing cardiac surgery called coronary artery bypass graft (CABG)
- have had an attack of asthma, hives, itching, skin rash or a runny nose after taking aspirin or Non-Steroidal Anti-Inflammatory Drugs (NSAIDs, medicines used to treat pain and inflammation), including other Coxib medicines.
Many medicines used to treat headache, period pain and other aches and pains contain aspirin or an NSAID.
If you are allergic to aspirin, NSAIDs, or other Coxib medicines and take Celebrex, these symptoms may be severe. - have an allergy to:
- celecoxib
- any of the ingredients listed at the end of this leaflet
- sulfonamides, a group of medicines which include, for example, certain antibiotics (if you are not sure if you are taking one of these medicines ask your pharmacist).
Symptoms of an allergic reaction to these medicines may include:
- asthma, wheezing or shortness of breath
- swelling of the face, lips or tongue which may cause difficulty in swallowing or breathing
- hives, itching or skin rash
- fainting.
If you are allergic to sulfonamides or any of the capsule ingredients and take Celebrex, these symptoms may be severe.
Ask your doctor or pharmacist if any of this applies to you. Celebrex may not be suitable for you if any of the conditions below apply to you. You:
- are already taking an NSAID
- have an ulcer or gastric bleeding
- have Irritable Bowel Disease
- have heart failure
- have had a heart attack, a "mini" stroke or stroke or blood vessel disease affecting circulation of blood to your brain or limbs, especially in the last 3 months.
Do not take Celebrex if the expiry date printed on the packaging has passed, even though the capsules may look alright. If you take this medicine after the expiry date has passed, it may not work as well.
Do not take Celebrex if the packaging is torn or shows signs of tampering.
If you are not sure if you should be taking Celebrex, talk to your doctor.
Before you start to take it
You must tell your doctor if you have any of the following health problems:
- you currently have diabetes
- high blood pressure
- high cholesterol levels
- heart failure or have a history of heart problems
- stroke or a history of stroke
- problems with the circulation in your limbs
- you have any allergies to:
- any other medicines
- any other substances such as foods, dyes or preservatives. - you are pregnant or intend to become pregnant.
NSAIDs, which are related medicines, have been associated with reversible infertility in some women.
Use of NSAIDs in early pregnancy can increase the risk of spontaneous abortion.
There is no information on the use of Celebrex during pregnancy.
Celebrex may affect your developing baby if taken during pregnancy. Particular caution should be exerted from the twentieth week of the pregnancy.
Celebrex use is not recommended in pregnancy unless your doctor considers it essential. If you are taking Celebrex while pregnant, you may need to be closely monitored by your doctor. Discuss any questions you may have with your doctor. - you are breast-feeding or intend to breast-feed
Small amounts of celecoxib passes into breast milk, therefore taking Celebrex during breastfeeding should be discussed with your doctor. - you have any other health problems including:
- liver or kidney problems
- asthma, hives, itching, skin rash or a runny nose
- high blood pressure
- fluid retention or other medical conditions that can cause fluid retention
- peptic ulcer (i.e. stomach or duodenal ulcer), a recent history of one, or have had peptic ulcers before
- vomiting blood or material that looks like coffee grounds
- bleeding from the rectum (back passage), have black sticky bowel motions (stools) or bloody diarrhoea
- bowel problems such as ulcerative colitis. - you are taking Celebrex together with any medicines used to treat high blood pressure and some other heart problems such as ACE inhibitors, angiotensin receptor antagonists, beta blockers and diuretics (also called fluid or water tablets).
When taken together these medicines can cause kidney problems. - you drink large amounts of alcohol
- you are a smoker
- you currently have an infection.
If you are given Celebrex while you have an infection, it may hide some of the signs of an infection.
If you have not told your doctor or pharmacist about these things, tell them before you start taking Celebrex.
Taking other medicines
Tell your doctor or your pharmacist if you are taking any other medicines, including medicines you buy without a prescription from a pharmacy, supermarket or health food shop.
