What is in this leaflet
This leaflet answers some common questions about CERVIDIL. It does not contain all the available information. It does not take the place of talking to your doctor or midwife.
All medicines have risks and benefits. Your doctor has weighed the risks of you being given CERVIDIL against the benefits he/she expects it will have for you.
If you have any concerns about being given this medicine, ask your doctor or midwife.
Keep this leaflet in a safe place. You may need to read it again.
What CERVIDIL is used for
CERVIDIL is for women who have a normal pregnancy and are near their due date for delivery. It is used to prepare for induction of labour.
CERVIDIL is a pessary (vaginal insert) containing dinoprostone, also known as Prostaglandin E2 or PGE2. Prostaglandin E2 also occurs naturally in the body. Prostaglandin E2 is important for the changes that take place before labour begins.
CERVIDIL is used to prepare the cervix (the neck of the womb, at the top of the birth canal) to allow the baby to pass through. This process is called "cervical ripening".
This medicine is available only with a doctor's prescription.
Your doctor may have prescribed CERVIDIL for another purpose.
Ask your doctor or midwife if you have any questions about why CERVIDIL has been prescribed for you.
How it works
CERVIDIL works by:
- softening and opening the cervix (neck of the womb)
- setting off contractions (in the body of the womb)
- releasing dinoprostone continuously to the cervix at the appropriate rate.
This allows softening and opening of the cervix to progress.
When the doctor decides no further dinoprostone is required, the pessary (vaginal insert) is removed by pulling on the withdrawal tape.
Before you are given CERVIDIL
Your doctor will decide if CERVIDIL is suitable for you.
CERVIDIL should be administered only by trained personnel, in hospital, with appropriate obstetrical care and facilities for the required monitoring.
When you must not be given it
You must not be given CERVIDIL if you have an allergy to dinoprostone or any of the ingredients (e.g. urethane) listed at the end of this leaflet.
Symptoms of an allergic reaction may include:
- shortness of breath, wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
You must not be given CERVIDIL if:
- you are carrying more than one baby
- your labour has already started
- your waters have broken
- there is any reason why you should not have a vaginal delivery, for example, genital herpes
- the baby's head is not well down in the pelvis
- the baby is not in the normal position for birth, or
- if it is suspected, or tests show, your baby is unwell or not growing, or
- the head of the baby is too big or the size of your pelvis is too small for normal delivery, or
- you have contractions that are unusually strong and/or long (known as "hypertonic contractions" or "hyperstimulation of the uterus").
You must not be given CERVIDIL if you have had any of the following:
- previous surgical operation on the womb, for example, a caesarean section, or
- surgery to the neck of the womb (cervix), or
- previous rupture of the cervix
- any vaginal discharge or unexplained vaginal bleeding during the current pregnancy.
You must not be given CERVIDIL if you have untreated pelvic inflammatory disease
(also known as PID); usually caused by an infection of the internal female sex organs; it may result in, for example, pain and tenderness of the stomach and fever.
You should not be given CERVIDIL:
- if the packaging is torn or shows signs of tampering
- after the expiry date (EXP) printed on the pack.
If you are not sure whether you should be given CERVIDIL, talk to your doctor.
Taking other medicines
Tell your doctor or midwife if you are taking/using any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop. Some medicines and CERVIDIL may interfere with each other.
Your doctor or midwife may have more information on medicines to be careful with or avoid while using CERVIDIL.
You must not be given CERVIDIL if you are being given, or are to be given intravenously within the next thirty minutes, medicines to make the muscles of your womb contract or bring labour, e.g. oxytocin.
Medication with aspirin and other non-steroidal anti-inflammatory drugs (known as NSAIDs) should be stopped before administration of CERVIDIL. Some examples of NSAIDs are Naprosyn and Voltaren.
Tell your doctor or midwife if you have allergies to:
- any other medicines
- any other substances, such as foods, preservatives or dyes.
Before you are given CERVIDIL tell your doctor or midwife if you are over 35 years of age or if you have had any medical conditions, especially the following:
- lung, liver or kidney problems
- asthma
- epilepsy (convulsions or fits)
- unexplained genital bleeding during current pregnancy
- glaucoma (raised pressure in the eye)
- abnormally strong contractions of your womb during previous labour, or
- previous excessively short labour and delivery time
- heart or blood pressure problems
- previous complications during pregnancy
- you have had more than three full term deliveries.
- gestational diabetes
- your pregnancy is past 40 weeks gestation.
When CERVIDIL must be removed
The pessary (vaginal insert) should be removed immediately:
- if contractions are considered too sustained or excessive, or
- if labour commences.
