What is in this leaflet
This leaflet answers some common questions about CRINONE. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using CRINONE against the benefits this medicine is expected to have for you.
If you have any questions about using this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What CRINONE is used for
CRINONE is a vaginal gel that contains natural progesterone.
Progesterone is a hormone that is produced by the ovaries during the second half of the menstrual cycle.
It is an essential hormone for preparing the uterus (womb) for pregnancy and on-going support of pregnancy.
The lack of normal progesterone production by the ovaries in the second half of the menstrual cycle is called luteal phase defect. Women who have this defect may have difficulties in becoming pregnant and may have a higher risk of miscarriage.
CRINONE is used to supplement progesterone in women who have luteal phase defect. CRINONE is also used to supplement or replace your own natural progesterone in Assisted Reproductive Technology procedures (e.g. IVF).
Your doctor may have prescribed CRINONE for another purpose.
Ask your doctor if you have any questions why CRINONE has been prescribed for you.
This medicine is available only with a doctor's prescription.
CRINONE is not addictive.
Before you use CRINONE
When you must not use it:
Do not use CRINONE if:
- you have ever had an allergic reaction to progesterone or to any of the ingredients listed at the end of this leaflet
Some of the symptoms of an allergic reaction may include:
- rash (itching or hives) on the skin
- swelling of the face, lips, tongue or other parts of the body
- shortness of breath, wheezing or troubled breathing or a tight feeling in your chest
Do not use CRINONE if you have, or have had any of the following conditions:
- cancer of the breast and genital organs
- porphyria
- you have abnormal vaginal bleeding that has not been diagnosed, or is due to a recent or current missed abortion or miscarriage
- you have or have ever had blood clots or a stroke
- you have liver disease or abnormal laboratory results
Do not use CRINONE if you are breastfeeding.
Do not use CRINONE after the expiry date printed on the label of the pre-filled applicator and the carton. If you are taking this medicine after the expiry date has passed, it may not work.
Do not use CRINONE if the packaging is torn or shows signs of tampering.
Do not use CRINONE if the product appears to be discoloured, or the product does not look quite right.
If it has expired or is damaged, return it to your pharmacist or doctor for disposal.
If you are not sure whether you should start using CRINONE, talk to your doctor or pharmacist.
If you have not told your doctor about any of the above, tell them before you start using CRINONE.
CRINONE is not for use in children.
Do not use CRINONE to treat other complaints unless your doctor says it is safe.
Do not give this medicine to anyone else.
Before you use it
Tell your doctor or pharmacist if you have allergies to:
- any other medicines
- any other substances, such as foods, preservatives or dyes
See your doctor for a check-up before you start to use CRINONE. The check-up should include a Pap smear, pelvic examination and breast examination.
You must tell your doctor or pharmacist if:
- you are breast feeding
- you are or have been very depressed
- you are using other vaginal medications
- you suffer from migraines, epilepsy or asthma
- you suffer from heart, kidney or liver disease
If in doubt, consult your doctor or pharmacist before using CRINONE.
Taking other medicines
Tell your doctor if you are taking any other medicines, including medicines you buy without a prescription from a pharmacy, supermarket or health food shop.
CRINONE should not be used at the same time as other vaginal preparations.
If you forget to use it
If you forget a dose of CRINONE, use it as soon as you remember, and then continue on the same schedule as before.
Do not use more than the recommended daily dose.
If you use too much (overdose)
Immediately contact your doctor or the Poisons Information Centre (in Australia telephone 131 126, in New Zealand telephone 0800 764 766) if you are concerned that you have used too much CRINONE.
While you are using CRINONE
Things you must do:
Be sure to keep all your doctor's appointments so your progress can be checked.
Tell any other doctors or pharmacists you visit that you are using CRINONE.
If you are about to start taking any new medicines, tell the doctor or pharmacist that you are using CRINONE.
Things you must not do:
Do not give this medicine to anyone else; it may harm them even if the reasons for using it seem to be the same as yours.
Things to be careful of:
Be careful driving or operating machinery until you know how CRINONE affects you. Some people feel drowsy and sleepy when using CRINONE.
If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
Side Effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using CRINONE.
CRINONE helps most women with a lack of progesterone, but it may have unwanted side effects in a few people.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Tell your doctor if you notice any of the following side effects when using CRINONE:
Very common side effects
- cramps, abdominal pain, perineal pain (around the genitals & the back passage)
- headache
- breast enlargement or breast pain
- feelings of severe sadness and unworthiness, decreased sexual drive, sleepiness, feeling emotional
- constipation, nausea
- passing urine at night
Common side effects
- bloating, pain
- dizziness
- vaginal discharge, itching of the vaginal area, vaginal thrush
- diarrhoea, vomiting
- painful sexual intercourse
- painful joints
Other side effects may include vaginal spotting, vaginal irritation, mild application site reactions and hypersensitivity reactions manifesting as skin rash.
This is not a complete list of all possible side effects. Others may occur in some people and there may be some side effects not yet known.
Tell your doctor if you notice anything else that is making you feel unwell, even if it is not on this list.