Some medicines and Celebrex may interfere with each other. These include:
- any medicines used to treat high blood pressure and some other heart problems such as ACE inhibitors, angiotensin receptor antagonists, beta blockers or diuretics (also called fluid or water tablets)
- digoxin, a medicines used to treat abnormal heart beats and some other heart problems
- fluconazole, an antifungal agent
- lithium, a medicine used to treat some types of depression
- warfarin or similar medicines including Eliquis (apixaban), Xarelto (rivaroxaban) or Pradaxa (dabigatran), medicines used to stop blood clots
- aspirin or salicylates, medicines used to treat pain
- antacids, medicines used to treat indigestion
- dextromethorphan, a medicine used to treat dry coughs
- some medicines used to treat diabetes
- methotrexate, a medicine used to treat arthritis and some cancers
- cyclosporin, a medicine used to suppress the immune system
- corticosteroids such as prednisolone, medicines that are used to reduce inflammation
- certain medicines used to treat pain and inflammation called non-steroidal anti-inflammatory drugs (NSAIDs).
Your doctor may need to adjust the dosage of these medicines, or provide additional advice if you are also taking Celebrex.
How to take Celebrex
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the label, ask your doctor or pharmacist for help.
How much to take
Osteoarthritis
200 mg once daily or 100 mg twice daily.
Rheumatoid arthritis
100 mg twice daily.
Your doctor may increase the dose to 200 mg twice a day for a short period of time if you have a flare up.
Ankylosing spondylitis
100 mg twice daily or 200 mg once daily.
Menstrual cramps or period pain
400 mg as a single dose on the first day and 200 mg twice daily on following days. You may take Celebrex for up to 5 days.
Muscle and joint injuries or after surgery
400 mg as a first dose followed by 200 mg once or twice daily as required. You may take Celebrex for up to 5 days.
How to take it
Swallow the capsules whole with a full glass of water. Celebrex can be taken with or without food.
When to take it
Take your medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
If you need to take an antacid, take it at least 2 hours before or 2 hours after your dose of Celebrex
How long to take it
Depending on your condition, you may need Celebrex for a few days or for longer periods.
Celebrex will not cure your condition but should help control pain, swelling and stiffness.
Keep taking Celebrex for as long as your doctor advises.
Do not exceed the dose recommended by your doctor.
Your risk of developing heart or blood vessel diseases (e.g., heart attack) may increase with dose and duration of use even if you don't have a history of heart or blood vessel disease.
If you need to take Celebrex for a long time see your doctor for regular check-ups so that he/she can monitor your condition and treatment.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to. Otherwise, take it as soon as you remember, then go back to taking your capsules as you would normally.
Do not take a double dose to makeup for the dose you missed.
If you take too much (overdose)
Immediately telephone your doctor or Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at your nearest hospital if you think you or anyone else may have taken too much Celebrex. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
If you take too much Celebrex, you may feel tired, drowsy, sick, vomit, and have stomach pain. You may also have difficulty breathing and feel faint.
While you are using Celebrex
Things you must do
If you become pregnant while taking Celebrex, tell your doctor immediately.
If you are about to start any new medicines, tell your doctor and pharmacist that you are taking Celebrex.
Tell all doctors, dentists and pharmacists who are treating you that you are taking Celebrex.
If you develop any skin rash (e.g. hives, spots) while being treated with Celebrex, contact your doctor immediately. The onset of these events, if they occur, can occur at any time, but most often occur in the first month of treatment.
If you are about to have any blood tests, tell your doctor that you are taking this medicine.
Things you must not do
Do not give Celebrex to anyone else, even if they have the same symptoms or condition as you.
Do not take Celebrex to treat any other complaints unless your doctor tells you to.
Side effects
Check with your doctor as soon as possible if you have any problems while taking Celebrex, even if you do not think the problems are connected with the medicine or are not listed in this leaflet.
Like other medicines, Celebrex can cause some side effects. If they occur, most are likely to be minor and temporary.
Ask your doctor or pharmacist any questions you may have.
Tell your doctor if you notice any of the following:
- stomach pain, diarrhoea, indigestion, wind
- swollen hands, ankles and feet, unexplained weight gain
- dizziness
- sore throat, runny nose, sinusitis, upper respiratory tract infection.
Tell your doctor immediately or go to the Accident and Emergency at your nearest hospital if you notice any of the following:
- skin rash, including hives, raised red, itchy spots
- blistering and bleeding in the lips, eyes, mouth, nose and genitals
- swelling, blistering or peeling of the skin, which may be accompanied by fever, chills, headache, sore throat, diarrhoea, aching joints and muscles
- muscles weakness
- other signs of allergic reaction such as wheezing, swelling of the face, lips, mouth, tongue or throat which may cause difficulty in swallowing or breathing
- collapse or fainting, shortness of breath or tiredness, irregular heartbeat, chest pain, swollen or sore leg veins
- severe stomach or throat pain, vomiting blood or black sticky bowel motions
- bleeding or bruising more than usual, reddish or purple blotches under the skin
- nausea, lethargy, itchiness, flu-like symptoms or yellowing of the skin or eyes (jaundice)
- signs of anaemia such as tiredness, being short of breath and looking pale
- loss or deterioration of hearing
- confusion
- redness, irritation or watering of the eye(s)
- experience sensations with any of the senses (sight, sound, touch, taste or feel) which may not be real
- severe or persistent headache, fever, stiff neck, sensitivity to light and vomiting
- sudden severe headache, loss of consciousness, sudden tingling, numbness or paralysis on one side the face, arm, leg or body, difficulty speaking, understanding, reading or writing, loss of coordination or balance.
