What is in this leaflet
This leaflet answers some of the common questions people ask about CRESTOR. It does not contain all the information that is known about CRESTOR.
It does not take the place of talking to your doctor and pharmacist.
All medicines have risks and benefits. Your doctor will have weighed the risks of you taking CRESTOR against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What CRESTOR is for
CRESTOR is used to lower high cholesterol levels.
Even though you may have normal cholesterol, CRESTOR can also be used to reduce the risk of you having a stroke or heart attack if you are a man 50 or more years old or a women 60 or more years old and have at least 2 risk factors for having a heart attack or stroke, such as high blood pressure, low levels of good cholesterol (HDL), smoking or a family history of premature coronary heart disease. Your doctor may also do a blood test to measure a substance called C Reactive Protein to help decide if you should be given CRESTOR for this use.
Cholesterol and triglycerides
Everyone has cholesterol and triglycerides in their blood. They are fatty substances needed by the body for many things.
Triglycerides are an energy source for the body. Cholesterol is used for such things as building cells, making bile acids (which help to digest foods) and making some hormones.
There are different types of cholesterol. Too much of the "bad" cholesterol (LDL) can block the blood vessels that supply your heart and brain with blood, and can cause heart attack, angina and stroke. The "good" cholesterol (HDL) helps to remove the bad cholesterol from the blood vessels. High levels of triglycerides can be associated with a low level of "good" cholesterol and may increase the risk of heart disease.
How CRESTOR works
CRESTOR belongs to a group of medicines known as HMG-CoA reductase inhibitors (also known as 'statins'). It lowers the "bad" cholesterol, and raises the "good" cholesterol when exercise and changes to diet are not enough on their own.
Cholesterol is present in many foods and is also made by your body. CRESTOR does not reduce the cholesterol that comes from fat in food. Because of this, when you are taking CRESTOR, you need to follow a low-fat diet, control your weight and exercise regularly.
High cholesterol is also more likely to occur with certain diseases or if you have a family history of high cholesterol.
Your doctor will have explained why you are being treated with CRESTOR and told you what dose to take. Your doctor may need to check your cholesterol levels before prescribing CRESTOR or changing your dose.
Follow all directions given to you by your doctor carefully. They may differ from the information contained in this leaflet.
Your doctor may prescribe this medicine for another use. Ask your doctor if you want more information.
CRESTOR is not addictive.
CRESTOR is not recommended for use in children as its effects in children have not been established.
Before you use CRESTOR
When you must not use it
Do not use CRESTOR if you are pregnant or intend to become pregnant. Ask your doctor about effective methods of contraception.
If you become pregnant, stop taking CRESTOR as soon as you find out and see your doctor immediately.
Do not use CRESTOR if you are breast feeding. We do not know if your baby can take in CRESTOR from breast milk if you are breast feeding.
Do not use CRESTOR if you have active liver disease or if tests show you have elevated levels of liver enzymes which may show that you have a problem with your liver.
Do not use CRESTOR 40 mg if you have:
- low thyroid hormone levels (hypothyroidism)
- a personal or family history of hereditary muscular disorders
- a previous history of muscular problems from using other lipid-lowering agents
- a history of very heavy alcohol use
- Asian heritage
- been prescribed another class of lipid lowering agent called a fibrate
- been prescribed any medicine containing fusidic acid
- severe kidney impairment
- situations that may increase CRESTOR blood levels
Do not use after the use by (expiry) date printed on the pack. It may have no effect at all, or worse, an entirely unexpected effect if you take it after the expiry date.
Do not use CRESTOR if the packaging is torn or shows signs of tampering.
Do not use it to treat any other complaints unless your doctor tells you to. Do not give this medicine to anyone else.
Before you start to use it
You must tell your doctor if:
- you have any allergies to
- any other statins such as simvastatin (e.g. ZOCOR, LIPEX); pravastatin (e.g. PRAVACHOL); atorvastatin (e.g. LIPITOR); fluvastatin (e.g. VASTIN)
- any ingredients listed at the end of this leaflet
If you have an allergic reaction, you may get a skin rash, hay fever, difficulty in breathing or feel faint.
- you have any of these medical conditions
- liver problems
- kidney problems
- low thyroid hormone levels (hypothyroidism)
- a personal or family history of muscle disorders
- a history of muscle problems from using other lipid-lowering agents
It may not be safe for you to take CRESTOR if you have any of these conditions. Your doctor may do a blood test to check if you have any problems, and may adjust the dose of CRESTOR.
- you have any unexplained aches or pains in your muscles
- you regularly drink large amounts of alcohol
Excessive alcohol consumption may not be safe in patients taking CRESTOR.
