What is in this leaflet
This leaflet answers some common questions about Chemists’ Own Sinus and Nasal Decongestant. It does not contain all the available information and it does not take the place of talking to your doctor or pharmacist.
Keep this leaflet with the medicine. You may want to read it again later.
What Chemists’ Own Sinus and Nasal Decongestant is used for
Chemists’ Own Sinus and Nasal Decongestant provides effective relief of nasal and sinus congestion due to:
- allergic (seasonal) rhinitis
- vasomotor (perennial) rhinitis
- sinusitis
- the common cold and flu
Pseudoephedrine belongs to a group of medicines called sympathomimetic decongestants.
It works by reducing congestion in the upper respiratory tract, including the nose, nasal passages and sinuses, and making it easier to breathe.
Ask your pharmacist or doctor if you have any questions about this medicine. Your pharmacist or doctor may have given it for another reason.
This medicine is only available from your pharmacist.
Before you use Chemists’ Own Sinus and Nasal Decongestant
When you must not take it
Do not take Chemists’ Own Sinus and Nasal Decongestant if you have an allergy to:
- any medicine containing pseudoephedrine
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not take this medicine if you have:
- very high blood pressure
- severe coronary artery disease (heart disease caused by poor blood flow or narrowing of the blood vessels of the heart)
- taken monoamine oxidase inhibitors, medicines used to treat depression, in the last 14 days
Do not take this medicine if you are pregnant or plan to become pregnant. It may affect your developing baby if you take it during pregnancy. Your pharmacist or doctor will discuss the benefits and possible risks of taking the medicine during pregnancy.
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your pharmacist or doctor.
Before your start to take it
Tell your pharmacist or doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor or pharmacist if you have or have had any of the following medical conditions:
- high blood pressure
- overactive thyroid gland
- diabetes
- heart disease and poor blood flow in the blood vessels of the heart
- glaucoma (high pressure in the eyes)
- prostate problems
- liver or kidney disease
Ask your pharmacist or doctor about taking pseudoephedrine if you are breastfeeding. Small amounts of pseudoephedrine pass into the breast milk. Your pharmacist or doctor will discuss the potential benefits and risks of taking the medicine if you are breastfeeding.
If you have not told your doctor or pharmacist about any of the above, tell them before you use Chemists’ Own Sinus and Nasal Decongestant.
Taking other medicines
Tell your pharmacist or doctor if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Chemists’ Own Sinus and Nasal Decongestant may interfere with each other. These include:
- medicines used to treat depression
- medicines used to treat heart conditions
- medicines used to treat high blood pressure
- medicines used to treat urinary tract infections and bladder problems
- medicines used to treat behavioural disorders
- phenylephrine, a medicine used to treat congestion
- appetite suppressants
These medicines may be affected by Chemists’ Own Sinus and Nasal Decongestant or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist will have more information on medicines to be careful with or avoid while taking this medicine.
How to take Chemists’ Own Sinus and Nasal Decongestant
Follow all directions given to you by your pharmacist or doctor carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask your pharmacist or doctor for help.
Adults & Children over 12 years: Take 1 tablet 3 to 4 times a day when necessary (maximum 4 tablets in 24 hours).
Not recommended for children under 12 years.
How to take it
Swallow tablets with a glass of water.
When to take it
Take the recommended dose 3 to 4 times a day, as required.
How long to take it
Do not use for more than 7 days except on medical advice.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, you may take a dose as soon as you remember if you think you need it.
Do not take a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your pharmacist or doctor.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have taken too much Chemists’ Own Sinus and Nasal Decongestant. Do this even if there are no signs of discomfort or poisoning.
While you are using Chemists’ Own Sinus and Nasal Decongestant
Things you must do
Talk to your pharmacist or doctor if your symptoms persist, worsen, or if new symptoms appear. Your pharmacist or doctor will assess your condition and decide if you should continue to take the medicine.
Things you must not do
Do not take Chemists’ Own Sinus and Nasal Decongestant to treat any other complaints unless your pharmacist or doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not take more than the recommended dose unless your pharmacist or doctor tells you to.
Things to be careful of
Be careful driving or operating machinery until you know how Chemists’ Own Sinus and Nasal Decongestant affects you. This medicine may cause dizziness in some people. If this happens, do not drive or operate machinery.
Side effects
Tell your pharmacist or doctor as soon as possible if you do not feel well while you are taking Chemists’ Own Sinus and Nasal Decongestant.
This medicine helps most people with symptoms of sinus and nasal congestion, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your pharmacist or doctor to answer any questions you may have.
Tell your pharmacist or doctor if you notice any of the following and they worry you:
- headache
- drowsiness
- dizziness
- difficulty sleeping
- nervousness
- excitability
- feeling of extreme happiness
- restlessness
- fear or anxiety
- change in heart rate
- rapid or irregular heart beat
- raised blood pressure
- tremor
- hallucinations
- dry mouth
- nausea
- vomiting
The above list includes the more common side effects of your medicine. They are usually mild and short-lived.
Children and people over 65 years of age may have an increased chance of getting side effects.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- urine retention
- pain when passing urine
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- skin reddening, blisters, rash, itching or hives on the skin
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare.
Tell your pharmacist or doctor if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
Storage
Keep your tablets in the blister pack until it is time to take them. If you take the tablets out of the blister pack they may not keep well.
Keep it in a cool dry place where the temperature stays below 30°C.
Do not store any medicines in the bathroom or in moist areas, or do not leave them in the car on hot days. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard or at least one-and-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking Chemists’ Own Sinus and Nasal Decongestant, or the tablets have passed their expiry date, ask you pharmacist what to do with any that are left over.
Product description
What it looks like
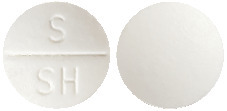
Chemists’ Own Sinus and Nasal Decongestant is a small white tablet with break bar & S/SH on one side.
Active ingredients
Each tablet contains Pseudoephedrine Hydrochloride 60mg
Inactive ingredients
- Carnauba wax
- Microcrystalline cellulose
- Crospovidone
- Dextrates
- Hypromellose
- Macrogol
- Magnesium stearate
- Colloidal anhydrous silica
Contains Sugars
Supplier
Chemists’ Own Sinus and Nasal Decongestant is supplied by:
Arrotex Pharmaceuticals Pty Ltd
15 – 17 Chapel Street
Cremorne, Victoria, 3121.
www.arrotex.com.au
This leaflet was updated in August 2023.
The Australian Registration Number for Chemists’ Own Sinus and Nasal Decongestant is AUST R 93484
Published by MIMS October 2023


 Chemical name: [S-(R*,R*)]-a-[1-(methylamino)ethyl] benzenemethanol hydrochloride.
Chemical name: [S-(R*,R*)]-a-[1-(methylamino)ethyl] benzenemethanol hydrochloride.