What is in this leaflet
This leaflet answers some common questions about CELSENTRI.
It does not contain all the information available. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using CELSENTRI against the benefits they expect it will have for you.
If you have any concerns about using this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What CELSENTRI is used for
What CELSENTRI does
CELSENTRI is used in combination with other anti-HIV medicines to treat HIV. It reduces the amount of HIV in your body and helps your immune system.
CELSENTRI is not a cure for HIV or AIDS. You should take appropriate precautions to prevent transmission.
Ask your doctor if you have any questions about why CELSENTRI has been prescribed for you. Your doctor may have prescribed CELSENTRI for another medical condition.
This medicine is available only with a doctor's prescription.
How CELSENTRI works
CELSENTRI's active ingredient (maraviroc) belongs to a group of medicines called 'CCR5 blockers'.
CELSENTRI stops the HIV-1 virus entering the CD-4 cells in your blood (also called T-cells). These are the cells in your immune system that the HIV virus attacks.
CELSENTRI works by blocking the most common entry point into the CD-4 cells - called the 'CCR5 receptor'. Because the virus cannot enter the cell, it cannot attack it, and this prevents further damage to your immune system.
CELSENTRI only stops the HIV-1 virus entering the cell, not HIV-2 (another rarer kind of the HIV virus). CELSENTRI also only stops types of HIV-1 that enter using the CCR5 receptor. Because of this, your doctor would have done a blood test to check what strain of HIV-1 you have before they prescribed CELSENTRI.
CELSENTRI has not been studied in children less than 18 years of age.
More treatment-naïve patients in clinical studies using CELSENTRI had treatment failures and developed resistance to lamivudine compared to patients using efavirenz.
Before you take CELSENTRI
Use this sheet to check for things that might affect whether you use CELSENTRI, including:
- allergies
- other medical conditions
- pregnancy and breastfeeding
- other medicines you are taking
When you must not take it
Do not take CELSENTRI if you have an allergy to:
- any medicine containing maraviroc (the active ingredient of CELSENTRI)
- any of the other ingredients listed at the end of this leaflet
Symptoms of an allergic reaction may include:
- shortness of breath, wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not take this medicine if the expiry date printed on the pack has passed.
Do not take CELSENTRI if the packaging is torn or shows signs of tampering.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you have allergies to:
- any other medicines,
- any other substances such as foods, preservatives or dyes
Tell your doctor about all of your medical conditions, especially if you have the following:
- liver problems or Hepatitis B or Hepatitis C infection, as your liver function may need to be closely monitored
- a history of low blood pressure, low blood pressure on standing (postural hypotension) or you are taking any medicine to lower blood pressure
- kidney problems
- heart problems
Tell your doctor if you are pregnant or intend to become pregnant. It is not known if CELSENTRI can harm your unborn child. If you become or plan to become pregnant while taking CELSENTRI your doctor will discuss the risks and benefits of taking it.
Tell your doctor if you are breast-feeding or plan to breast-feed. You should not breast-feed if you are infected with HIV because the virus may be transmitted through the breast milk. It is not known whether the active ingredient in CELSENTRI can pass into your breast milk. Therefore, mothers must not breast-feed during treatment with CELSENTRI.
If you have not told your doctor about any of the above, tell them before you start taking CELSENTRI.
Taking other medicines
Some medicines may be affected by CELSENTRI or may affect how well it works. You may need to use different amounts of your medicine, or you may need to take different medicines. Your doctor will advise you.
Tell your doctor if you are taking any other medicines. Tell your doctor about:
- all prescription medicines
- all medicine you buy over-the-counter from a pharmacy or supermarket
- all complementary and alternative therapies
- anything you buy from a health food shop
Tell your doctor if you are taking any of the following:
- other medicines to treat HIV or Hepatitis C infections (e.g. efavirenz, etravirine, raltegravir, lopinavir, darunavir, delavirdine, elvitegravir, atazanavir, nelfinavir, indinavir, saquinavir, boceprevir or telaprevir)
- medicines to treat fungal infections (e.g. ketoconazole and itraconazole)
- medicines to treat bacterial infections (e.g. clarithromycin, telithromycin, rifampicin and rifabutin)
- medicines containing St. John's wort (Hypericum perforatum). St. John's wort can prevent CELSENTRI from working properly. Therefore, you should not take St. John's wort together with CELSENTRI
- medicines to treat seizures (fits) (e.g. carbamazepine, phenobarbital and phenytoin
Talk to your doctor if you are not sure whether you should take this medicine.