Taking other medicines
Tell your doctor if you are taking any other medicines including
- ciclosporin (e.g. SANDIMMUN and NEORAL), used for example, after organ transplant
- antacids (medicines used to treat heartburn and indigestion). CRESTOR can be taken 2 hours before or 2 hours after taking an antacid
- warfarin (e.g. COUMADIN and MAREVAN), used to stop blood clots
- clopidogrel (e.g. PLAVIX), a medicine used to prevent blood clots
- gemfibrozil (e.g. LOPID, JEZIL, and AUSGEM), used to lower blood lipids
- fusidic acid (eg FUCIDIN) used to treat some infections
- various protease inhibitors used in combination with ritonavir to treat HIV infection (e.g. KALETRA)
- simeprevir (OLYSIO), a medicine used for treatment of chronic hepatitis C
- eltrombopag (REVOLADE), used to increase your platelet count in your blood
- medicines that you buy at the chemist, supermarket or health food shop, including herbal medicines.
Your doctor will consider if CRESTOR should be used together with any of these medicines, or may wish to adjust the dose of CRESTOR or the other medicines. These medicines may affect the way CRESTOR works.
Your doctor or pharmacist can tell you what to do if you are taking any of these medicines.
If you have not told your doctor about any of these things, tell them before you take CRESTOR.
Effects on driving and using machinery
Be careful driving a car or operating machinery until you know if CRESTOR affects you. CRESTOR generally does not cause any problems with your ability to drive a car or operate machinery. However, as with many other medicines, CRESTOR may cause dizziness in some people.
Using CRESTOR
How to take it
Depending on your condition and ethnic background, your doctor will decide the most appropriate starting dose for you.
If you have high cholesterol, your doctor will probably start you on 5 mg or 10 mg tablet taken once daily. Your doctor will then monitor your cholesterol and triglyceride levels during your treatment, and, if needed, may increase your CRESTOR dose to 20 mg once daily. For most patients a maximum 20 mg CRESTOR daily dose is sufficient to treat high cholesterol.
A small number of patients may need to further increase their CRESTOR dose to 40 mg once daily, for example patients whose high cholesterol is hereditary.
If your cholesterol is not high but you have risks for having a heart attack or stroke, your doctor may start you on 20 mg.
Your doctor will advise you on the dose that's right for your condition. The daily dose of CRESTOR must not exceed 40 mg daily.
DO NOT INCREASE OR ADJUST YOUR CRESTOR DOSE YOURSELF.
Take CRESTOR once a day, at about the same time each day. Keeping a regular time for taking CRESTOR will help to remind you to take it.
Swallow each tablet whole with a drink of water.
CRESTOR can be taken at any time of the day. It does not matter whether you take CRESTOR with food or on an empty stomach.
While taking CRESTOR you also need to follow a low-fat diet, control your weight and exercise regularly.
How long to take it
You must continue to take it as directed.
CRESTOR helps lower your blood cholesterol and triglycerides. It does not cure your condition. If you stop taking CRESTOR, your cholesterol and triglycerides levels may rise again.
You may have to take cholesterol-lowering medicines for the rest of your life.
If you forget to take it
If you forget to take a dose of CRESTOR, take it as soon as you remember, as long as it is more than 6 hours before your next dose is due. Otherwise, wait until your next dose is due and take it as normal.
Do not double the dose to make up for the one you missed.
If you have trouble remembering when to take your medicine, ask your pharmacist for some hints.
Overdose
Telephone your doctor or the Poisons Information Centre (13 11 26), or go to Accident and Emergency at your nearest hospital immediately if you think that you or anyone else may have taken too much CRESTOR even if there are no signs of discomfort or poisoning.
While you are using it
Things you must do
Have your blood cholesterol and triglycerides checked when your doctor says so to make sure CRESTOR is working.
Stop taking CRESTOR and contact your doctor immediately if you become pregnant while you are taking CRESTOR.
Things you must not do
Do not stop taking CRESTOR unless you have discussed it with your doctor.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking CRESTOR.
CRESTOR helps most people with too much cholesterol, but it may have unwanted side effects in a few people.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- headache
- constipation
- dizziness
- nausea (feeling sick)
- stomach pain
- unusual tiredness
- itchy skin
- memory loss
- stiff or painful joints (arthralgia)
These side effects are usually mild.
Tell your doctor if you notice a significant increase in your need to urinate or if you are significantly more hungry or thirsty than usual.
Tell your doctor immediately or go to Accident and Emergency at your nearest hospital if you notice any of the following:
- aching muscles, muscle tenderness or weakness not caused by exercise, particularly if you also have a fever or generally feel unwell
- difficulty breathing, swelling of the face, eyelids or lips
- difficulty breathing, coughing, particularly if you also feel generally unwell (e.g. fatigue, weight loss, fever).
These are all serious side effects. You may need urgent medical attention
Serious side effects are rare.
Tell your doctor if you notice anything else that is making you feel unwell. Some people may get other side effects while taking CRESTOR.
Do not be alarmed by this list of side effects. You may not experience any of them.
After using it
Storage
Keep your tablets in the blister pack until it is time to take them. If you take CRESTOR out of the blister pack it will not keep well.
