What is in this leaflet
This leaflet answers some common questions about CRESEMBA.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking CRESEMBA against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Read this leaflet carefully before taking CRESEMBA and keep this leaflet with the medicine. You may need to read it again.
What CRESEMBA is used for
This medicine used to treat fungal infections such as:
- Invasive aspergillosis (as-pur-ji-losis), an infection caused by a fungus called Aspergillus (as-pur-jilus).
- Mucormycosis (mu-cor-mi-cosis), an infection caused by a fungus called Mucorales (mu-cor-alus) where the treatment with amphotericin (am-fo-teri-cin) B is not appropriate.
This medicine belongs to a group of medicines called triazole antifungals.
This medicine works by preventing the growth of the fungal organisms causing your infection.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is not addictive.
This medicine is available only with a doctor's prescription.
This medicine may affect your ability to drive a car or operate machinery. You may feel confused, tired or sleepy.
There is not enough information to recommend the use of this medicine for children under the age of 18 years.
Before you start to use it
When you must not take it
Do not take CRESEMBA if you have an allergy to:
- any medicine containing isavuconazole (as isavuconazonium sulfate).
- any of the ingredients listed at the end of this leaflet.
- any other similar medicines such as ketoconazole, fluconazole, itraconazole, voriconazole or posaconazole .
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not take CRESEMBA if you are taking any of the following medicines:
- ketoconazole (e.g. Nizoral), a medicine used to treat fungal infections.
- ritonavir (e.g. Norvir, Kaletra), a medicine used to treat HIV infection where the dose is greater than 200 mg or more twice a day.
- rifabutin (e.g. Mycobutin), an antibiotic used to treat tuberculosis.
- rifampicin (e.g. Rifadin, Rimycin), a medicine used to treat tuberculosis and other infections.
- carbamazepine (e.g. Tegretol), a medicine used to treat epilepsy.
- long acting barbiturates such as phenobarbital, medicines used to treat severe insomnia and seizures.
- phenytoin (e.g. Dilantin), a medicine used to treat epilepsy.
- efavirenz (e.g. Stocrin) and etravine (e.g. Intelence), medicines used to treat HIV infection.
- St. John's Wort, a herbal medicine.
- nafcillin (e.g. Nafcil), a medicine used to bacterial infections.
Do not take this medicine if you have familial short QT syndrome (Disorder of the heart). It may cause your heart to beat irregularly.
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes or any other medicines, especially antifungal medicines such as itraconazole (Sporanox), fluconazole (Diflucan), posaconazole (Noxafil) or ketoconazole (Nizoral) (not all brands given).
Tell your doctor if you have or have had any of the following medical conditions:
- heart problems
- any problems affecting your liver
- any problems affecting your kidneys
Your doctor should do blood tests to check your liver before you start and while you are having treatment with CRESEMBA.
Tell your doctor if you are pregnant or plan to become pregnant. CRESEMBA should not be taken during pregnancy, unless indicated by your doctor. Effective contraception should be used in woman of childbearing potential. Your doctor can discuss with you the risks and benefits involved.
Tell your doctor if you are breast-feeding. CRESEMBA should not be taken whilst breastfeeding, unless indicated by your doctor. Your doctor can discuss with you the risks and benefits involved.
If you have not told your doctor about any of the above, tell your doctor before you start taking CRESEMBA.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines should not be taken with CRESEMBA. These include:
- ketoconazole (e.g Nizoral), a medicine used to treat fungal infections.
- ritonavir (e.g. Norvir, Kaletra), a medicine used to treat HIV infection where the dose is greater than 200 mg or more twice a day.
- rifabutin (e.g. Mycobutin), an antibiotic used to treat tuberculosis.
- rifampicin (e.g. Rifadin, Rimycin), a medicine used to treat tuberculosis and other infections.
- carbamazepine (e.g. Tegretol), a medicine used to treat epilepsy.
- long acting barbiturates such as phenobarbital, medicines used to treat severe insomnia and seizures.
- phenytoin (e.g. Dilantin), a medicine used to treat epilepsy.
- efavirenz (e.g. Stocrin) and etravine (e.g. Intelence), medicines used to treat HIV infection.