These are serious side effects. You may need urgent medical attention.
Not all of these side effects have been reported with Celebrex but have been seen with similar medicines.
Other side effects not listed above may occur in some people.
Do not be alarmed by this list of possible side effects. You may not get any of them.
Tell your doctor if you notice anything else that is making you feel unwell, even if it is not on this list.
After taking Celebrex
Storage
Keep your capsules where young children cannot reach them. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Keep Celebrex in a cool, dry place where the temperature stays at or below 25°C. Do not store it, or any other medicine, in the bathroom or near a sink. Do not leave it in the car or on window sills. Heat and dampness can destroy some medicines.
Keep your capsules in their blister pack until it is time to take them. If you take the capsules out of their container they may not keep well.
Disposal
If your doctor tells you to stop taking Celebrex or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
Celebrex 100 mg - opaque, white capsules with two blue bands marked 7767 and 100.
The 100 mg capsules come in blister packs of 60.
Celebrex 200 mg - opaque, white capsules with two gold bands marked 7767 and 200.
The 200 mg capsules come in blister packs of 10 and 30.
Ingredients
Active ingredient
- Celebrex 100 mg - 100 mg celecoxib/capsule
- Celebrex 200 mg - 200 mg celecoxib/capsule.
Other ingredients
- lactose monohydrate
- sodium lauryl sulfate
- povidone
- croscarmellose sodium
- magnesium stearate
- gelatin
- titanium dioxide
- iron oxide yellow CI 77492 (200 mg capsule)
- indigo carmine CI 73015 (100 mg capsule).
Celebrex does not contain sucrose, gluten, tartrazine or other azo dyes.
Supplier
Celebrex is supplied in Australia by:
Pfizer Australia Pty Ltd
SYDNEY NSW 2000.
Toll Free Number: 1800 675 229.
www.pfizer.com.au.
Australian Registration Number:
Celebrex 100 mg - AUST R 67901.
Celebrex 200 mg - AUST R 67902.
This document was last revised in August 2021.
® Registered Trademark.
© Copyright 2019.
pfccelec10821.
Published by MIMS October 2021




 In placebo- or active-controlled clinical trials, the discontinuation rate due to adverse events was 7.1% for patients receiving Celebrex and 6.1% for patients receiving placebo. Among the most common reasons for discontinuation due to adverse events in the Celebrex treatment groups were dyspepsia and abdominal pain (cited as reasons for discontinuation in 0.8% and 0.7% of Celebrex patients, respectively). Among patients receiving placebo, 0.6% discontinued due to dyspepsia and 0.6% withdrew due to abdominal pain.
In placebo- or active-controlled clinical trials, the discontinuation rate due to adverse events was 7.1% for patients receiving Celebrex and 6.1% for patients receiving placebo. Among the most common reasons for discontinuation due to adverse events in the Celebrex treatment groups were dyspepsia and abdominal pain (cited as reasons for discontinuation in 0.8% and 0.7% of Celebrex patients, respectively). Among patients receiving placebo, 0.6% discontinued due to dyspepsia and 0.6% withdrew due to abdominal pain. The following serious adverse events have occurred rarely in patients taking Celebrex. See Table 4.
The following serious adverse events have occurred rarely in patients taking Celebrex. See Table 4.



 Celebrex (4-fold and 2-fold greater than the recommended OA and RA doses, respectively) was also associated with a significantly lower incidence of clinically relevant decreases in haemoglobin (> 20 g/L) or haematocrit (≥ 10 points) than ibuprofen and diclofenac regardless of aspirin use (Figure 2).
Celebrex (4-fold and 2-fold greater than the recommended OA and RA doses, respectively) was also associated with a significantly lower incidence of clinically relevant decreases in haemoglobin (> 20 g/L) or haematocrit (≥ 10 points) than ibuprofen and diclofenac regardless of aspirin use (Figure 2). In the original registration studies, the incidence of serious upper gastrointestinal complications (bleeding, perforation, gastric outlet obstruction) with Celebrex is not significantly different from placebo and is approximately 8-fold less than with non-specific COX inhibitors.