Your doctor or pharmacist has more information on medicines to be careful with or to avoid taking together with CELSENTRI.
How to take CELSENTRI
Take the medicine only as prescribed by your doctor. This may differ from the information contained in this leaflet.
How much to take
The usual dose of CELSENTRI is 150 mg, 300 mg or 600 mg twice a day.
This depends on the other medicines that you are taking with CELSENTRI. Always take the dose recommended by your doctor.
Your doctor will tell you the dosage for the other anti-HIV medicines and whether or not you should take them at a different time to CELSENTRI.
How to take it
Swallow your CELSENTRI tablets whole. Do not chew the tablets.
CELSENTRI tablets may be taken with or without food.
If you do not understand the instructions, ask your doctor or pharmacist for help.
When to take it
Take your medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
It is very important to take all your anti-HIV medicines as prescribed and at the right time of day. This can help your medicines work better. It also lowers the chance of your medicines becoming less effective in fighting HIV (also known as drug resistance).
How long to take it
Take the medicine for as long as your doctor tells you to, even if you feel well.
This medicine helps to control your condition, but does not cure it.
If you forget to take it
If it is almost time for your next dose skip the dose you missed and take your next dose when you are meant to. Otherwise, take it as soon as you remember, and then go back to taking your tablet(s) as you would normally.
If you are not sure whether to skip the dose, talk to your doctor or pharmacist.
Do not take a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you find it hard to remember when to take your tablets, ask your doctor or pharmacist for help.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) or go to Accident and Emergency at your nearest hospital, if you think you or anyone else may have taken too much CELSENTRI. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
Symptoms of an overdose may include dizziness or light-headedness when you stand or sit up quickly. If this happens, lie down until you feel better. When you get up, do so as slowly as possible.
While you are taking CELSENTRI
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking CELSENTRI.
Tell any other doctors, dentists, and pharmacists who are treating you that you are taking CELSENTRI.
Tell your doctor immediately if you become pregnant while taking CELSENTRI.
You need to use safe sexual practices while taking CELSENTRI. CELSENTRI has not been shown to decrease the chance of giving HIV to your partner.
Things you must not do
Do not stop taking CELSENTRI or change the dose, even if you feel well, without telling your doctor.
Do not take any other medicine without telling your doctor.
Do not use CELSENTRI to treat any other complaints unless your doctor tells you to.
Do not give CELSENTRI to anyone else, even if they have the same condition as you.
Things to be careful of
Be careful driving or operating machinery until you know how CELSENTRI affects you. CELSENTRI may cause dizziness or light-headedness in some people. Make sure you know how you react to CELSENTRI before you drive a car, cycle, operate machinery or do anything else that could be dangerous.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using CELSENTRI, even if you do not think the problem is connected with the medicine or is not listed in this leaflet.
Other side effects not listed here may affect some people.
This medicine helps most people with the HIV infection but it may have unwanted side effects in some people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
The side effects in this sheet are divided into:
- common side effects
- serious side effects
- very serious side effects
Tell your doctor if you notice anything that is making you feel unwell or if your condition changes.
It is often difficult to tell whether side effects are the result of taking CELSENTRI, effects of the HIV disease or side effects of other medicines you may be taking. For this reason it is very important to report any change in your condition. Your doctor may want to change your dose or advise you to stop taking CELSENTRI.
Tell your doctor if
The following list shows the more common side effects of CELSENTRI.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- diarrhoea
- nausea or vomiting
- stomach pain or discomfort
- indigestion
- constipation
- dizziness
- abnormal sense of taste
- problems sleeping or abnormal sleep
- rash
- loss of appetite
- muscle spasms or pain
- cough
- joint pain
- fever
- colds, upper respiratory tract infections or flu-like symptoms
Tell your doctor as soon as possible if
The following list includes serious side effects which may require medical attention.