It should also be removed
- prior to amniotomy
- after the waters break (spontaneous rupture of the membranes)
- if there is any suggestion of maternal or fetal complications, or
- if unwanted side-effects occur
- if, after 24 hours the cervix has not changed adequately for delivery.
How CERVIDIL is given
How much is given
CERVIDIL is given as one pessary (vaginal insert) inserted once only. Each CERVIDIL insert contains 10 mg dinoprostone.
Over the maximum recommended usage period of 24 hours, the insert gradually releases dinoprostone.
Over the maximum recommended usage period of 24 hours, the insert gradually releases about 0.3 mg of dinoprostone per hour.
How it is given
CERVIDIL must only be given under the supervision of a doctor.
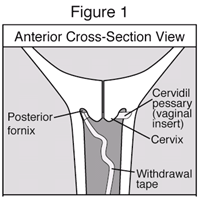
CERVIDIL is inserted into the vagina. The pouch containing the active ingredient is positioned up at the top of the vagina behind the cervix. This is called the "posterior fornix" (See Figure 1).
The tape for withdrawal hangs from the entrance to the vagina.
Before CERVIDIL is used, careful assessment of the cervix is necessary.
After insertion of CERVIDIL the following must be monitored regularly:
- changes in the cervix
- presence or absence of contractions
- frequency, duration and strength of contractions
- baby's health.
If there is any suggestion of maternal or fetal complications, or if adverse effects occur, the CERVIDIL pessary (vaginal insert) should be removed. This is done by gently pulling on the withdrawal tape, until the whole device is removed from the vagina.
Administration
CERVIDIL looks like a small slim tampon, with a very long attached tape. CERVIDIL must not be used without this tape, which is used to withdraw the pessary (vaginal insert).
Your doctor or midwife will coat the CERVIDIL with a little lubricating jelly before putting it in your vagina. The tampon-like end, which holds the medicine, is placed behind the neck of the womb (cervix), in the area known as the "posterior fornix" of the vagina. The medicine gradually passes from the device into the upper vagina. The continuing concentration of medicine in the fluids around the cervix causes the cervix to become softer and gradually open.
The attached withdrawal tape is left hanging out of the entrance to your vagina. Your doctor or midwife can therefore easily pull out the CERVIDIL pessary (vaginal insert) when it is time to do so, or if it needs to be removed for any reason.
While you are being given CERVIDIL
Things you must do
You will be lying down while CERVIDIL is put in. You should remain lying down for at least 30 minutes afterwards. Your doctor or midwife will advise you when you can get up again. The pessary (vaginal insert) should be left in place for no longer than 24 hours.
While the pessary (vaginal insert) is in place, you will be checked frequently. Examples of what is being checked include but are not limited to:
- the neck of the womb (cervix)
- the strength and frequency of any contractions
- the health of your unborn baby.
Removal
The pessary (vaginal insert) is removed by gently pulling the withdrawal tape.
How long it is used for
Your doctor or midwife will remove the pessary (vaginal insert) when you no longer need it or after 24 hours.
For example, they may remove it because:
- your labour has started
- your doctor wants to use a different medicine to help your womb (uterus) contract e.g. oxytocin
- your waters have broken
- your uterus is contracting too strongly
- your baby is starting to get distressed.
OVERDOSAGE
Your medical attendants will be alert for any signs of overdose. Your doctor, midwife or pharmacist have information on how to recognise and treat an overdose. Initial treatment of overdose is removal of the pessary (vaginal insert). Other treatment is also available.
Contact the Poisons Information Centre on 131 126 for further advice on overdose management.
Side effects
Tell your doctor or midwife as soon as possible if you do not feel well while you are being given CERVIDIL.
All medicines can have side effects. Sometimes they are serious, most of the time they are not.
In most people, CERVIDIL helps prepare the cervix (neck of womb) for birth. It may have unwanted side effects in a few people.
Tell your doctor or midwife immediately if you notice any of the following:
- headache
- dizziness
- itching
- bleeding, possibly from multiple sites of your body
- fever
- nausea or vomiting
- very strong or very frequent contractions of the womb
- bluish coloration of the fingers
- sudden bruising.
The above list includes serious side effects. Your doctor may decide to remove the CERVIDIL pessary (vaginal insert) if these side effects occur.
Tell your doctor or midwife if you notice any other side effects.
Rarely, rupture of the womb has been reported in association with the use of CERVIDIL. However, most of these patients should not have been given CERVIDIL. See the section beginning "When you must not be given it" above.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Ask your doctor or midwife to answer any questions you may have.