In the original registration studies, the incidence of serious upper gastrointestinal complications (bleeding, perforation, gastric outlet obstruction) with Celebrex is not significantly different from placebo and is approximately 8-fold less than with non-specific COX inhibitors.
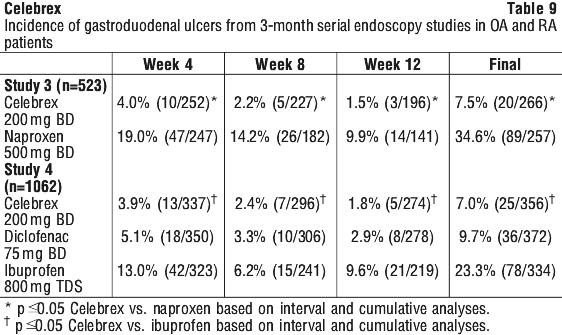
 Figure 4 and Table 9 summarise data from two 12-week studies that enrolled patients in whom baseline endoscopies revealed no ulcers. Patients underwent interval endoscopies every 4 weeks to give information on ulcer risk over time.
Figure 4 and Table 9 summarise data from two 12-week studies that enrolled patients in whom baseline endoscopies revealed no ulcers. Patients underwent interval endoscopies every 4 weeks to give information on ulcer risk over time.
 One randomised and double-blinded 6-month study in 430 RA patients was conducted in which an endoscopic examination was performed at 6 months. The results are shown in Figure 5.
One randomised and double-blinded 6-month study in 430 RA patients was conducted in which an endoscopic examination was performed at 6 months. The results are shown in Figure 5. The correlation between findings of endoscopic studies and the relative incidence of clinically serious upper GI events that may be observed with different products, has not been fully established.
The correlation between findings of endoscopic studies and the relative incidence of clinically serious upper GI events that may be observed with different products, has not been fully established.
 Because of its lack of platelet effects, Celebrex is not a substitute for aspirin for CV prophylaxis.
Because of its lack of platelet effects, Celebrex is not a substitute for aspirin for CV prophylaxis.

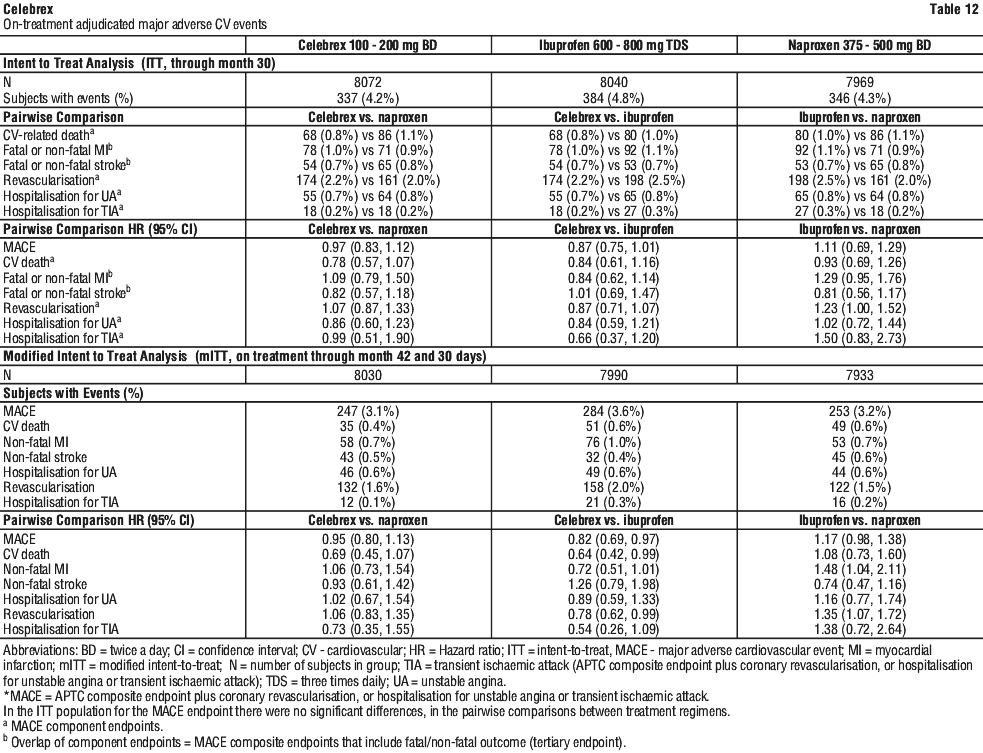 The analysis of gastrointestinal events for ITT and mITT are described in Table 13.