- St. John's Wort, a herbal medicine used to treat depression.
- nafcillin (e.g. Nafcil), a medicine used to bacterial infections.
Some medicines and CRESEMBA may interfere with each other. These include:
- rufinamide (e.g. Banzel), a medicine used to treat seizures.
- aprepitant (e.g. Emend), a medicine used to treat nausea and vomiting.
- prednisone (e.g. Panafcort), a steroidal medicine use to treat a variety of conditions.
- pioglitazone (e.g. Actos), a medicine use in the treatment of diabetes.
- ciclosporin (e.g Sandimmun), sirolimus (e.g. Rapamune) and tacrolimus (e.g. Prograf), medicines used to prevent organ transplant rejection or to treat certain problems with the immune system.
- digoxin (e.g. Lanoxin, Sigmaxin), medicine used to various heart conditions.
- colchicine (e.g. Colgout), a medicine used in the treatment of gout.
- dabigatran etexilate (e.g. Pradaxa), a medicine used in the treatment of blood clots (Deep Vein Thrombosis and Pulmonary Embolism).
- clarithromycin (e.g. Klacid) used to treat bacterial infections.
- saquinavir, indinavir, nevirapine, fosamprenavir, lopinavir/ritonavir combination (e.g. Invirase, Crixvan, Viramune, Kaletra, Telzir) used to treat HIV infection.
- alfentanil, fentanyl (e.g. Rapifen, Fentora, Sublimaze, Actiq, Abstral), used to treat strong pain.
- vincristine, vinblastine, used to treat cancer.
- mycophenolate mofetil (e.g. Cellcept), used in transplant patients.
- midazolam (e.g. Hypnovel), used to treat severe insomnia.
- bupropion (e.g. Zyban) used for smoking cessation and to treat depression.
- metformin (e.g. Diabex, Formet, Glucophage), a medicine used in the treatment of diabetes
- daunorubicin (e.g. Cerubidine), cyclophosphamide (e.g. Endoxan), doxorubicin (e.g. Adriamycin), irinotecan (e.g. Campto), topotecan (e.g. Hycamtin) and mitoxantrone (e.g. Onkotrone), medicines used in treating cancer.
- imatinib (e.g. Glivec) and lapatinib (e.g. Tyverb) medicines used in the treatment of cancer.
- lovastatin (e.g. Mevacor), a medicine used in the treatment of high cholesterol (Hypercholesterolaemia).
These medicines may be affected by CRESEMBA or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How to take CRESEMBA
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How much to take
Your doctor will tell you how much to take during the loading and maintenance phase.
Adults
The usual dose of CRESEMBA Capsules in adults is 200 mg (2 capsules) every 8 hours for 6 doses (First 48 hours - Starting Dose). Then 200 mg (2 capsules) ONCE daily thereafter (Usual Dose).
The usual dose of CRESEMBA IV Injection in adults is 200 mg (1 reconstituted vial, diluted further for IV infusion) every 8 hours for 6 doses (First 48 hours - Starting Dose). Then 200mg (1 reconstituted vial, diluted further for IV infusion) ONCE daily thereafter (Usual Dose).
How to take it
CRESEMBA needs to be taken regularly to be effective.
CRESEMBA Capsules
CRESEMBA can be taken with or without food.
CRESEMBA capsules should be swallowed whole with a full glass of water. Do not chew, crush, dissolve or open the capsules.
CRESEMBA IV Injection
CRESEMBA IV Injection is given as a slow injection into a vein (intravenous infusion). It must only be given by a doctor or trained nurse. It is a powder which is mixed with sterile water for injections and diluted further by your pharmacist, nurse or doctor.
When to take it
Take your medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
How long to take it
The length of time you take CRESEMBA will depend on the type of infection you have.
If you have weakened immune system or a difficult infection, you may need to be on CRESEMBA for longer periods of time (maybe longer than 6 months).
You doctor may switch you from CRESEMBA IV Injection to CRESEMBA Capsules once your condition improves.
Continue taking CRESEMBA for as long as your doctor tells you. Do not stop taking CRESEMBA because you are feeling better.