Keep it in a cool dry place where the temperature stays below 30°C.
Do not store it or any other medicine in the bathroom or near a sink. Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it. A locked cupboard, at least one-and-a-half metres above the ground, is a good place to store medicines.
Do not leave it in the car on hot days.
Disposal
Ask your pharmacist what to do with any CRESTOR tablets you have left over if your doctor tells you to stop taking them, or you find that the expiry date has passed.
Product description
What CRESTOR looks like
CRESTOR 5 mg are yellow, round, film-coated, biconvex tablets engraved "ZD4522 5" on one side.
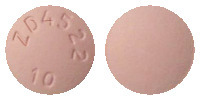
CRESTOR 10 mg are pink, round, film-coated, biconvex tablets engraved "ZD4522 10" on one side.
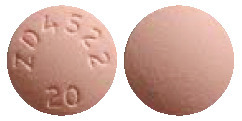
CRESTOR 20 mg are pink, round, film-coated, biconvex tablets engraved "ZD4522 20" on one side.
CRESTOR 40 mg are pink, oval, film-coated, biconvex tablets, engraved "ZD4522" on one side and "40" on the other side.
CRESTOR tablets are available in blister packs of 30 tablets.
Ingredients:
CRESTOR contains rosuvastatin calcium as the active ingredient, equivalent to rosuvastatin 5 mg, 10 mg, 20 mg and 40 mg plus:
Lactose monohydrate
Microcrystalline cellulose
Calcium phosphate
Crospovidone
Magnesium stearate
Hypromellose
Triacetin
The 5 mg tablets are coloured with titanium dioxide and iron oxide yellow.
The 10 mg, 20 mg and 40 mg tablets are coloured with titanium dioxide and iron oxide red.
Manufacturer
CRESTOR is supplied in Australia by:
A. Menarini Australia Pty Ltd
Level 8, 67 Albert Avenue
Chatswood NSW 2067
Telephone: 1800 644 542
Australian Registration Numbers:
CRESTOR 5 mg - AUST R 119120
CRESTOR 10 mg - AUST R 119128
CRESTOR 20 mg - AUST R 119130
CRESTOR 40 mg - AUST R 119135
This leaflet was prepared in June 2021.
® CRESTOR is a trade mark of A. Menarini Australia Pty Ltd in Australia and New Zealand
For the most up to date version of this leaflet, please go to www.menarini.com.au/cmi
[vA04-0]
Published by MIMS August 2021





 The percent change from baseline in HDL-C at week 6 is shown in Figure 2.
The percent change from baseline in HDL-C at week 6 is shown in Figure 2. The mean percent change in HDL-C from baseline to week 6 for each statin treatment group represented in Figure 2 is summarised with 95% CI in Table 3.
The mean percent change in HDL-C from baseline to week 6 for each statin treatment group represented in Figure 2 is summarised with 95% CI in Table 3. Table 4 summarises the pooled lipid variable data for rosuvastatin 5 and 10 mg from 5 Phase III efficacy trials (Trials 24-28).
Table 4 summarises the pooled lipid variable data for rosuvastatin 5 and 10 mg from 5 Phase III efficacy trials (Trials 24-28).


 There were no statistically significant reductions in the rate of noncardiovascular death or the incidence of bone fractures in the rosuvastatin treated group compared to placebo.
There were no statistically significant reductions in the rate of noncardiovascular death or the incidence of bone fractures in the rosuvastatin treated group compared to placebo. The individual components of the primary end point are presented in Figure 4. Rosuvastatin significantly reduced the risk of nonfatal myocardial infarction, nonfatal stroke, and arterial revascularisation procedures. There were no significant treatment differences between the rosuvastatin and placebo groups for death due to cardiovascular causes or hospitalisations for unstable angina.
The individual components of the primary end point are presented in Figure 4. Rosuvastatin significantly reduced the risk of nonfatal myocardial infarction, nonfatal stroke, and arterial revascularisation procedures. There were no significant treatment differences between the rosuvastatin and placebo groups for death due to cardiovascular causes or hospitalisations for unstable angina. In a post hoc subgroup analysis of JUPITER subjects (n = 1405; rosuvastatin = 725, placebo = 680) with a hsCRP ≥ 2 mg/L and no other traditional risk factors (smoking, BP ≥ 140/90 or taking antihypertensives, low HDL-C) other than age, after adjustment for high HDL-C, there was no significant treatment benefit with rosuvastatin treatment.
In a post hoc subgroup analysis of JUPITER subjects (n = 1405; rosuvastatin = 725, placebo = 680) with a hsCRP ≥ 2 mg/L and no other traditional risk factors (smoking, BP ≥ 140/90 or taking antihypertensives, low HDL-C) other than age, after adjustment for high HDL-C, there was no significant treatment benefit with rosuvastatin treatment.