Ask your doctor or pharmacist if you do not understand anything in this list.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
After using CRINONE
Storage
Keep this medicine where young children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Keep CRINONE in a cool place where it stays below 25°C and is not exposed to extreme heat or cold.
Do not store it, or any other medicine, in a bathroom or near a sink.
Do not leave it in the car. Heat and dampness can destroy some medicines.
Each applicator must be used once only. Use a new applicator for each treatment.
Disposal
If your doctor tells you to stop using CRINONE, or the gel has passed its expiry date, ask the pharmacist what to do with any leftover drug.
Product description
What it looks like
CRINONE is a soft, white to off-white gel. It is supplied in an applicator with a twist-off top. The applicator contains a plastic ball. Each applicator is sealed in a wrapper and contains a single dose of CRINONE.
Each applicator contains 1.45 g of gel and delivers a constant dose of 1.125 g of CRINONE gel containing 90 mg (8% gel) of progesterone.
CRINONE is available in boxes containing 6, 15 or 18 applicators*.
* Not all pack sizes may be available.
Ingredients
Active ingredients:
Progesterone
Other ingredients:
Hydrogenated palm glycerides,
Glycerol,
Carbomer 974P,
Sorbic acid,
Polycarbophil,
Sodium hydroxide,
Purified water,
Light liquid paraffin.
Australian Registration number
AUST R 83166
Supplier
CRINONE is supplied in Australia by:
Merck Healthcare Pty Ltd
Suite 1, Level 1, Building B
11 Talavera Road
Macquarie Park NSW 2113
E-mail: [email protected]
Phone: 1800 633 463
CRINONE is supplied in New Zealand by:
Healthcare Logistics
58 Richard Pearse Drive
Airport Oaks
Auckland
E-mail: [email protected]
Phone: 0800 426 252
This leaflet was prepared in March 2020.
How to use CRINONE
CRINONE is applied directly from the applicator deep into the vagina. The applicator is designed to deliver a pre-measured dose of progesterone gel. A small amount of gel will be left in the applicator after usage. Do not be concerned because you will still be receiving the required dose.
How much to use and how often to use it
The amount of CRINONE used depends on the condition being treated.
Your doctor may prescribe one applicator of CRINONE either daily or twice daily and will advise you when to start treatment. If pregnancy occurs treatment may continue for up to 12 weeks or as directed by your doctor.
CRINONE should be used at the same time each day. If used twice a day, one applicator of CRINONE should be used in the morning and the other at night.
How to use CRINONE
Follow these steps carefully or as instructed by your doctor:
- Wash your hands well with soap and water.
- Remove the applicator from the sealed wrapper.
- DO NOT REMOVE THE TWIST-OFF TAB AT THIS TIME.
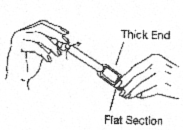
- Hold the applicator by the thick end. Shake down several times like a thermometer to ensure that the contents are at the thin end.
- The plastic ball in the thin end helps the gel to move down to the tip of the applicator. The ball also helps to control the release of the gel. The applicator is designed so that the ball cannot exit it.
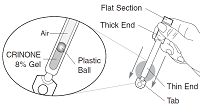
- Hold the applicator by the flat section of the thick end. Twist off the tab at the thin end of the applicator and discard it.
DO NOT squeeze the thick end while twisting off the tab. This may release gel.
- Carefully insert the applicator into the vagina while you are in a sitting position or when lying on your back with the knees bent. Gently insert the thin end of the applicator well into the vagina.
- Squeeze the thick end of the applicator firmly to push the gel into the vagina. Remove the applicator and discard into a waste container. There may be some gel left in the applicator. Do not worry; you will still be receiving the correct dose.
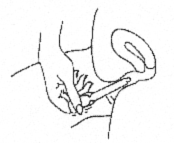
CRINONE coats the vaginal lining to provide long-lasting release of progesterone.
Published by MIMS July 2020

 In a further study, comprised two crossover comparisons of progesterone pharmacokinetics in 20 healthy postmenopausal women, 10 of the subjects received a dose of 45 mg, on two different occasions; once as intravaginal gel (Crinone 4%) and on the other as an intramuscular injection (IMI). The other 10 subjects received a dose of 90 mg by each route. The pharmacokinetic parameters are shown in Table 2.
In a further study, comprised two crossover comparisons of progesterone pharmacokinetics in 20 healthy postmenopausal women, 10 of the subjects received a dose of 45 mg, on two different occasions; once as intravaginal gel (Crinone 4%) and on the other as an intramuscular injection (IMI). The other 10 subjects received a dose of 90 mg by each route. The pharmacokinetic parameters are shown in Table 2. These pharmacokinetic parameters show that for the 45 mg and 90 mg dose the gel is 28% and 20% bioavailable in comparison with the IMI route, respectively.
These pharmacokinetic parameters show that for the 45 mg and 90 mg dose the gel is 28% and 20% bioavailable in comparison with the IMI route, respectively. Chemical formula: C21H30O2. Molecular weight: 314.5. Melting range: 127-131°C.
Chemical formula: C21H30O2. Molecular weight: 314.5. Melting range: 127-131°C.