If you do not complete the full course prescribed by your doctor, the infection may not clear completely or your symptoms may return.
If you forget to take it
If it is nearly time to take your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose that you missed.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
CRESEMBA IV Injection will be given to you under close medical supervision. It is unlikely that a dose would be missed.
However, tell your doctor or pharmacist if you think that a dose has been forgotten.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have taken too much CRESEMBA. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
Symptoms of an overdose may include: headache, feeling dizzy, restless or sleepy, tingling, reduced sense of touch or sensation in the mouth, problems being aware of things, hot flushes, anxiety, joint pain, changes in the way things taste, dry mouth, diarrhoea, vomiting, feeling your heart beat, faster heart rate, being more sensitive to light.
While you are using CRESEMBA
Things you must do
Stop taking CRESEMBA and tell your doctor immediately if you develop: a rash or blisters, swelling of your lips, mouth, tongue or throat with difficulty breathing - these may be signs of an allergic reaction while taking CRESEMBA.
If the symptoms of your infection do not improve within few days or if they become worse, tell your doctor.
Make sure you follow your doctor's instructions and keep all appointments, including blood tests. Your doctor should monitor the function of your liver and kidneys using blood tests.
If you are about to be started on any medicine, remind your doctor and pharmacists who treat you that you are taking CRESEMBA.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
If you become pregnant while taking this medicine, tell your doctor immediately.
If you are about to have any blood tests, tell your doctor that you are taking this medicine. It may interfere with the results of some tests.
Keep all of your doctor's appointments so that your progress can be checked.
Things you must not do
Do not take CRESEMBA to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop taking your medicine or lower the dosage without checking with your doctor. If you stop taking it suddenly or not complete the course as prescribed by your doctor, your condition may worsen or your symptoms may return.
Things to be careful of
Be careful driving or operating machinery until you know how CRESEMBA affects you. This medicine may cause confusion, tiredness and sleepiness in some people.
If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking CRESEMBA.
This medicine helps most people with fungal infections, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor as soon as possible if you notice any of the following:
- low potassium in your blood,
- decreased appetite,
- hallucinations (delirium),
- headache,
- sleepiness,
- inflamed veins that could lead to blood clots,
- shortness of breath or sudden and severe difficulty breathing
- feeling sick (nausea), being sick (vomiting), diarrhoea, stomach pain,
- changes in blood tests of liver function,
- rash, itching,
- kidney failure,
- chest pain, feeling tired or sleepy.
The above list includes the more common side effects of your medicine.
Tell your doctor as soon as possible if you notice any of the following:
- reduced white blood cells - can increase your risk of infection and fever,
- reduced blood cells called 'platelets' - can increase your risk for bleeding or bruising,
- reduced red blood cells - can make you feel weak or short of breath or make your skin pale,
- severe reduction in blood cells - can make you feel weak, cause bruising or make infections more likely,
- rash, swelling of your lips, mouth, tongue or throat with difficulty breathing (hypersensitivity),
- low blood sugar levels,
- low blood levels of magnesium,
- low levels in the blood of a protein called 'albumin',
- not getting the right goodness from your diet (malnutrition),
- depression, difficulty sleeping,
- seizure, fainting or feeling faint, dizziness,
- sensation of tingling, tickling, or pricking of the skin (paraesthesia),
- altered mental state (encephalopathy),
- changes in taste (dysgeusia),
- feeling of 'spinning' or being dizzy (vertigo),
- heart beat problems - may be too fast or uneven, or extra heart beats – this may show in your heart tracing (electrocardiogram or ECG),
- problems with the blood circulation,
- low blood pressure,
- wheezing, very fast breathing, coughing up blood or blood-stained sputum, nose bleeding,
- indigestion,
- constipation,
- feeling bloated (abdominal distension),
- enlarged liver,
- inflammation of the liver,
- problems with the skin, red or purple spots on the skin (petechiae), inflamed skin (dermatitis), hair loss,
- back pain,
- feeling weak, very tired, or sleepy or generally out of sorts (malaise).