The analysis of gastrointestinal events for ITT and mITT are described in Table 13.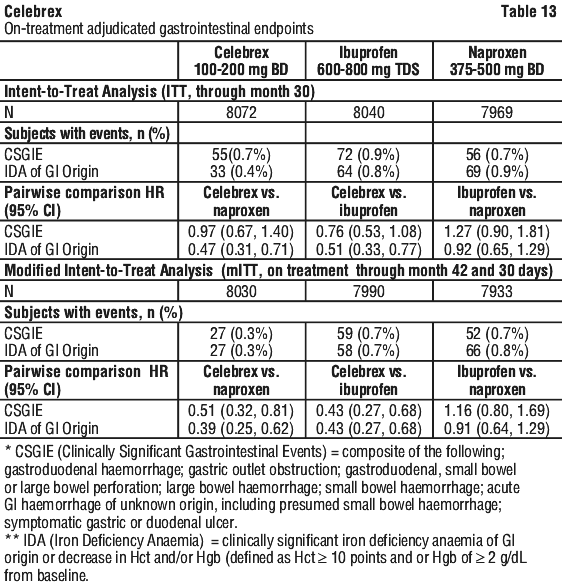 In the ITT population for the CSGIE endpoint there were no significant differences, in the pairwise comparisons between treatment regimens (data not shown). For the endpoint of iron deficiency anaemia of GI origin, significant differences (celecoxib vs naproxen; celecoxib vs ibuprofen) and non-significant differences (ibuprofen vs naproxen) were observed in a manner consistent with the data presented in Table 13.
In the ITT population for the CSGIE endpoint there were no significant differences, in the pairwise comparisons between treatment regimens (data not shown). For the endpoint of iron deficiency anaemia of GI origin, significant differences (celecoxib vs naproxen; celecoxib vs ibuprofen) and non-significant differences (ibuprofen vs naproxen) were observed in a manner consistent with the data presented in Table 13.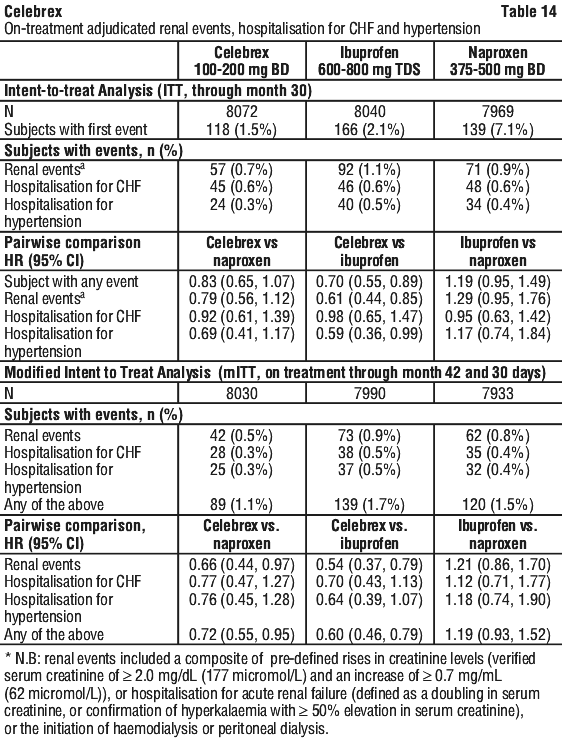 In the ITT population for the endpoint of clinically significant renal events, only the pairwise comparison between celecoxib and ibuprofen was significant, HR 0.61 (0.44, 0.85), no significant differences were observed between treatment regimens in the incidence of hospitalisation for congestive heart failure, and a significantly lower incidence of hospitalisation for hypertension was observed between celecoxib and ibuprofen, HR 0.59 (0.36, 0.99).
In the ITT population for the endpoint of clinically significant renal events, only the pairwise comparison between celecoxib and ibuprofen was significant, HR 0.61 (0.44, 0.85), no significant differences were observed between treatment regimens in the incidence of hospitalisation for congestive heart failure, and a significantly lower incidence of hospitalisation for hypertension was observed between celecoxib and ibuprofen, HR 0.59 (0.36, 0.99).
