What is in this leaflet
This leaflet answers some common questions about CLOFEN.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking CLOFEN against the benefits expected for you.
If you have any concerns about taking this medicine, talk to your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What CLOFEN is used for
CLOFEN belongs to a group of medicines called muscle relaxants.
This medicine is used to reduce stiffness and/or spasms in your muscles.
CLOFEN is used to control muscle spasms in conditions such as:
- multiple sclerosis
- spinal cord damage resulting from disease or physical injury
Because this medicine reduces spasms and the pain that goes with them, it helps to make you more mobile and better able to manage your daily activities.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
CLOFEN is only available with a doctor's prescription.
Before you take CLOFEN
When you must not take it
Do not take CLOFEN if you have an allergy to:
- any medicine containing baclofen
- any of the ingredients listed at the end of this leaflet
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you are pregnant or plan to become pregnant or are breastfeeding or wish to breastfeed. There is very little information on the use of this medicine in pregnancy or breastfeeding. Your doctor can discuss with you the risks and benefits involved.
If you have to take CLOFEN when you are pregnant, your baby may have convulsions and other symptoms related to sudden discontinuation of the medicine just after delivery.
Tell your doctor if you have or have had any of the following medical conditions:
- a mental illness
- Parkinson's disease
- seizures (fits) from any cause
- stiffness and restriction of movement in a group of muscles
- stomach ulcers
- stroke or other blood circulation disease
- heart disease
- kidney disease
- liver disease
- lung problems which make breathing difficult
- diabetes
- porphyria, a rare disturbance in the production of porphyrin, a pigment important for liver function and blood formation
- history of alcoholism, drink alcohol to excess or you have a history of drug abuse or dependence. Some people treated with baclofen have had thoughts of harming or killing themselves or have tried to kill themselves. Most of these people also had depression, had been using alcohol excessively or were prone to having thoughts of killing themselves. If you have thoughts of harming or killing yourself at any time, speak to your doctor straightaway or go to the hospital. Also, ask a relative or close friend to tell you if they are worried about any changes in your behaviour and ask them to read this leaflet.
- high blood pressure
Your doctor may want to take special precautions if you have any of the above conditions. These precautions may include additional tests during or prior to taking CLOFEN.
If you have not told your doctor about any of the above, tell them before you start taking CLOFEN.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from a pharmacy, supermarket or health food shop.
Some medicines and CLOFEN may interfere with each other. These include:
- medicines that can make you sleepy, such as medicines used to help you sleep or calm you down (sleeping tablets or sedatives), pain relievers and medicines for hay fever or allergy, cough and cold, blocked nose
- some medicines used to treat mood disorders, including tricyclic antidepressants, lithium and monoamine oxidase inhibitors (MAOIs)
- medicines used to treat diabetes
- medicines for high blood pressure
- medicines used to treat Parkinson's disease, including selegiline, levodopa and carbidopa
You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How to take CLOFEN
Follow all directions given to you by your doctor and pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the bottle, ask your doctor or pharmacist for help.
How much to take
Treatment is usually started in hospital with small doses of CLOFEN. The dose is then gradually increased to an amount that works best for you. For example, CLOFEN may be started at a dose of 15 mg per day, then increased slowly to anywhere from 30 to 75 mg a day. Sometimes, doses up to 100 mg a day may be needed.
If you are under the age of 16 or over 65 or you have kidney disease, your doctor may start you on a lower dose and increase it more gradually to prevent unwanted side effects.
How to take it
Swallow the tablets during meals with a little liquid.
CLOFEN is usually taken in at least 3 divided doses throughout the day. But your doctor may tell you to take it more or less often, depending on your situation.
How long to take it
Continue taking your medicine for as long as your doctor tells you.
Your doctor will check your progress to make sure the medicine is working for you and will discuss with you how long your treatment should continue.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital, if you think you or anyone else may have taken too much CLOFEN.
Do this even if there are no signs of discomfort or poisoning.
The main symptoms of overdose are: drowsiness, breathing difficulties, consciousness disorders and being unconscious (coma).
Other symptoms may include: feeling confused, hallucinations, agitation, convulsions, blurred vision, unusual muscle slackness, sudden contraction of the muscles, poor or absent reflexes, high or low blood pressure, slow, fast or irregular heartbeat, low body temperature, nausea, vomiting, diarrhoea, or excessive salivation, trouble breathing during sleep (sleep apnoea), pain in your muscles, fever and dark urine (rhabdomyolysis).
If you have kidney disease and have accidently taken more tablets than your doctor has prescribed, you may experience some neurological symptoms of overdose such as drowsiness, feeling confused or hallucinations.
While you are taking CLOFEN
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking CLOFEN.
Tell any other doctors, dentists and pharmacists who treat you that you are taking CLOFEN.
If you become pregnant while taking CLOFEN, tell your doctor immediately. Your doctor can discuss with you the risks of taking it while you are pregnant.
Be sure to keep all of your doctor's appointments so that your progress can be checked. To help prevent unwanted side effects from happening, your doctor may want to do some tests from time to time during the course of your treatment.