The above list includes serious side effects that may require medical attention.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- swelling of the face, lips or tongue which may cause difficulty in swallowing or breathing
- sudden and severe difficulty breathing
- fainting, confusion, seizures or fits
- sudden or severe itching, skin rash, hives or blisters
- signs of kidney failure such as tiredness, lack of appetite and reduced or greatly increased amount of urine
These may be signs of a serious allergic reaction or side effect. You may need urgent medical attention or hospitalisation. These side effects are rare.
Tell your doctor if you notice any other side effects.
Other side effects not listed above may also occur in some people.
After using CRESEMBA
Storage
CRESEMBA Capsules
Keep your capsules in the original pack until it is time to take them. If you take the capsules out of the pack they may not keep well.
Keep your capsules in a cool dry place where the temperature stays below 25°C.
Do not store CRESEMBA or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep your medicine where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
CRESEMBA IV Injection
CRESEMBA IV Injection will be stored in the pharmacy or on the hospital ward. It is kept in a refrigerator where the temperature stays between 2-8°C.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
CRESEMBA Capsules
CRESEMBA Capsules come in one strength, 100 mg.
The capsules have a reddish-brown body marked with '100' in black ink and a white cap marked with "C" in black ink.
CRESEMBA Capsules 100 mg are available in cartons that contain 14 capsules. Each carton contains 2 aluminium/aluminium blisters pack, with 7 capsules each.
Each capsule pocket is connected to a pocket that contains 'desiccant' to protect the capsule from moisture.
Do not puncture the blister containing the desiccant.
Do not swallow or use the desiccant.
CRESEMBA Powder for Injection
CRESEMBA IV comes as a white to yellow powder in a clear glass vial with a rubber stopper and an aluminium cap with a plastic seal which contains 200 mg isavuconazole.
Ingredients
Active Ingredients
CRESEMBA Capsules 100 mg contains 100 mg of isavuconazole (as 186.3 mg isavuconazonium sulfate) as the active ingredient.
CRESEMBA IV Injection 200 mg contains 200 mg of isavuconazole (as 372.6 mg isavuconazonium sulfate) as the active ingredient.
Inactive Ingredients
CRESEMBA Capsules 100 mg contains the following other ingredients:
Capsule Contents
- magnesium citrate
- microcrystalline cellulose
- purified talc
- colloidal anhydrous silica
- stearic acid
Capsule Shell
- hypromellose
- purified water
- iron oxide red (E172) (capsule body only)
- titanium dioxide (E171)
- gellan gum
- potassium acetate
- disodium edetate
- sodium lauryl sulfate
Printing Ink
- shellac
- propylene glycol
- potassium hydroxide
- iron oxide black (E172)
CRESEMBA IV Injection 200 mg contains the following other ingredients:
- mannitol
- sulfuric acid (for pH-adjustment)
Supplier
CRESEMBA is supplied in Australia by:
Pfizer Australia Pty Ltd
Sydney NSW
Toll Free Number: 1800 675 229
www.pfizer.com.au
Australian Registration Numbers
CRESEMBA Capsules 100 mg AUST R 305452
CRESEMBA IV Injection 200 mg AUST R 305480
Date of Preparation
This leaflet was prepared in January 2022
® = Registered Trademark
© Pfizer Australia Pty Ltd
Published by MIMS March 2022



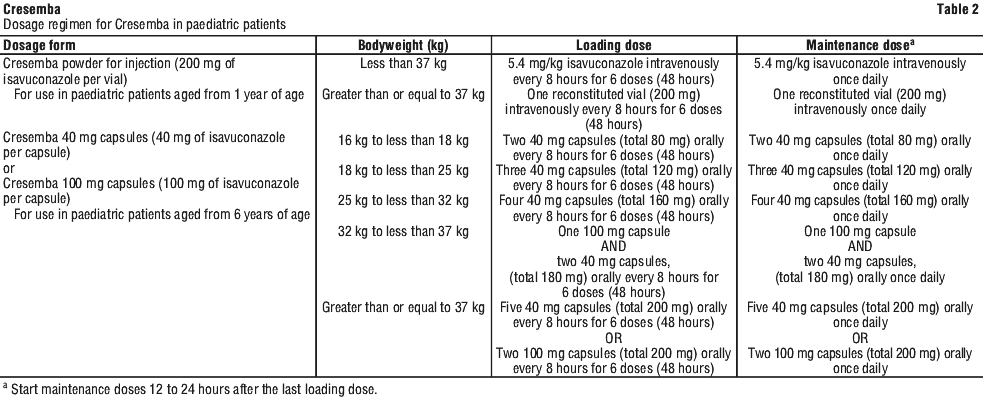 The maximum of any individual loading or daily maintenance dose to be administered to any paediatric patient is 200 mg isavuconazole.