Product description
What it looks like
The CERVIDIL pessary (vaginal insert) is:
- a thin, flat rectangle, with rounded corners
- slightly thicker than a large postage stamp
- beige in colour
- contained within a pouch
- made so that, when the pouch becomes moist, the active ingredient (dinoprostone) comes out very slowly. The pouch forms one end of a long tape
- pouch and tape are made of knitted polyester (off-white in colour)
- tape allows withdrawal of the insert at the end of dosing.
NOTE: After recommended use, when CERVIDIL is removed from the vagina, the tampon-like end will have become larger. It absorbs fluid and becomes 2-3 times its original size.
Ingredients
The active ingredient in CERVIDIL is dinoprostone.
The active ingredient is within a plastic (polyurethane) sustained release insert which contains:
- hexanetriol/macrogol 8000/ isocyanate cross-linked hydrogel copolymer.
The insert is held within a pouch in continuity with a withdrawal tape. Pouch and tape are made of knitted polyester yarn.
Presentation
CERVIDIL is in an aluminium/polyethylene foil sachet, each containing 1 pessary (vaginal insert).
Storage
CERVIDIL is kept in a freezer and removed immediately before use. It is stored in the hospital.
Disposal
The used pessary (vaginal insert) should be disposed of as clinical waste.
Sponsor
Ferring Pharmaceuticals Pty Ltd
Suite 2, Level 1, Building 1
20 Bridge Street
Pymble, NSW 2073, Australia.
AUST R 81391 - CERVIDIL dinoprostone 10 mg pessary sachet
This leaflet was prepared in January 2018.
#37784-v2B.
CERVIDIL® is a registered trademark of Ferring B.V.
® = Registered trademark
Published by MIMS February 2018


 Cervidil should be used with caution in patients with compromised cardiovascular function.
Cervidil should be used with caution in patients with compromised cardiovascular function. (See Table 2.)
(See Table 2.) In study 101-801, five minute Apgar scores were below 7 in 1.8% (646/658) of studied neonates whose mothers received Cervidil for up to 12 hours. In Miso-Obs-303, where Cervidil was inserted up to 24 hours, five minute Apgar scores of below 7 were reported in neonates of 1% of participants.
In study 101-801, five minute Apgar scores were below 7 in 1.8% (646/658) of studied neonates whose mothers received Cervidil for up to 12 hours. In Miso-Obs-303, where Cervidil was inserted up to 24 hours, five minute Apgar scores of below 7 were reported in neonates of 1% of participants. An increased risk of post-partum DIC has been reported in patients whose labour was induced by pharmacological means, either with dinoprostone or oxytocin.
An increased risk of post-partum DIC has been reported in patients whose labour was induced by pharmacological means, either with dinoprostone or oxytocin.
 Approximately 60% of the women in these two Phase 3 trials required cervical ripening to continue beyond 12 hours and a total of 302 women had the insert in situ for 24 hours during the two trials (83 in Miso-Obs-004 and 219 in Miso-Obs-303). Table 6 shows the efficacy results in both trials, where results for the DVI group are consistent in these two Phase 3 trials.
Approximately 60% of the women in these two Phase 3 trials required cervical ripening to continue beyond 12 hours and a total of 302 women had the insert in situ for 24 hours during the two trials (83 in Miso-Obs-004 and 219 in Miso-Obs-303). Table 6 shows the efficacy results in both trials, where results for the DVI group are consistent in these two Phase 3 trials. A total of 8.38% and 11.7% of subjects in the DVI groups delivered vaginally within 12 hours which increased to 33.97% and 42.7% by 24 hours in the Miso-Obs-303 and Miso-Obs-004 trials, respectively. There were 66.0% and 59.6% of subjects in the DVI groups who achieved the composite endpoint for cervical ripening at 12 hours in Miso-Obs-303 and Miso-Obs-004 trials, respectively. In Miso-Obs-303, the percentage of subjects in the DVI group with cervical ripening success increased to 86.8% at 24 hours.
A total of 8.38% and 11.7% of subjects in the DVI groups delivered vaginally within 12 hours which increased to 33.97% and 42.7% by 24 hours in the Miso-Obs-303 and Miso-Obs-004 trials, respectively. There were 66.0% and 59.6% of subjects in the DVI groups who achieved the composite endpoint for cervical ripening at 12 hours in Miso-Obs-303 and Miso-Obs-004 trials, respectively. In Miso-Obs-303, the percentage of subjects in the DVI group with cervical ripening success increased to 86.8% at 24 hours. Molecular formula: C20H32O5. Molecular weight: 352.5.
Molecular formula: C20H32O5. Molecular weight: 352.5.