Tell your doctor as soon as possible if you notice any of the following:
- weakness, tiredness, headaches, being short of breath, dizziness and looking pale, fainting
- signs of frequent infections such as fever, severe chills, sore throat or mouth ulcers
- loss of appetite, vomiting and/or upper right stomach pain, feeling generally unwell, fever, itching, yellowing of the skin and eyes, passing less or more urine than normal, or a change of the colour of your urine, dark coloured urine
- unexpected muscle aches or pain, paralysis or weakness
- signs of infections such as:
- meningitis - fever, nausea, vomiting, headache, stiff neck and sensitivity to bright light
- pneumonia - fever, chills, shortness of breath, cough, phlegm and occasionally blood
- bleeding problems such as:
- bleeding or bruising more easily than normal, nosebleeds
- bleeding from the back passage (rectum)
- convulsions, fits or seizures, shaking or tremors
- swollen glands in the neck, armpit or groin
- any mental problems, such as
- epilepsy or fits
- loss of reflexes
- hallucinations
- inability to move the muscles in the face
- tingling or burning sensation in the hands or feet
- lower back pain
- feeling dizzy, faint or light headed when standing up
Go to hospital if
The following list contains very serious side effects. You may need urgent medical attention or hospitalisation.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- chest pain or angina
- severe upper stomach pain, often with nausea or vomiting
- shortness of breath, wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- blisters and peeling skin around the mouth, nose, eyes and genitals
- widespread skin rash, itching or hives on the skin
- signs of stroke - collapse, numbness or weakness of the arms or legs, headache, dizziness and confusion, visual disturbance, difficulty swallowing, slurred speech and loss of speech
- meningitis - fever, nausea, vomiting, headache, stiff neck and sensitivity to bright light
Changes in your immune system.
A condition called Immune Reconstitution Syndrome can happen when you start taking HIV medicines. Your immune system may get stronger and could begin to fight infections that have been hidden in your body such as pneumonia, herpes virus or tuberculosis. Tell your doctor if you develop new symptoms after starting your HIV medicines.
Possible chance of infection or cancer.
Although there is no evidence from the clinical trials of an increase in serious infections or cancer, CELSENTRI affects other immune system cells and therefore may potentially increase your chance of getting other infections or cancer.
After using CELSENTRI
Storage
Keep the CELSENTRI tablets in their pack until it is time to take them. If you take the tablets out of the pack they may not keep well.
Keep CELSENTRI in a cool dry place where the temperature stays below 30 degrees C.
Do not store CELSENTRI or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop using CELSENTRI or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product Description
What it looks like
CELSENTRI film-coated tablets are blue in colour and oval-shaped. The tablets are marked with "MVC" followed by the tablet strength on one tablet side.
CELSENTRI 150 mg and 300 mg film-coated tablets are supplied in a polyvinyl chloride/foil blister pack or a polyvinyl chloride/child resistant foil blister pack in a carton of 60 tablets.
Ingredients
The active ingredient in CELSENTRI is maraviroc. Each tablet contains either 150 mg or 300 mg of maraviroc.
The inactive ingredients are:
- microcrystalline cellulose
- calcium hydrogen phosphate
- sodium starch glycollate
- magnesium stearate
The film-coat [Opadry II Blue (85G20583)] contains:
- indigo carmine CI73015
- soya lecithin
- macrogol 3350
- polyvinyl alcohol
- talc
- titanium dioxide
Supplier
CELSENTRI is supplied in Australia by:
ViiV Healthcare Pty Ltd
Level 4, 436 Johnston Street
Abbotsford Victoria 3067
Australia
Australian Registration Numbers
CELSENTRI 150 mg film-coated tablets in blister packs AUST R 137329
CELSENTRI 300 mg film-coated tablets in blister packs AUST R 137331
Trademarks are owned by or licensed to the ViiV Healthcare group of companies.
© 2019 ViiV Healthcare group of companies or its licensor.
This leaflet was prepared on 28 March 2019
Version 8.0
Published by MIMS October 2019







 MOTIVATE 1 and MOTIVATE 2 were unblinded after the Week 48 visit of the last enrolled patient, and eligible patients could switch to an open-label phase of Celsentri twice daily extending to week 96. A subsequent observational phase extending to 5 years on treatment was conducted to assess the incidence of the following long-term safety selected endpoints; death, AIDS defining events, hepatic failure, MI/cardiac ischemia, malignancies, rhabdomyolysis, and other serious infectious events. The incidence of these selected endpoints was consistent with those seen at earlier time points in the studies and listed in Table 6.