The maximum of any individual loading or daily maintenance dose to be administered to any paediatric patient is 200 mg isavuconazole.




 There are currently insufficient data to set clinical breakpoints for other Aspergillus species or for any Mucorales species.
There are currently insufficient data to set clinical breakpoints for other Aspergillus species or for any Mucorales species. Overall success at end-of-treatment (EOT) was assessed by a blinded, independent Data Review Committee (DRC) using pre-specified clinical, mycological, and radiological criteria. In the subgroup of patients with proven or probable invasive aspergillosis confirmed by serology, culture or histology, overall success at EOT was seen in 35% of isavuconazole-treated patients compared to 38.9% of voriconazole-treated patients (see Table 8).
Overall success at end-of-treatment (EOT) was assessed by a blinded, independent Data Review Committee (DRC) using pre-specified clinical, mycological, and radiological criteria. In the subgroup of patients with proven or probable invasive aspergillosis confirmed by serology, culture or histology, overall success at EOT was seen in 35% of isavuconazole-treated patients compared to 38.9% of voriconazole-treated patients (see Table 8).
 Patients were treated with isavuconazole intravenously or via oral administration at the recommended doses. Median treatment duration was 102 days for patients classified as primary, 33 days for refractory, and 85 days for intolerant (see Section 4.2 Dose and Method of Administration).
Patients were treated with isavuconazole intravenously or via oral administration at the recommended doses. Median treatment duration was 102 days for patients classified as primary, 33 days for refractory, and 85 days for intolerant (see Section 4.2 Dose and Method of Administration).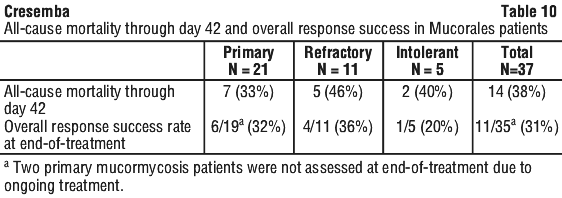
 As shown in Table 12, the absolute bioavailability of isavuconazole following oral administration of a single dose of Cresemba is 98%. Based on these findings, intravenous and oral dosing can be used interchangeably.
As shown in Table 12, the absolute bioavailability of isavuconazole following oral administration of a single dose of Cresemba is 98%. Based on these findings, intravenous and oral dosing can be used interchangeably.
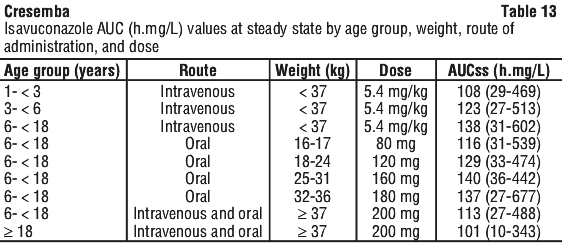 The predicted exposures for paediatric patients, regardless of route of administration and age group, were comparable to exposures at steady-state (AUCss) from a clinical study conducted in adult patients with infections caused by Aspergillus species and other filamentous fungi (mean AUCss = 101.2 h.mg/L with standard deviation (SD) = 55.9, see Table 13).
The predicted exposures for paediatric patients, regardless of route of administration and age group, were comparable to exposures at steady-state (AUCss) from a clinical study conducted in adult patients with infections caused by Aspergillus species and other filamentous fungi (mean AUCss = 101.2 h.mg/L with standard deviation (SD) = 55.9, see Table 13).