If your muscle spasms come back, tell your doctor. Your doctor may be able to change the dose of CLOFEN to make it work better for you.
Things you must not do
Do not stop taking CLOFEN suddenly. This medicine is not habit-forming but stopping it suddenly may bring on severe spasms and other unwanted symptoms, such as nervousness, feeling confused, hallucinations, abnormal thinking or behaviour, convulsions, uncontrollable twitching, jerking or writhing movements, fast heartbeat, high body temperature, pain in muscles, fever and dark urine. The excessive stiffness (spasms) in your muscles may also get worse.
If CLOFEN must be stopped, your doctor will reduce the dose gradually over a period of 1 to 2 weeks so that these unwanted effects are avoided.
Do not take CLOFEN to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if their symptoms seem to be similar to yours.
Things to be careful of
Be careful driving, operating machinery or doing other jobs that require you to be alert while you are taking CLOFEN until you know how it affects you. This medicine may cause sleepiness and decreased alertness in some people, especially at the start of treatment.
Be careful when drinking alcohol while taking CLOFEN. The combination may make you feel more sleepy and less alert than usual.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking CLOFEN.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of these side effects.
Side effects occur mainly at the start of treatment or if the dose is too high or increased too quickly. They can often be relieved by lowering the dose.
If you are over 65 years of age, you should be especially careful while taking this medicine. Report any side effects promptly to your doctor. As people grow older, they are more likely to get side effects from medicines.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- daytime sleepiness or drowsiness
- tiredness, lack of energy
- dizziness, light-headedness
- spinning sensation (vertigo)
- mental confusion
- headache
- difficulty sleeping, nightmares
- nausea (feeling sick), vomiting, retching
- constipation, diarrhoea, stomach pain
- loss of appetite
- stuffy or blocked nose
- dry mouth
- change in sense of taste
- misuse, abuse and dependence
- suicide or suicide attempts
- numbness or tingling in hands and feet
- muscle weakness, spasms or pain
- problems with coordination and balance
- difficulty in speaking
- swelling of ankles due to fluid build-up
- blurred vision or double
- ringing in the ears
- frequent urination or bed-wetting
- excessive sweating
- weight gain
- impotence or inability to ejaculate
- increased blood sugar
- low body temperature
Tell your doctor immediately or go to Accident and Emergency at the nearest hospital if you notice any of the following:
- signs of an allergic reaction such as rash, itching or hives on the skin; swelling of the face, lips, tongue or other parts of the body; shortness of breath or wheezing
- slow or difficulty breathing
- irregular heart beat (fast or slow)
- chest pain
- uncontrollable muscle spasms affecting the eyes, head, neck or body
- fainting or seizures (fits)
- depression or other severe mood or mental changes
- hallucinations (hearing or seeing things that are not there)
- being unable to urinate or pain upon urinating; blood in the urine
- symptoms following sudden discontinuation of the medicine (drug withdrawal syndrome).
Refer to "Things you must not do" above
The above side effects could be serious. You may need medical attention.
Tell your doctor if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
After taking CLOFEN
Storage
Keep your tablets in the bottle until it is time to take them. If you take the tablets out of the bottle they may not keep well.
Keep your tablets in a cool dry place where the temperature stays below 25°C.
Do not store CLOFEN or any other medicine in the bathroom or near a sink. Do not leave it in the car or on window sills. Heat and dampness can destroy some medicines.
Keep Clofen where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine, or your tablets have passed their expiry date, ask your pharmacist what to do with any that are left over.
Product description
What it looks like
CLOFEN tablets are available in 2 strengths:
- CLOFEN 10 - round, white, scored tablet, marked "BN" over 00"10" on one side and "G" on the other.
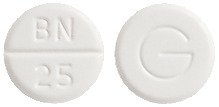
- CLOFEN 25 - round, white, scored tablet, marked "BN" over "25" on one side and "G" on the other.
Each bottle contains 100 tablets.
Ingredients
The active ingredient in CLOFEN is baclofen.
- Each CLOFEN 10 contains 10 mg of baclofen
- Each CLOFEN 25 contains 25 mg of baclofen
The tablets also contain the following inactive ingredients:
- lactose monohydrate
- microcrystalline cellulose
- calcium hydrogen phosphate
- sodium starch glycollate
- colloidal anhydrous silica
- magnesium stearate
The tablets are gluten free.
Manufacturer
Alphapharm Pty Ltd
Level 1, 30 The Bond
30 - 34 Hickson Road
Millers Point NSW 2000
www.mylan.com.au
Australian registration numbers:
CLOFEN 10 - AUST R 42146
CLOFEN 25 - AUST R 42147
- This leaflet was prepared in March 2020.
clofen_cmi\Mar20/00
Published by MIMS May 2020


 Chemical name: (3RS)-4-amino-3- (4-chlorophenyl) butanoic acid.
Chemical name: (3RS)-4-amino-3- (4-chlorophenyl) butanoic acid.