MOTIVATE 1 and MOTIVATE 2 were unblinded after the Week 48 visit of the last enrolled patient, and eligible patients could switch to an open-label phase of Celsentri twice daily extending to week 96. A subsequent observational phase extending to 5 years on treatment was conducted to assess the incidence of the following long-term safety selected endpoints; death, AIDS defining events, hepatic failure, MI/cardiac ischemia, malignancies, rhabdomyolysis, and other serious infectious events. The incidence of these selected endpoints was consistent with those seen at earlier time points in the studies and listed in Table 6.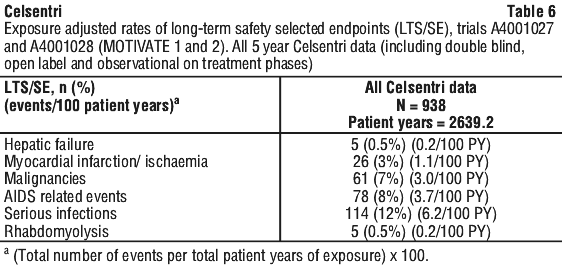

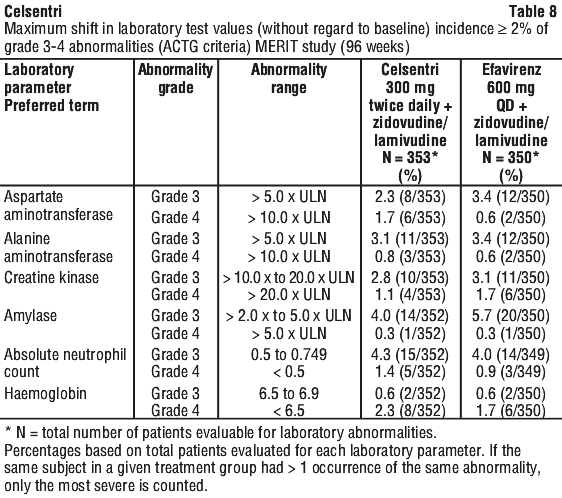
 Limited numbers of patients from races other than Caucasian were included in the pivotal clinical studies, therefore very limited data are available in these patient populations.
Limited numbers of patients from races other than Caucasian were included in the pivotal clinical studies, therefore very limited data are available in these patient populations.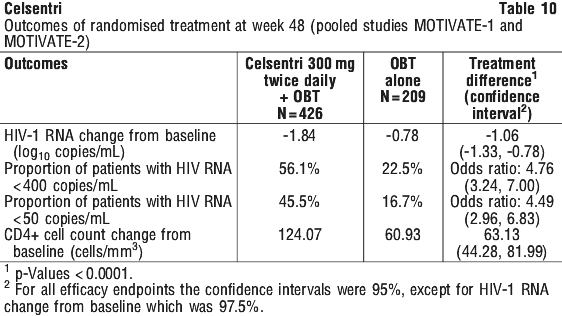

 The treatment outcomes through week 48 for the treatment-naïve study (MERIT) are shown in Table 13. Treatment outcomes (responders) include reanalysis of the screening samples using a more sensitive tropism assay, enhanced sensitivity Trofile HIV tropism assay, which became available after the Week 48 analysis.
The treatment outcomes through week 48 for the treatment-naïve study (MERIT) are shown in Table 13. Treatment outcomes (responders) include reanalysis of the screening samples using a more sensitive tropism assay, enhanced sensitivity Trofile HIV tropism assay, which became available after the Week 48 analysis. The primary efficacy endpoints were defined as the percentage of patients with HIV-1 RNA undetectable by the standard method (< 400 copies/mL and < 50 copies/mL). After 48 weeks of combination therapy with zidovudine/lamivudine, maraviroc 300 mg BID demonstrated non-inferiority to efavirenz 600 mg QD in the proportion of patients with undetectable viral load measured at < 400 copies/mL but not at < 50 copies/mL (lower bound of 97.5% CI > -10% for non-inferiority). However, reanalysis of the data following rescreening of the samples using the enhanced sensitivity tropism assay demonstrated non-inferiority for maraviroc 300 mg BID compared to efavirenz 600 mg QD in the proportion of patients with viral loads of < 400 copies/mL and < 50 copies/mL.
The primary efficacy endpoints were defined as the percentage of patients with HIV-1 RNA undetectable by the standard method (< 400 copies/mL and < 50 copies/mL). After 48 weeks of combination therapy with zidovudine/lamivudine, maraviroc 300 mg BID demonstrated non-inferiority to efavirenz 600 mg QD in the proportion of patients with undetectable viral load measured at < 400 copies/mL but not at < 50 copies/mL (lower bound of 97.5% CI > -10% for non-inferiority). However, reanalysis of the data following rescreening of the samples using the enhanced sensitivity tropism assay demonstrated non-inferiority for maraviroc 300 mg BID compared to efavirenz 600 mg QD in the proportion of patients with viral loads of < 400 copies/mL and < 50 copies/mL. The median increase from baseline in CD4+ cell counts at week 96 was 184 cells/mm3 for the Celsentri arm compared to 155 cells/mm3 for the efavirenz arm.
The median increase from baseline in CD4+ cell counts at week 96 was 184 cells/mm3 for the Celsentri arm compared to 155 cells/mm3 for the efavirenz arm.
